Advanced Electrochemical Manufacturing of Seven-Membered Ring Sulfonamide Intermediates for Global Pharma Supply Chains
Introduction to Next-Generation Sulfonamide Synthesis
The pharmaceutical industry continuously seeks innovative synthetic methodologies to access complex heterocyclic scaffolds that serve as critical building blocks for new drug candidates. Patent CN115747837A introduces a groundbreaking electrochemical synthesis method specifically designed for constructing sulfonamide polycyclic compounds containing a seven-membered ring skeleton. This technology addresses a long-standing challenge in organic synthesis, where the formation of medium-sized rings, particularly seven-membered rings, is often hindered by unfavorable entropy and transannular interactions. By leveraging anodic oxidation to generate reactive sulfonyl radicals in situ, this method enables a seamless radical tandem cyclization that constructs a complex 6-7-5 fused ring system in a single operational step.
The significance of this invention extends beyond academic curiosity; it offers a practical, green, and efficient route for producing high-value pharmaceutical intermediates. Sulfonamide motifs are ubiquitous in medicinal chemistry, appearing in a vast array of bioactive molecules including diuretics, antibiotics, and antiviral agents. The ability to rapidly assemble polycyclic sulfonamide cores with high structural diversity provides medicinal chemists with powerful tools for lead optimization. Furthermore, the electrochemical nature of the reaction aligns perfectly with the industry's shift towards sustainable manufacturing practices, eliminating the need for hazardous chemical oxidants and reducing the environmental footprint of the synthesis process.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes to access fused sulfonamide systems often rely heavily on transition metal catalysis, utilizing precious metals such as palladium, rhodium, or copper to facilitate C-H activation or cross-coupling reactions. These conventional methods frequently require stoichiometric amounts of external oxidants, such as hypervalent iodine reagents or peroxides, to regenerate the active catalytic species. This reliance introduces significant drawbacks, including the generation of large quantities of toxic metal waste, high costs associated with catalyst removal to meet stringent pharmaceutical purity standards, and safety concerns related to the handling of strong oxidants. Additionally, constructing seven-membered rings via traditional thermal cyclization is notoriously difficult due to kinetic and thermodynamic barriers, often resulting in low yields and poor selectivity.
The Novel Approach
The electrochemical methodology disclosed in the patent represents a paradigm shift by replacing chemical oxidants with electricity. In this novel approach, the benzenesulfinate anion is directly oxidized at the anode surface to generate a highly reactive sulfonyl radical without the need for any metal catalyst. This radical species then initiates a cascade reaction with an N,N-disubstituted sulfonamide substrate containing both alkene and alkyne functionalities. The process proceeds through a sophisticated sequence of radical additions and cyclizations, ultimately forging the challenging seven-membered ring alongside a five-membered ring and the existing six-membered aromatic core. This metal-free strategy not only simplifies the reaction setup but also drastically improves the atom economy and reduces the E-factor of the synthesis.
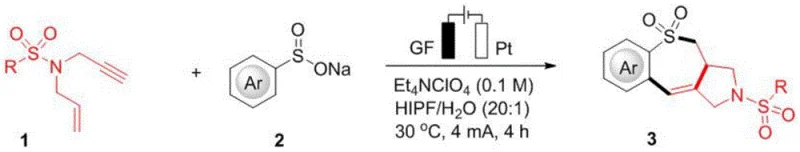
Mechanistic Insights into Electrooxidative Radical Tandem Cyclization
Understanding the mechanistic pathway is crucial for R&D teams aiming to optimize this process for specific API intermediates. The reaction begins at the anode, where the sodium aryl sulfinate undergoes a single-electron oxidation to form a sulfonyl radical species. This electrophilic radical preferentially attacks the electron-rich terminal alkene moiety of the N-allyl-N-propargyl sulfonamide substrate, generating a transient carbon-centered radical intermediate. This initial addition is kinetically favored over attack on the alkyne, setting the stage for the subsequent cyclization events. The resulting alkyl radical then undergoes an intramolecular 5-exo-dig cyclization onto the pendant alkyne group, forming a vinyl radical and establishing the five-membered ring component of the final scaffold.
