Advanced Synthesis of Cyanoalkyl-Substituted Nitrogen Heterocycles for Pharmaceutical Applications
The pharmaceutical and agrochemical industries rely heavily on nitrogen-containing heterocyclic compounds, which serve as critical scaffolds in over 84% of FDA-approved drug molecules. The ability to efficiently functionalize these cores, particularly through C-H bond activation, is a cornerstone of modern medicinal chemistry. Patent CN111116465A introduces a groundbreaking synthetic methodology for preparing cyanoalkyl-substituted nitrogen heterocycles, addressing long-standing challenges in late-stage functionalization. This technology utilizes a silver-catalyzed radical system that offers a superior alternative to traditional Minisci reactions and emerging photocatalytic methods. By leveraging cycloalkanone oximes as radical precursors in conjunction with an Ag+/S2O8 2- oxidation system, this process achieves high substrate adaptability while maintaining mild reaction conditions. For R&D directors and procurement specialists, this represents a significant opportunity to access complex intermediates with improved cost-efficiency and supply chain reliability.
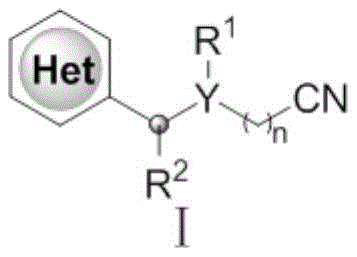
The construction of carbon-carbon bonds on electron-deficient nitrogen heterocycles has historically been dominated by the Minisci reaction, which typically requires harsh acidic conditions and stoichiometric oxidants. While effective, conventional methods often suffer from poor regioselectivity and limited functional group tolerance. More recently, visible-light photocatalysis emerged as a milder alternative; however, it introduces new bottlenecks for industrial adoption. Photocatalytic processes often rely on expensive iridium or ruthenium complexes, require specialized reactor setups to ensure uniform light penetration, and face significant difficulties when scaling from milligram to kilogram quantities due to the Beer-Lambert law limitations. These factors collectively drive up the cost of goods sold (COGS) and extend lead times for active pharmaceutical ingredient (API) intermediates.
In stark contrast, the novel approach detailed in the patent data circumvents these limitations by employing a thermally driven silver-catalyzed radical generation system. This method operates effectively at temperatures between 20°C and 30°C, eliminating the need for energy-intensive heating or cooling cycles. The use of inexpensive silver salts, such as silver nitrate, coupled with common oxidants like sodium persulfate, drastically reduces raw material costs compared to precious metal photocatalysts. Furthermore, the reaction proceeds in a biphasic solvent system of dichloromethane and water, which not only facilitates product separation but also aligns with green chemistry principles by reducing the reliance on purely organic solvent systems. This shift from photochemical to thermal radical chemistry fundamentally simplifies the engineering requirements for commercial manufacturing.
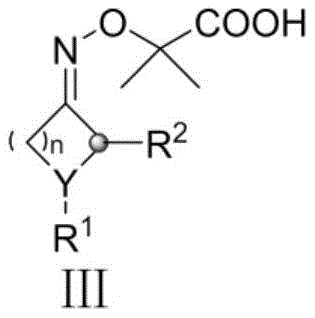
Mechanistically, the success of this transformation hinges on the efficient generation of cyanoalkyl radicals from cycloalkanone oxime esters. The process initiates with the oxidation of the silver(I) catalyst to a high-valent silver(II) species by the persulfate oxidant. This active silver species then interacts with the cycloalkanone oxime precursor, triggering a single-electron transfer that generates an iminyl radical intermediate. Crucially, this iminyl radical undergoes a rapid beta-scission of the carbon-carbon bond within the strained ring system, resulting in the extrusion of an acetonyl fragment and the formation of a nitrogen-centered radical. Subsequent fragmentation releases the desired cyanoalkyl radical, which is nucleophilic in nature. This radical selectively attacks the electron-deficient positions of the protonated nitrogen heterocycle, followed by rearomatization to yield the final cyanoalkyl-substituted product. This intricate cascade allows for the precise installation of functionalized alkyl chains that are otherwise difficult to introduce via classical nucleophilic substitution.
Impurity control is another critical aspect where this mechanism offers distinct advantages. The specificity of the radical ring-opening step minimizes the formation of side products associated with non-selective hydrogen abstraction often seen in traditional radical halogenation methods. Additionally, the use of trifluoroacetic acid or p-toluenesulfonic acid ensures the proper protonation state of the heterocyclic substrate, enhancing reactivity while suppressing polymerization or oligomerization of the radical species. The reaction conditions are sufficiently mild to preserve sensitive functional groups such as esters, halides, and protected amines, which is essential for the synthesis of advanced intermediates where downstream diversification is required. This high level of chemoselectivity translates directly to simplified purification workflows and higher overall yields in multi-step synthesis campaigns.
