Advanced Palladium-Catalyzed Carbonylation for High-Purity 3-Benzylidene-2,3-dihydroquinolone Manufacturing
Advanced Palladium-Catalyzed Carbonylation for High-Purity 3-Benzylidene-2,3-dihydroquinolone Manufacturing
The pharmaceutical industry continuously seeks robust and efficient synthetic routes for nitrogen-containing heterocycles, particularly those serving as core scaffolds for bioactive molecules. Patent CN113735826A introduces a groundbreaking preparation method for 3-benzylidene-2,3-dihydroquinolone compounds, a structural motif prevalent in potent analgesic and antitumor agents. As illustrated in the biological context, these quinolone derivatives form the backbone of significant therapeutic candidates, such as analgesic agent A and antitumor agent B, highlighting their critical role in modern drug discovery pipelines. 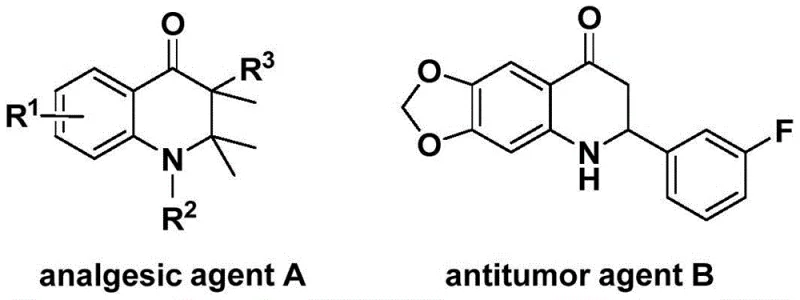 . This novel methodology leverages a palladium-catalyzed carbonylation reaction, utilizing N-pyridinesulfonyl-o-iodoaniline and allenes as primary building blocks. By replacing hazardous gaseous carbon monoxide with a solid surrogate, this process not only enhances laboratory safety but also streamlines the operational workflow, making it an attractive option for reliable pharmaceutical intermediate suppliers aiming to optimize their manufacturing portfolios.
. This novel methodology leverages a palladium-catalyzed carbonylation reaction, utilizing N-pyridinesulfonyl-o-iodoaniline and allenes as primary building blocks. By replacing hazardous gaseous carbon monoxide with a solid surrogate, this process not only enhances laboratory safety but also streamlines the operational workflow, making it an attractive option for reliable pharmaceutical intermediate suppliers aiming to optimize their manufacturing portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 2,3-dihydroquinolone skeleton has relied on classical cyclization strategies that often suffer from significant drawbacks regarding efficiency and scalability. Traditional approaches frequently necessitate harsh reaction conditions, including extreme temperatures or the use of highly toxic reagents, which pose severe challenges for environmental compliance and operator safety in a commercial setting. Furthermore, many existing protocols exhibit poor functional group tolerance, limiting the diversity of substituents that can be introduced onto the aromatic rings without compromising the reaction yield. This lack of versatility forces R&D teams to develop lengthy, multi-step protection and de-protection sequences, drastically increasing the overall cost of goods and extending the lead time for high-purity pharmaceutical intermediates. Additionally, the direct use of carbon monoxide gas in carbonylation reactions requires specialized high-pressure equipment and rigorous safety protocols, creating a substantial barrier to entry for many contract manufacturing organizations.
The Novel Approach
In stark contrast, the technology disclosed in CN113735826A offers a transformative solution by employing a mild, palladium-catalyzed carbonylation strategy that operates under atmospheric pressure conditions. This innovative route utilizes 1,3,5-trimesic acid phenol ester (TFBen) as a safe and effective carbon monoxide substitute, thereby eliminating the need for handling toxic CO gas while maintaining high reaction efficiency. The process demonstrates exceptional substrate compatibility, successfully accommodating a wide range of electronic and steric environments on the allene component, including electron-donating groups like methyl and methoxy, as well as electron-withdrawing halogens. 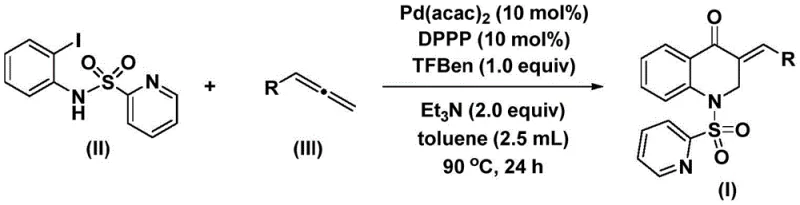 . By simplifying the post-treatment procedure to basic filtration and column chromatography, this method significantly reduces processing time and waste generation. For procurement managers, this translates to a more predictable supply chain with reduced dependency on specialized hazardous material logistics, ultimately driving down the total cost of acquisition for these valuable heterocyclic building blocks.
. By simplifying the post-treatment procedure to basic filtration and column chromatography, this method significantly reduces processing time and waste generation. For procurement managers, this translates to a more predictable supply chain with reduced dependency on specialized hazardous material logistics, ultimately driving down the total cost of acquisition for these valuable heterocyclic building blocks.
Mechanistic Insights into Palladium-Catalyzed Carbonylative Cyclization
The success of this synthetic route lies in the intricate dance of the palladium catalytic cycle, which orchestrates the formation of multiple bonds in a single operational step. The mechanism initiates with the oxidative addition of the palladium(0) species into the carbon-iodine bond of the N-pyridinesulfonyl-o-iodoaniline substrate, generating a reactive aryl-palladium intermediate. This key intermediate then undergoes migratory insertion with carbon monoxide, which is released in situ from the thermal decomposition of the TFBen additive, to form an acyl-palladium complex. Subsequently, the allene substrate coordinates to the metal center and inserts into the acyl-palladium bond, constructing the new carbon-carbon bond that defines the exocyclic double bond of the final product. The cycle concludes with an intramolecular nucleophilic attack or reductive elimination step that closes the six-membered ring and regenerates the active palladium catalyst, ready to enter the next turnover. This elegant cascade allows for the rapid assembly of complex molecular architectures from simple precursors with high atom economy.