Following the formation of the five-membered ring, the vinyl radical intermediate engages in a remarkable 7-endo-trig cyclization onto the adjacent aromatic ring. This step is the key to constructing the seven-membered ring skeleton, a transformation that is rarely observed in standard radical chemistry due to ring strain and entropy. The resulting cyclohexadienyl-type radical is then subjected to a second anodic oxidation event, losing another electron to form a cationic intermediate. Finally, deprotonation restores aromaticity to the benzene ring, yielding the stable, fused 6-7-5 polycyclic sulfonamide product. This dual-oxidation mechanism highlights the precise control offered by electrochemical parameters in driving complex multistep transformations.
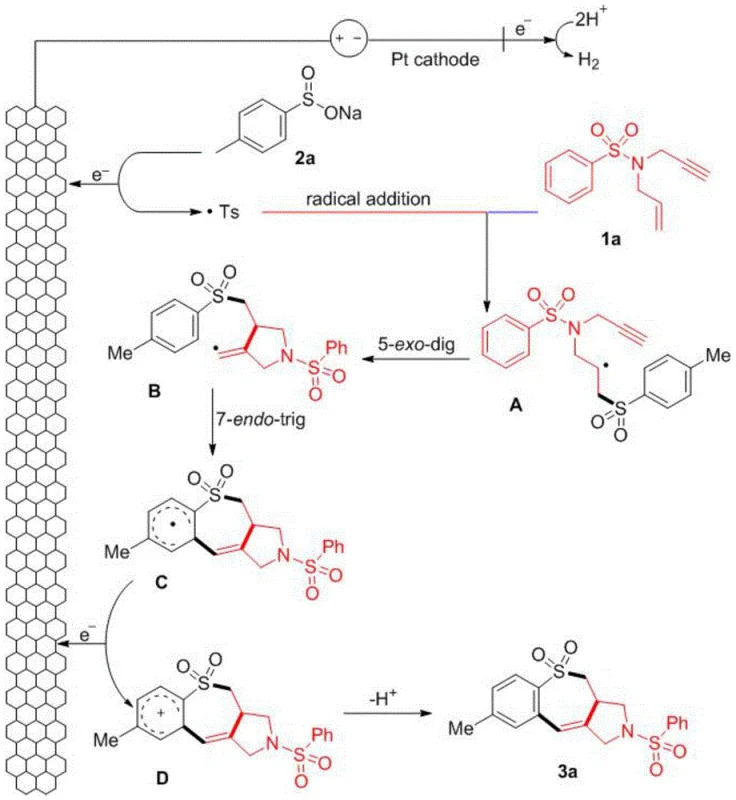
How to Synthesize Sulfonamide Polycyclic Compounds Efficiently
The experimental protocol described in the patent demonstrates the robustness and reproducibility of this electrochemical transformation. For R&D professionals looking to implement this synthesis, the process utilizes a simple undivided cell configuration equipped with a graphite felt (GF) anode and a platinum (Pt) cathode. The reaction is conducted in a mixed solvent system of hexafluoroisopropanol (HFIP) and water, which plays a critical role in stabilizing radical intermediates and facilitating proton transfer. Under optimized conditions involving a constant current of 4 mA at a mild temperature of 30°C, the reaction reaches completion within 4 hours, delivering the target polycyclic products in good to excellent yields. Detailed standardized synthesis steps are provided in the guide below.
- Prepare the reaction mixture by combining N,N-disubstituted sulfonamide substrates and sodium aryl sulfinate salts in a mixed solvent system of hexafluoroisopropanol (HFIP) and water.
- Add tetraethylammonium perchlorate as the supporting electrolyte and install a graphite felt (GF) anode and platinum (Pt) cathode into the undivided cell.
- Apply a constant current of 4 mA at 30°C for 4 hours to drive the anodic oxidation and radical cascade cyclization, followed by silica gel column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this electrochemical technology offers tangible strategic benefits that extend beyond mere technical novelty. The elimination of transition metal catalysts removes a major cost driver and supply chain bottleneck, as precious metals are subject to significant price volatility and geopolitical supply risks. Furthermore, the absence of heavy metals simplifies the purification workflow, potentially reducing the number of processing steps required to meet regulatory limits for residual metals in pharmaceutical ingredients. This streamlining of the downstream process translates directly into reduced manufacturing lead times and lower operational expenditures.
