Advanced Nickel-Catalyzed Carbonylation for Scalable Indole Pharmaceutical Intermediates
Advanced Nickel-Catalyzed Carbonylation for Scalable Indole Pharmaceutical Intermediates
The pharmaceutical industry continuously seeks robust and efficient synthetic routes for privileged scaffolds, among which the indole nucleus stands out as a cornerstone structure found in numerous bioactive molecules. As detailed in the recent patent CN115286553A, a novel preparation method for indole compounds has been developed that leverages nickel-catalyzed carbonylation cyclization. This technology represents a significant leap forward in synthetic methodology, offering a streamlined one-pot approach to constructing complex indole derivatives from readily available 2-alkynyl nitrobenzenes and arylboronic acid pinacol esters. The strategic importance of this innovation cannot be overstated, given that indole motifs are integral to a wide array of therapeutic agents ranging from antiviral drugs like Arbidol to anti-tumor and anti-inflammatory agents. By utilizing a nickel catalyst system combined with a cobalt carbonyl carbon monoxide source, this method circumvents many of the logistical and economic hurdles associated with traditional carbonylation processes, positioning it as a highly attractive option for the manufacture of high-purity pharmaceutical intermediates.
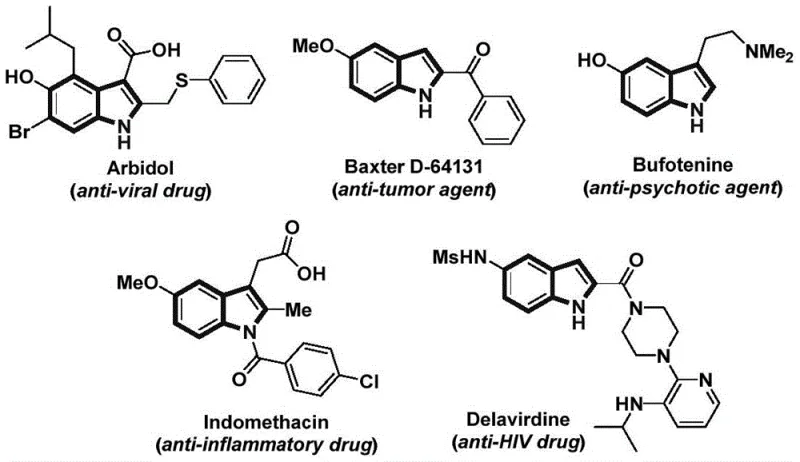
The structural diversity achievable through this synthetic route is exemplified by the broad spectrum of biologically active indole-containing compounds currently in clinical use or development. As illustrated in the reference data, molecules such as Arbidol, Baxter D-64131, and Indomethacin all share this critical heterocyclic core, underscoring the universal demand for efficient indole synthesis technologies. The ability to access these scaffolds through a unified, catalytic protocol allows medicinal chemists and process engineers to rapidly iterate on lead compounds without being bottlenecked by cumbersome synthetic steps. Furthermore, the compatibility of this method with various functional groups ensures that late-stage functionalization is feasible, thereby accelerating the drug discovery timeline. For procurement and supply chain stakeholders, understanding the versatility of this platform is crucial, as it implies a single, adaptable manufacturing process can serve multiple product lines, enhancing overall operational agility and reducing the complexity of the chemical supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of indole derivatives via carbonylation has been plagued by significant technical and economic challenges that hinder widespread industrial adoption. Traditional methods often rely on precious metal catalysts such as palladium, which not only incur substantial raw material costs but also pose severe contamination risks that necessitate expensive purification steps to meet stringent pharmaceutical purity standards. Moreover, many conventional carbonylation protocols require the direct use of carbon monoxide gas, a highly toxic and hazardous reagent that demands specialized high-pressure equipment and rigorous safety protocols, thereby inflating capital expenditure and operational overhead. Additionally, older synthetic routes frequently suffer from poor atom economy and limited substrate scope, often failing to tolerate sensitive functional groups or requiring harsh reaction conditions that lead to decomposition and low yields. These inefficiencies create a ripple effect throughout the supply chain, resulting in longer lead times, higher waste generation, and ultimately, increased costs for the final active pharmaceutical ingredient, making the search for alternative methodologies a top priority for forward-thinking chemical enterprises.
