Advanced Ag-Catalyzed Cyanoalkylation for Complex Nitrogen Heterocycles Manufacturing
The pharmaceutical and fine chemical industries continuously seek robust methodologies for constructing complex nitrogen-containing scaffolds, which are ubiquitous in bioactive molecules. Patent CN111116465B discloses a groundbreaking synthetic approach for preparing cyanoalkyl substituted nitrogen heterocyclic compounds, addressing critical limitations in current late-stage functionalization techniques. This technology utilizes an efficient Ag+/S2O8 2- catalytic system to achieve direct cyanoalkylation of various electron-deficient nitrogen heterocycles under mild conditions. Unlike traditional methods that often rely on harsh reagents or difficult-to-scale photochemical processes, this invention offers a practical pathway using readily available cycloalkanone oximes as radical precursors. The versatility of this method allows for the modification of diverse substrates including quinolines, pyridines, and quinoxalines, significantly expanding the chemical space accessible for drug discovery and development programs.
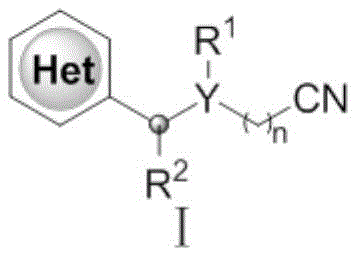
The strategic importance of this synthesis lies in its ability to introduce cyanoalkyl groups, which serve as versatile handles for further chemical transformations into amines, acids, or tetrazoles. As noted in the background art, over 60% of FDA-approved drug molecules contain nitrogen heterocyclic structures, making efficient functionalization methods highly valuable. The disclosed process operates effectively at temperatures ranging from 0-80°C, with optimal results observed between 20-30°C, ensuring energy efficiency and safety. By leveraging a silver-catalyzed radical mechanism, the invention bypasses the need for pre-functionalized starting materials, thereby reducing step counts and overall waste generation in the synthesis of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the functionalization of nitrogen heterocycles has relied heavily on Minisci-type reactions or transition-metal catalyzed cross-couplings, which often present significant operational challenges. Conventional Minisci reactions typically require strong acidic conditions and stoichiometric amounts of oxidants, which can lead to poor functional group tolerance and substrate decomposition. Furthermore, recent advancements in photocatalytic C-H activation, while powerful, introduce substantial barriers to industrial adoption. Photocatalytic systems necessitate specialized reactor designs to ensure uniform light penetration, a constraint that becomes exponentially difficult to manage during scale-up from gram to kilogram scales. The reliance on expensive photocatalysts and the potential for batch-to-batch variability due to light intensity fluctuations further complicate the manufacturing process. Additionally, many existing methods utilize carboxylic acids or alkyl halides that may not provide the specific cyanoalkyl motif required for downstream medicinal chemistry optimization without additional synthetic steps.
The Novel Approach
The methodology described in CN111116465B represents a paradigm shift by employing a silver-catalyzed oxidative system that operates independently of light irradiation. This novel approach utilizes cycloalkanone oximes as stable and tunable radical precursors, which decompose under the influence of the Ag+/S2O8 2- system to release cyanoalkyl radicals. The reaction proceeds smoothly in a biphasic solvent system comprising dichloromethane and water, facilitating easy workup and product isolation. By avoiding the complexities of photochemistry, this method ensures consistent reaction kinetics regardless of reactor size, directly addressing the scalability bottleneck. The use of inexpensive silver salts like silver nitrate and common oxidants like sodium persulfate drastically reduces raw material costs compared to precious metal photocatalysts. Moreover, the mild reaction temperature of 25°C preserves sensitive functional groups on the heterocyclic core, enabling the synthesis of complex derivatives that would otherwise degrade under harsher thermal or acidic conditions typical of older protocols.
Mechanistic Insights into Ag-Catalyzed Radical Cyanoalkylation
The core of this transformative synthesis lies in the unique interaction between the silver catalyst and the persulfate oxidant to generate reactive radical species. The mechanism initiates with the oxidation of Ag(I) to Ag(II) by the persulfate anion (S2O8 2-), creating a potent single-electron oxidant in situ. This high-valent silver species then interacts with the cycloalkanone oxime precursor, triggering a single-electron transfer that leads to the formation of an iminyl radical intermediate. Subsequent fragmentation of this intermediate involves the cleavage of the carbon-carbon bond adjacent to the oxime nitrogen, releasing acetone and generating the desired nucleophilic cyanoalkyl radical. This radical species is then captured by the protonated nitrogen heterocycle, which is activated by the added acid (such as trifluoroacetic acid) to increase its electrophilicity. The final rearomatization step yields the cyanoalkyl substituted product while regenerating the proton source, completing the catalytic cycle.
Understanding the impurity profile is critical for R&D teams aiming to implement this chemistry, particularly regarding the selectivity of the radical addition. The reaction conditions are finely tuned to favor mono-substitution, minimizing the formation of di- or tri-alkylated byproducts which can be difficult to separate. The choice of acid plays a pivotal role here; acids with appropriate pKa values, such as trifluoroacetic acid or p-toluenesulfonic acid, ensure sufficient protonation of the heterocycle without causing degradation of the acid-sensitive cyano group or the oxime precursor. Furthermore, the biphasic solvent system helps to moderate the concentration of radical species in the organic phase, reducing the likelihood of radical-radical coupling side reactions. This controlled environment ensures high purity of the final intermediate, reducing the burden on downstream purification processes and improving the overall mass balance of the manufacturing campaign.
