Advanced Palladium-Catalyzed Carbonylation for Scalable 3-Benzylidene-2,3-Dihydroquinolone Production
Introduction to Next-Generation Quinolone Synthesis
The development of efficient synthetic routes for nitrogen-containing heterocycles remains a cornerstone of modern medicinal chemistry, particularly for scaffolds exhibiting potent biological activities. As highlighted in recent literature, 2,3-dihydroquinolone compounds serve as critical molecular frameworks found in various bioactive agents, ranging from analgesic drugs to antitumor therapeutics. The structural versatility of these compounds allows for significant modulation of pharmacological properties, making them highly sought-after targets for pharmaceutical R&D teams globally. However, traditional synthetic methodologies often suffer from limitations such as harsh reaction conditions, poor atom economy, or the requirement for hazardous reagents, which can impede the rapid discovery and development of new drug candidates. Addressing these challenges, Chinese Patent CN113735826A discloses a novel and robust preparation method for 3-benzylidene-2,3-dihydroquinolone compounds. This innovation leverages a palladium-catalyzed carbonylation strategy that not only streamlines the synthetic pathway but also enhances safety and scalability, positioning it as a valuable asset for reliable pharmaceutical intermediate suppliers aiming to optimize their manufacturing portfolios.
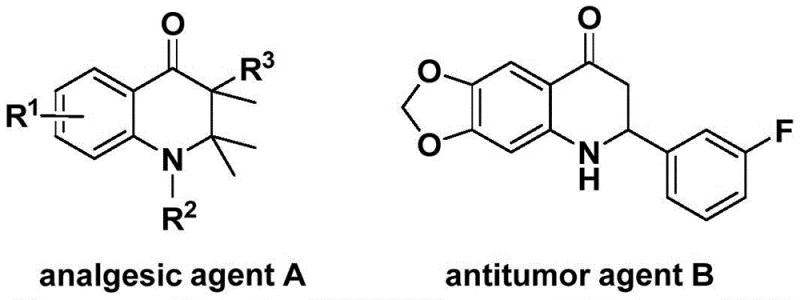
The patent specifically outlines a transition metal-catalyzed approach that utilizes N-pyridinesulfonyl-o-iodoaniline and allenes as key starting materials. By employing a solid carbon monoxide surrogate instead of gaseous CO, the method mitigates significant safety risks associated with high-pressure gas handling. This technological advancement is particularly relevant for the production of high-purity OLED materials and specialty chemicals where trace impurities can be detrimental. The ability to rapidly construct the quinolone core with high regioselectivity and yield offers a compelling solution for cost reduction in API manufacturing, enabling faster time-to-market for downstream drug products. Furthermore, the broad substrate scope demonstrated in the patent suggests that this methodology can be adapted for the commercial scale-up of complex polymer additives and other fine chemicals, providing a versatile platform for diverse industrial applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,3-dihydroquinolone derivatives has relied on classical cyclization reactions or multi-step sequences that often involve unstable intermediates and rigorous purification protocols. Many conventional carbonylation processes require the direct use of carbon monoxide gas, which poses severe safety hazards due to its toxicity and flammability, necessitating specialized high-pressure reactors and extensive safety infrastructure. These requirements not only increase capital expenditure but also complicate the regulatory compliance landscape for manufacturing facilities. Additionally, traditional methods frequently exhibit limited functional group tolerance, leading to side reactions and lower overall yields when complex substrates are employed. The reliance on stoichiometric amounts of hazardous reagents or expensive transition metals without efficient recycling mechanisms further exacerbates the environmental footprint and operational costs. For procurement managers, these inefficiencies translate into higher raw material costs and longer lead times, creating bottlenecks in the supply chain for critical pharmaceutical intermediates.
The Novel Approach
In stark contrast, the method described in Patent CN113735826A introduces a paradigm shift by utilizing a palladium-catalyzed system with a solid CO surrogate, 1,3,5-trimesic acid phenyl ester (TFBen). This approach effectively decouples the carbonylation step from the dangers of gaseous CO, allowing the reaction to proceed under atmospheric pressure or mild conditions in standard glassware or reactors. The use of TFBen ensures a controlled release of carbon monoxide in situ, which improves the reaction kinetics and selectivity towards the desired 3-benzylidene-2,3-dihydroquinolone product. Moreover, the catalytic system employs readily available ligands such as DPPP and a robust palladium source, Pd(acac)2, which are cost-effective and easy to source. This novel route significantly simplifies the post-treatment process, as the reaction mixture can be directly purified via column chromatography after simple filtration. For supply chain heads, this translates to reduced lead time for high-purity pharmaceutical intermediates and enhanced supply continuity, as the process is less susceptible to disruptions caused by hazardous material transport regulations.