How to Synthesize Cyanoalkyl-Substituted Nitrogen Heterocycles Efficiently
The operational simplicity of this synthetic route makes it highly attractive for process chemistry teams aiming to transition from discovery to development scales. The standard protocol involves dissolving the nitrogen heterocycle and the cycloalkanone oxime precursor in a mixed solvent system, typically dichloromethane and water in a 1:2 volume ratio. Under an inert atmosphere, catalytic amounts of silver nitrate and stoichiometric sodium persulfate are added along with an acid promoter. The reaction mixture is stirred at ambient temperature (25°C) for approximately 12 hours, allowing sufficient time for the radical cascade to reach completion. Post-reaction workup is straightforward, involving quenching with saturated sodium bicarbonate solution and extraction with ethyl acetate. The crude material is then purified via silica gel column chromatography using petroleum ether and ethyl acetate gradients. Detailed standardized synthesis steps are provided in the guide below.
- Mix the nitrogen-containing heterocyclic compound and cycloalkanone oxime compound in a solvent system comprising dichloromethane and water under inert gas protection.
- Add silver nitrate catalyst, sodium persulfate oxidant, and trifluoroacetic acid to the mixture to initiate the radical cyanoalkylation reaction at 20-30°C.
- Quench the reaction with saturated sodium bicarbonate, extract with ethyl acetate, and purify the crude product via column chromatography to obtain the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this silver-catalyzed methodology offers tangible benefits beyond mere technical feasibility. The elimination of photocatalytic equipment removes a significant capital expenditure barrier, allowing existing stainless steel reactor infrastructure to be utilized without modification. This compatibility with standard batch processing equipment significantly reduces the lead time for process validation and technology transfer. Moreover, the reagents employed—silver nitrate, sodium persulfate, and simple organic acids—are commodity chemicals available from multiple global suppliers, mitigating the risk of single-source dependency that often plagues specialized photocatalyst supply chains.
- Cost Reduction in Manufacturing: The economic impact of replacing expensive photocatalysts with commodity silver salts cannot be overstated. Photocatalysts often cost hundreds of dollars per gram and may require recycling protocols that add complexity to the process. In contrast, the silver catalyst loading in this method is low (0.1 to 0.3 equivalents relative to substrate), and the oxidant is exceptionally cheap. Furthermore, the ability to run the reaction at room temperature eliminates energy costs associated with cryogenic cooling or high-temperature heating. The simplified workup procedure, which avoids complex distillation or specialized filtration required for catalyst removal in other systems, further drives down operational expenses and labor hours per kilogram of product.
- Enhanced Supply Chain Reliability: Supply continuity is paramount for API manufacturing. This synthesis route relies on robust, shelf-stable reagents that do not require cold chain logistics or protection from ambient light during storage. The broad substrate scope means that a single set of operating parameters can be applied to a wide library of heterocyclic cores, from quinolines to pyridazines. This standardization reduces the need for custom process development for each new analog, accelerating the timeline from benchtop to pilot plant. The use of water as a co-solvent also reduces the total volume of flammable organic solvents required, improving safety profiles and lowering insurance and compliance costs associated with hazardous material handling.
- Scalability and Environmental Compliance: Scaling photochemical reactions often requires switching from batch to continuous flow reactors to overcome light penetration limits, a change that demands significant engineering resources. This thermal radical method scales linearly in traditional batch reactors, facilitating a smoother path to commercial production. Additionally, the reduced solvent load and the absence of heavy metal photocatalysts simplify waste stream management. The aqueous waste generated is easier to treat compared to organic streams laden with complex metal ligands. This alignment with environmental, social, and governance (ESG) goals is increasingly important for multinational corporations seeking sustainable supply chain partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this cyanoalkylation technology. These insights are derived directly from the experimental data and scope evaluation presented in the patent literature, ensuring that potential partners have a clear understanding of the process capabilities and limitations.
Q: What are the advantages of this Ag-catalyzed method over photocatalysis?
A: This method eliminates the need for expensive photocatalysts and specialized light equipment, significantly reducing capital expenditure and simplifying the scale-up process for industrial manufacturing.
Q: What types of nitrogen heterocycles are compatible with this synthesis?
A: The process demonstrates excellent substrate adaptability, successfully functionalizing quinolines, pyridines, pyridazines, quinoxalines, and pyrimidines with various substituents.
Q: Is this process suitable for large-scale production?
A: Yes, the reaction operates at mild temperatures (20-30°C) using cheap reagents like silver nitrate and sodium persulfate, making it highly amenable to commercial scale-up without complex engineering controls.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyanoalkyl Nitrogen Heterocycles Supplier
The technological advancements described in patent CN111116465A represent a pivotal shift in how complex nitrogen heterocycles can be manufactured for the life sciences sector. At NINGBO INNO PHARMCHEM, we recognize the strategic value of such innovations in accelerating drug discovery and optimizing production costs. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped to handle the specific requirements of radical chemistry, including rigorous safety protocols for oxidant handling and stringent purity specifications to meet global regulatory standards. Our rigorous QC labs ensure that every batch of cyanoalkyl-substituted intermediates meets the exacting criteria required for clinical and commercial applications.
We invite R&D and procurement leaders to collaborate with us to leverage this efficient synthetic route for your next project. By integrating this technology into your supply chain, you can achieve significant process intensification and cost optimization. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific molecule. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing expertise can support your long-term commercial goals.