How to Synthesize Cyanoalkyl Nitrogen Heterocycles Efficiently
Implementing this synthesis requires careful attention to reagent stoichiometry and mixing protocols to maximize yield and reproducibility. The patent outlines a robust procedure where the nitrogen heterocycle and the cycloalkanone oxime are dissolved in a mixture of dichloromethane and water, typically in a volume ratio of 1:2. The addition of the catalyst, oxidant, and acid must be managed to maintain the reaction temperature within the optimal 20-30°C range, preventing exothermic runaway. Detailed standardized synthesis steps see the guide below.
- Mix nitrogen-containing heterocycle and cycloalkanone oxime in a solvent mixture of dichloromethane and water under inert gas protection.
- Add silver nitrate catalyst, sodium persulfate oxidant, and trifluoroacetic acid to the reaction mixture.
- Stir at 20-30°C for 12 hours, then quench, extract, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement and supply chain leaders, the transition to this Ag-catalyzed protocol offers tangible benefits in terms of cost stability and operational flexibility. The elimination of photocatalytic equipment removes a significant capital expenditure barrier, allowing production to occur in standard glass-lined or stainless steel reactors already present in most multipurpose facilities. This compatibility with existing infrastructure drastically reduces the lead time required for technology transfer and process validation. Furthermore, the reliance on commodity chemicals like silver nitrate and sodium persulfate insulates the supply chain from the volatility often associated with specialized organometallic catalysts or custom-synthesized photoredox mediators. The simplicity of the workup procedure, involving basic extraction and crystallization or chromatography, minimizes solvent consumption and waste disposal costs, contributing to a more sustainable and economically viable manufacturing process.
- Cost Reduction in Manufacturing: The replacement of expensive photocatalysts with economical silver salts results in a substantial decrease in raw material costs per kilogram of product. By removing the need for specialized lighting arrays and cooling systems required for photochemistry, the process significantly lowers utility and maintenance expenses. The high atom economy of the radical fragmentation step ensures that the majority of the precursor mass is converted into the desired product or benign byproducts like acetone, reducing waste treatment liabilities. Overall, the streamlined process flow translates to a lower cost of goods sold (COGS), providing a competitive edge in pricing negotiations for bulk pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Sourcing strategies are simplified as the key reagents, including various substituted quinolines and cycloalkanone oximes, are commercially available from multiple global suppliers. This multi-sourcing capability mitigates the risk of supply disruptions that can occur with proprietary or single-source catalysts. The robustness of the reaction conditions means that slight variations in raw material quality do not critically impact the outcome, ensuring consistent batch-to-batch quality. Consequently, manufacturers can maintain tighter delivery schedules and reduce safety stock requirements, leading to a more agile and responsive supply chain capable of meeting fluctuating market demands without compromising on quality standards.
- Scalability and Environmental Compliance: The absence of light-dependent steps removes the geometric constraints typically limiting photochemical reactions, allowing for seamless scale-up from laboratory to pilot and commercial production scales. The mild operating temperatures reduce energy consumption for heating or cooling, aligning with green chemistry principles and corporate sustainability goals. Additionally, the use of water as a co-solvent reduces the total volume of organic solvents required, lowering the environmental footprint of the process. This compliance with increasingly stringent environmental regulations facilitates smoother regulatory approvals and enhances the company's reputation as a responsible chemical manufacturer committed to eco-friendly production practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this cyanoalkylation technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on reaction scope and operational parameters. Understanding these details is essential for project managers evaluating the feasibility of integrating this route into their existing portfolio of synthetic capabilities.
Q: What are the advantages of this Ag-catalyzed method over photocatalysis?
A: This method eliminates the need for expensive photocatalysts and specialized light equipment, solving scalability issues associated with light penetration in large reactors while maintaining mild reaction conditions.
Q: What is the role of the cycloalkanone oxime in this reaction?
A: The cycloalkanone oxime acts as a radical precursor. Under the Ag+/S2O8 2- system, it undergoes decarboxylation and C-C bond cleavage to generate the active cyanoalkyl radical required for substitution.
Q: Can this process be scaled for industrial production?
A: Yes, the process operates at mild temperatures (20-30°C) without light dependency, making it highly suitable for commercial scale-up in standard stainless steel reactors without complex engineering modifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyanoalkyl Nitrogen Heterocycles Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug development timelines. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are successfully translated into reliable industrial processes. We are equipped with rigorous QC labs and adhere to stringent purity specifications to guarantee that every batch of cyanoalkyl nitrogen heterocycles meets the highest industry standards. Our commitment to technical excellence allows us to navigate the complexities of radical chemistry, delivering high-quality intermediates that support your R&D and commercial manufacturing needs.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized synthesis can enhance your supply chain efficiency. Let us partner with you to drive innovation and cost-effectiveness in the production of complex nitrogen-containing pharmaceutical building blocks.