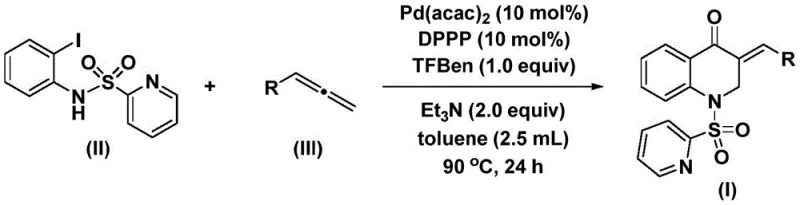
Mechanistic Insights into Pd-Catalyzed Carbonylation
The efficacy of this synthetic route is underpinned by a sophisticated catalytic cycle that ensures high turnover and selectivity. The mechanism initiates with the oxidative addition of the palladium(0) species into the carbon-iodine bond of the N-pyridinesulfonyl-o-iodoaniline substrate, generating a reactive aryl-palladium intermediate. This step is crucial as it activates the aromatic ring for subsequent functionalization. Following this activation, the carbon monoxide released from the decomposition of TFBen inserts into the palladium-carbon bond, forming an acyl-palladium species. This carbonyl insertion is the key step that constructs the ketone functionality inherent to the quinolone scaffold. The presence of the pyridine sulfonyl group on the nitrogen atom plays a dual role: it acts as a directing group to facilitate the initial oxidative addition and serves as a leaving group or stabilizing element during the cyclization process. The precise control over these elementary steps minimizes the formation of homocoupling byproducts or decarbonylation side reactions, which are common pitfalls in palladium-catalyzed carbonylations.
Subsequently, the allene substrate coordinates to the acyl-palladium intermediate, followed by migratory insertion to form an alkyl-palladium species. This step determines the regiochemistry of the exocyclic double bond in the final product, ensuring the formation of the 3-benzylidene motif rather than internal isomers. Finally, reductive elimination occurs to release the 3-benzylidene-2,3-dihydroquinolone product and regenerate the active palladium(0) catalyst, closing the catalytic cycle. The use of triethylamine as a base is essential to neutralize any acidic byproducts and maintain the catalytic activity of the palladium species throughout the reaction duration of 24 to 48 hours. Understanding this mechanistic pathway allows R&D directors to fine-tune reaction parameters, such as temperature and ligand choice, to optimize the impurity profile and maximize yield for specific derivatives, ensuring the production of high-purity pharmaceutical intermediates that meet stringent quality standards.
How to Synthesize 3-Benzylidene-2,3-Dihydroquinolone Efficiently
The practical implementation of this synthesis involves a straightforward one-pot procedure that is amenable to both laboratory screening and pilot-scale production. The process begins by combining the palladium catalyst, phosphine ligand, CO surrogate, and substrates in an organic solvent such as toluene, which has been identified as the optimal medium for solubility and reaction rate. The mixture is then heated to a moderate temperature of 90°C, balancing reaction speed with energy efficiency. Detailed standardized synthesis steps, including specific molar ratios and workup procedures, are provided in the technical guide below to ensure reproducibility and safety.
- Charge a reaction vessel with Pd(acac)2 (10 mol%), DPPP (10 mol%), TFBen (1.0 equiv), N-pyridinesulfonyl-o-iodoaniline, and the specific allene substrate in toluene.
- Add triethylamine (2.0 equiv) as the base and heat the mixture to 90°C under stirring for 24 to 48 hours to ensure complete conversion.
- Upon completion, filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the target 3-benzylidene-2,3-dihydroquinolone derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this palladium-catalyzed carbonylation method offers substantial strategic benefits for organizations focused on cost optimization and supply chain resilience. The elimination of gaseous carbon monoxide removes a major logistical and safety barrier, allowing for more flexible manufacturing site selection and reduced insurance premiums. Furthermore, the high reaction efficiency and broad substrate compatibility mean that a single platform technology can be used to produce a diverse library of quinolone derivatives, reducing the need for multiple specialized production lines. This versatility is a key driver for cost reduction in API manufacturing, as it maximizes asset utilization and minimizes changeover times between different product campaigns.
- Cost Reduction in Manufacturing: The use of commercially available and relatively inexpensive reagents, such as Pd(acac)2 and DPPP, combined with the high yields reported (often exceeding 80-90%), significantly lowers the cost of goods sold (COGS). By avoiding the need for specialized high-pressure equipment required for gaseous CO, capital expenditure is drastically reduced. Additionally, the simplified workup procedure reduces solvent consumption and waste disposal costs, contributing to a leaner and more economical production process that enhances overall profit margins for fine chemical intermediates.