The Novel Approach
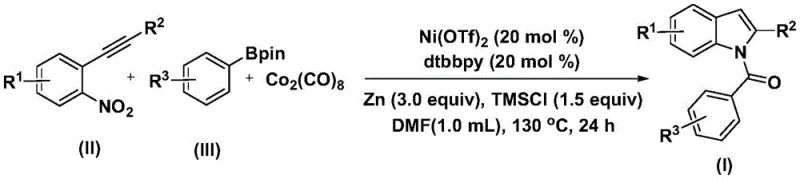
In stark contrast to these legacy issues, the methodology disclosed in patent CN115286553A introduces a transformative nickel-catalyzed system that effectively dismantles these barriers to entry. By substituting expensive palladium with earth-abundant nickel complexes, specifically nickel trifluoromethanesulfonate paired with a bipyridine ligand, the process achieves a dramatic reduction in catalyst cost while maintaining high catalytic activity. A particularly ingenious aspect of this invention is the use of cobalt carbonyl as a solid carbon monoxide substitute, which releases CO in situ under the reaction conditions, thereby eliminating the need for handling dangerous CO gas cylinders and enabling the reaction to proceed in standard laboratory glassware or reactors. The reaction operates efficiently at a moderate temperature of 130°C in polar aprotic solvents like DMF, demonstrating exceptional functional group tolerance across a wide range of substrates. This novel approach not only simplifies the operational workflow but also enhances the safety profile of the manufacturing process, making it an ideal candidate for the cost reduction in pharmaceutical intermediate manufacturing and facilitating the commercial scale-up of complex indole derivatives.
Mechanistic Insights into Nickel-Catalyzed Carbonylation Cyclization
A deep dive into the mechanistic underpinnings of this transformation reveals a sophisticated catalytic cycle that orchestrates the convergence of multiple reaction partners into the final indole architecture with high precision. The proposed mechanism initiates with the oxidative addition of the nickel catalyst into the arylboronic acid pinacol ester, generating a reactive aryl-nickel intermediate that serves as the foundation for subsequent bond-forming events. Crucially, the cobalt carbonyl additive acts as a reservoir for carbon monoxide, which inserts into the nickel-carbon bond to form an acyl-nickel species, effectively introducing the carbonyl functionality required for the amide linkage. Concurrently, the 2-alkynyl nitrobenzene substrate undergoes a reduction process mediated by the zinc reductant and trimethylsilyl chloride additive, converting the nitro group into a nucleophilic amine equivalent. This reduced species then attacks the electrophilic acyl-nickel intermediate, forging the critical carbon-nitrogen bond. The cycle concludes with a reductive elimination step that releases the linear amide precursor, which subsequently undergoes an intramolecular cyclization to yield the thermodynamically stable indole ring system. This intricate interplay of reduction, carbonylation, and cyclization within a single pot showcases the elegance of modern transition metal catalysis in constructing complex molecular architectures.
From a quality control and impurity profiling perspective, the robustness of this catalytic system offers distinct advantages for ensuring product integrity. The use of specific ligands such as 4,4'-di-tert-butyl-2,2'-bipyridine helps to stabilize the nickel center and suppresses off-cycle reactions that could lead to homocoupling byproducts or incomplete conversion. Furthermore, the stoichiometric balance of reagents, particularly the use of zinc as a terminal reductant and trimethylsilyl chloride as an activator, is optimized to drive the reaction to completion, minimizing the presence of unreacted starting materials in the crude mixture. The reaction conditions are sufficiently mild to prevent the degradation of sensitive substituents, such as halogens or alkoxy groups, which might otherwise decompose under more forcing conditions typical of classical indole syntheses. Consequently, the resulting crude product typically exhibits a cleaner impurity profile, simplifying the downstream purification process and reducing the burden on analytical teams to identify and quantify trace impurities. This level of control is paramount for a reliable pharmaceutical intermediate supplier, as it ensures consistent batch-to-batch reproducibility and adherence to strict regulatory specifications.
How to Synthesize Indole Compounds Efficiently
The practical implementation of this nickel-catalyzed protocol is designed to be accessible and scalable, bridging the gap between academic discovery and industrial application. The procedure involves a straightforward charging of reagents into a reaction vessel, followed by heating and a standard workup sequence, making it amenable to automation and large-batch processing. The detailed standardized synthesis steps below outline the precise molar ratios, solvent choices, and thermal parameters required to achieve optimal yields and purity, serving as a vital reference for process chemists looking to adopt this technology. By adhering to these guidelines, manufacturers can leverage the full potential of this method to produce high-value indole intermediates with maximum efficiency.
- Combine nickel catalyst, nitrogen ligand, reducing agent, additive, carbon monoxide substitute, 2-alkynyl nitrobenzene, and arylboronic acid pinacol ester in an organic solvent.
- Heat the reaction mixture to 130°C and maintain for 24 hours to ensure complete conversion.