- Cost Reduction in Manufacturing: The primary economic advantage lies in the substitution of expensive chemical reagents with electricity. By avoiding the purchase of stoichiometric oxidants and precious metal catalysts, the raw material costs are significantly lowered. Additionally, the simplified workup procedure, which avoids complex metal scavenging steps, reduces solvent consumption and waste disposal costs. The use of inexpensive electrode materials like graphite felt further contributes to a favorable cost profile, making the commercial scale-up of complex pharmaceutical intermediates more economically viable compared to traditional catalytic methods.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, namely N,N-disubstituted sulfonamides and sodium aryl sulfinates, are commercially available and can be sourced from multiple suppliers globally. This diversity in sourcing mitigates the risk of single-source dependency. Moreover, the reaction conditions are mild and do not require specialized high-pressure or cryogenic equipment, allowing for production in standard chemical manufacturing facilities. This flexibility ensures consistent supply continuity even during periods of market disruption, providing a reliable source of high-purity pharmaceutical intermediates for downstream drug formulation.
- Scalability and Environmental Compliance: Electrochemical processes are inherently scalable, particularly when transitioning from batch to continuous flow reactors. The ability to precisely control current density and residence time in flow systems allows for safe and efficient production at the multi-kilogram or ton scale. From an environmental perspective, this green synthesis method aligns with increasingly strict global regulations on industrial emissions and waste. The reduction in hazardous waste generation and the avoidance of toxic metal residues enhance the sustainability profile of the manufacturing process, facilitating easier regulatory approval and improving the corporate social responsibility standing of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrochemical synthesis technology. These answers are derived directly from the patent data and are intended to clarify the feasibility and advantages of adopting this method for large-scale production. Understanding these details is essential for stakeholders evaluating the integration of this novel pathway into their existing manufacturing portfolios.
Q: What are the primary advantages of this electrochemical method over traditional transition metal catalysis?
A: This electrochemical approach eliminates the need for expensive and toxic transition metal catalysts such as palladium or rhodium, as well as stoichiometric chemical oxidants. By using electrons as the reagent, the process significantly reduces heavy metal contamination risks, simplifies downstream purification, and lowers the overall E-factor, making it ideal for GMP pharmaceutical intermediate production.
Q: How does the reaction achieve the difficult formation of a seven-membered ring?
A: The synthesis utilizes a unique radical tandem cyclization strategy. It initiates with the anodic generation of a sulfonyl radical, which undergoes a sequential 5-exo-dig cyclization followed by a challenging 7-endo-trig cyclization. This specific pathway overcomes the entropic barriers typically associated with forming medium-sized rings, allowing for the efficient construction of the complex 6-7-5 fused skeleton.
Q: Is this process scalable for commercial manufacturing of pharmaceutical intermediates?
A: Yes, the process operates under mild conditions (30°C) using constant current electrolysis, which is highly amenable to scale-up in flow electrochemical reactors. The use of robust electrode materials like graphite felt and platinum, combined with readily available starting materials, ensures supply chain continuity and facilitates the transition from laboratory discovery to multi-kilogram commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sulfonamide Polycyclic Compounds Supplier
As the demand for complex heterocyclic scaffolds continues to grow in the pharmaceutical sector, partnering with an experienced CDMO is essential for successful commercialization. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can seamlessly transition from laboratory discovery to full-scale manufacturing. Our facility is equipped with state-of-the-art electrochemical reactors and rigorous QC labs capable of meeting stringent purity specifications required by global regulatory agencies. We are committed to delivering high-quality intermediates that accelerate your drug development timelines while maintaining the highest standards of safety and compliance.
We invite you to collaborate with our technical team to evaluate the feasibility of this electrochemical route for your specific project needs. By leveraging our expertise in process optimization and scale-up, we can help you realize substantial cost savings and efficiency gains. Please contact our technical procurement team to request a Customized Cost-Saving Analysis, specific COA data, and route feasibility assessments tailored to your target molecule. Let us be your trusted partner in navigating the complexities of modern pharmaceutical synthesis.