From an impurity control perspective, the choice of ligand and additives plays a pivotal role in ensuring the purity profile required for pharmaceutical applications. The use of 1,3-bis(diphenylphosphino)propane (DPPP) as a bidentate ligand stabilizes the palladium center, preventing the formation of palladium black and minimizing side reactions such as homocoupling of the aryl iodide. Furthermore, the presence of triethylamine acts as a base to neutralize acidic byproducts, maintaining the optimal pH for the catalytic cycle to proceed smoothly. The high yields observed across various substrates, ranging from 69% to 93%, indicate that the reaction pathway is highly selective, effectively suppressing the formation of regioisomers or over-carbonylated byproducts. This level of control is essential for R&D directors who require consistent quality and minimal impurity burdens to facilitate downstream purification and regulatory filing.
How to Synthesize 3-Benzylidene-2,3-dihydroquinolone Efficiently
The patented procedure outlines a straightforward protocol that balances reaction kinetics with practical operability, making it ideal for both laboratory optimization and pilot plant campaigns. The standard operating procedure involves charging a reaction vessel with the palladium catalyst, ligand, CO surrogate, base, and substrates in toluene, followed by heating to 90°C. The detailed standardized synthesis steps see the guide below, which breaks down the precise molar ratios and workup procedures necessary to achieve the reported high yields.
- Combine Pd(acac)2 catalyst, DPPP ligand, TFBen (CO source), triethylamine, N-pyridinesulfonyl-o-iodoaniline, and the specific allene substrate in toluene solvent.
- Heat the reaction mixture to 90°C and maintain stirring for 24 to 36 hours to ensure complete conversion of the starting materials.
- Upon completion, filter the mixture, mix with silica gel, and purify the crude product via column chromatography to isolate the target 3-benzylidene-2,3-dihydroquinolone.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and supply continuity, this manufacturing process presents a compelling value proposition driven by its operational simplicity and raw material accessibility. The elimination of high-pressure gas infrastructure and the use of bench-stable reagents significantly lower the capital expenditure required for production facilities. Moreover, the robustness of the reaction conditions ensures consistent batch-to-batch reproducibility, a critical factor for maintaining long-term supply contracts with major pharmaceutical clients. By adopting this technology, manufacturers can offer a more competitive pricing structure while adhering to stringent environmental, health, and safety (EHS) standards.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived primarily from the use of commercially available and inexpensive starting materials, such as substituted o-iodoanilines and allenes, which are readily sourced from the global chemical market. The replacement of toxic carbon monoxide gas with a solid surrogate eliminates the need for expensive gas containment systems and specialized safety training, leading to substantial operational cost savings. Additionally, the high conversion rates and simplified workup reduce the consumption of solvents and purification media, further driving down the variable costs associated with production. These efficiencies allow for a more aggressive pricing strategy without compromising margin, providing a distinct competitive advantage in the marketplace.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the broad substrate scope of this methodology, which allows for the flexible synthesis of diverse analogues using a common platform. Since the key reagents are stable solids or liquids with long shelf lives, inventory management becomes more predictable, reducing the risk of production delays caused by material degradation or scarcity. The ability to tolerate various functional groups means that supply disruptions for specific substituted precursors can often be mitigated by switching to alternative synthetic routes for the starting materials without altering the core coupling process. This flexibility ensures a steady flow of high-quality intermediates to downstream customers, safeguarding their own production schedules.
- Scalability and Environmental Compliance: The transition from gram-scale laboratory experiments to kilogram or ton-scale production is facilitated by the homogeneous nature of the reaction and the absence of hazardous gases. The process generates minimal hazardous waste, as the byproducts are largely organic and manageable through standard waste treatment protocols. This aligns perfectly with the growing industry demand for green chemistry solutions and helps manufacturers meet increasingly rigorous regulatory requirements regarding emissions and effluent discharge. The scalability of this route ensures that as demand for the final API grows, the supply of the intermediate can be ramped up seamlessly to meet market needs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for potential partners.
Q: What are the key advantages of this carbonylation method over traditional synthesis?
A: This method utilizes a solid CO surrogate (TFBen) instead of toxic carbon monoxide gas, significantly improving operational safety. It also demonstrates excellent substrate compatibility with various functional groups like halogens and methoxy groups, achieving yields up to 93%.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly states the process is simple to operate, uses cheap and easily obtainable starting materials, and has been validated for gram-level synthesis, indicating strong potential for industrial scale-up.
Q: What catalyst system is required for this transformation?
A: The optimal catalytic system identified involves bis(acetylacetone)palladium(II) [Pd(acac)2] as the catalyst and 1,3-bis(diphenylphosphino)propane (DPPP) as the ligand, typically used at 10 mol% loading each.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Benzylidene-2,3-dihydroquinolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient heterocycle synthesis in accelerating drug development timelines. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can support your project from early-stage clinical trials through to full-scale market launch. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, backed by our rigorous QC labs and state-of-the-art analytical capabilities. Our expertise in palladium-catalyzed transformations allows us to optimize this specific carbonylation route for maximum yield and minimal impurity formation, guaranteeing a supply of material that exceeds industry standards.
We invite you to collaborate with us to leverage this advanced technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your supply chain efficiency and reduce your overall cost of goods.