- Perform post-treatment including filtration and silica gel mixing, followed by column chromatography purification to isolate the target indole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this nickel-catalyzed synthesis route offers a compelling value proposition centered on cost optimization and supply security. The shift from precious metals to base metals fundamentally alters the cost structure of the synthesis, removing the volatility associated with palladium pricing and reducing the overall bill of materials. Additionally, the reliance on commercially available and stable starting materials, such as 2-alkynyl nitrobenzenes and arylboronic acid pinacol esters, ensures a resilient supply chain that is less susceptible to geopolitical disruptions or raw material shortages. The simplified operational requirements, which eliminate the need for high-pressure gas infrastructure, further contribute to significant capital expenditure savings and lower facility maintenance costs. These factors collectively enhance the economic viability of producing indole-based APIs, allowing companies to remain competitive in a margin-sensitive market while maintaining high standards of quality and safety.
- Cost Reduction in Manufacturing: The substitution of expensive palladium catalysts with nickel trifluoromethanesulfonate represents a direct and substantial decrease in catalyst costs, which is a major component of the overall production expense for many fine chemicals. Furthermore, the use of cobalt carbonyl as a solid CO source eliminates the need for specialized gas handling equipment and the associated safety compliance costs, leading to broader operational savings. The high reaction efficiency and yield reported in the patent data imply that less raw material is wasted, improving the overall mass balance and reducing the cost per kilogram of the final product. These cumulative savings can be reinvested into R&D or passed on to customers, strengthening the company's market position as a cost-effective partner.
- Enhanced Supply Chain Reliability: The starting materials for this reaction, including the nitrobenzene derivatives and boronic esters, are commodity chemicals that are widely produced and available from multiple global suppliers, mitigating the risk of single-source dependency. The robustness of the reaction conditions means that production is less likely to be interrupted by minor fluctuations in utility supplies or environmental conditions, ensuring a steady flow of goods to downstream customers. Moreover, the simplified workup procedure, which involves basic filtration and chromatography, reduces the turnaround time between batches, allowing for more responsive inventory management and shorter lead times for urgent orders. This reliability is critical for maintaining continuous manufacturing operations and meeting the just-in-time delivery expectations of major pharmaceutical clients.
- Scalability and Environmental Compliance: The protocol is inherently scalable, having been demonstrated to work effectively in standard solvents like DMF at temperatures that are easily achievable in large-scale reactors without exotic cooling or heating requirements. The avoidance of toxic carbon monoxide gas significantly lowers the environmental, health, and safety (EHS) footprint of the process, simplifying permitting and regulatory approval for new manufacturing lines. Additionally, the high atom economy and reduced waste generation align with green chemistry principles, helping companies meet their sustainability goals and reduce the costs associated with waste disposal and treatment. This alignment with modern environmental standards future-proofs the manufacturing process against increasingly stringent global regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this indole synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent literature, offering clarity on the practical aspects of the method. Understanding these details is essential for technical teams evaluating the feasibility of integrating this process into their existing manufacturing portfolios.
Q: What are the key advantages of this nickel-catalyzed method over traditional palladium catalysis?
A: This method utilizes earth-abundant nickel catalysts instead of expensive palladium, significantly reducing raw material costs. It also employs cobalt carbonyl as a safe carbon monoxide substitute, avoiding the handling of toxic CO gas directly, which enhances operational safety and simplifies regulatory compliance.
Q: What is the substrate scope for this indole synthesis reaction?
A: The reaction demonstrates excellent functional group tolerance. It accommodates various substituents on the 2-alkynyl nitrobenzene and the arylboronic acid pinacol ester, including alkyl, alkoxy, halogen, and trifluoromethyl groups, allowing for the diverse synthesis of complex indole scaffolds required in drug discovery.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is designed for scalability. It uses commercially available starting materials, operates at a moderate temperature of 130°C in standard solvents like DMF, and involves a straightforward workup procedure involving filtration and column chromatography, making it highly viable for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indole Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methods like the nickel-catalyzed carbonylation described in CN115286553A for the production of high-value pharmaceutical intermediates. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are seamlessly translated into robust industrial processes. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which employ state-of-the-art analytical techniques to verify identity and assess impurity profiles. By partnering with us, you gain access to a supply chain that is not only cost-effective but also technically superior, capable of handling the complexities of modern drug synthesis with precision and reliability.
We invite you to engage with our technical procurement team to discuss how this specific synthesis route can be tailored to your project needs. Whether you require a Customized Cost-Saving Analysis for your current supply chain or need to evaluate the technical feasibility of a new indole derivative, our experts are ready to provide comprehensive support. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and quality can drive success for your organization.