- Enhanced Supply Chain Reliability: The reliance on stable, solid reagents like TFBen and readily available allenes ensures a robust supply chain that is less vulnerable to disruptions compared to processes dependent on compressed gases or cryogenic liquids. The ability to source raw materials from multiple global suppliers further mitigates risk. For procurement managers, this means securing a reliable pharmaceutical intermediate supplier who can guarantee consistent delivery schedules and maintain inventory levels without the volatility associated with hazardous material logistics, thereby supporting uninterrupted downstream drug production.
- Scalability and Environmental Compliance: The mild reaction conditions and the absence of toxic gas emissions make this process inherently greener and easier to scale from gram to kilogram or ton quantities. The reduced environmental footprint aligns with increasingly stringent global regulations on industrial emissions and waste management. This facilitates smoother regulatory approvals and permits for commercial scale-up of complex pharmaceutical intermediates, ensuring long-term operational sustainability and minimizing the risk of production shutdowns due to environmental non-compliance.
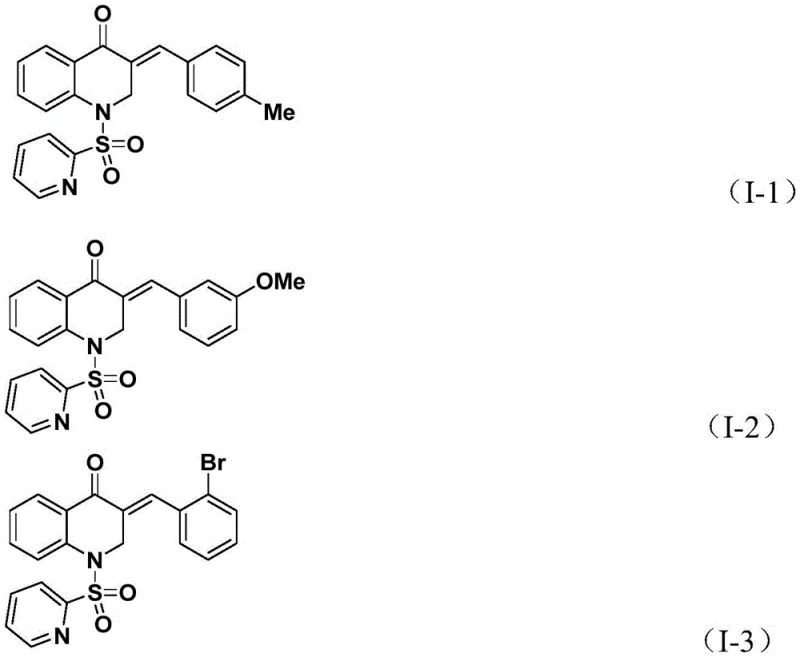
Frequently Asked Questions (FAQ)
To address common technical and commercial inquiries regarding this synthesis technology, we have compiled a set of questions based on the detailed experimental data and beneficial effects described in the patent documentation. These insights are designed to clarify the operational advantages and technical feasibility for potential partners and stakeholders evaluating this method for their own production needs.
Q: What is the primary advantage of using TFBen over carbon monoxide gas in this synthesis?
A: Using 1,3,5-trimesic acid phenyl ester (TFBen) as a solid carbon monoxide surrogate eliminates the need for handling toxic, high-pressure CO gas, significantly enhancing operational safety and simplifying equipment requirements for industrial scale-up.
Q: What is the substrate compatibility of this palladium-catalyzed method?
A: The method demonstrates excellent functional group tolerance, successfully accommodating various substituents on the aryl ring of the allene, including electron-donating groups like methyl and methoxy, as well as electron-withdrawing halogens like bromine and chlorine, with yields often exceeding 80%.
Q: Is this process suitable for large-scale manufacturing of pharmaceutical intermediates?
A: Yes, the process utilizes commercially available catalysts and reagents, operates at moderate temperatures (90°C), and avoids hazardous gases, making it highly amenable to commercial scale-up for the production of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Benzylidene-2,3-Dihydroquinolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the one described in Patent CN113735826A for the future of pharmaceutical manufacturing. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are seamlessly translated into robust industrial processes. We are committed to delivering high-purity pharmaceutical intermediates that adhere to stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch.
We invite you to collaborate with us to leverage this efficient synthesis route for your specific project requirements. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume needs, along with specific COA data and route feasibility assessments. Let us help you accelerate your drug development timeline with a supply partner dedicated to quality, safety, and commercial excellence.
