Advanced Carbonylation Strategy for Scalable Thioester Production in Pharmaceutical Manufacturing
Advanced Carbonylation Strategy for Scalable Thioester Production in Pharmaceutical Manufacturing
The landscape of organic synthesis is constantly evolving to meet the rigorous demands of the pharmaceutical industry, particularly regarding the efficient production of key structural motifs like thioesters. Patent CN113004181B introduces a groundbreaking methodology for preparing thioester compounds through a transition metal-catalyzed carbonylation reaction. This innovation addresses long-standing challenges in the field by utilizing inexpensive benzyl chloride compounds and sulfonyl chlorides as primary starting materials. Unlike traditional routes that rely on volatile and malodorous thiols, this novel approach leverages tungsten carbonyl as both a carbonyl source and a reducing agent. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this technology represents a significant leap forward in process safety and economic viability. The method demonstrates exceptional functional group tolerance and high reaction efficiency, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of thioester compounds has been predominantly achieved through the direct acylation of thiols with carboxylic acids or their derivatives. While chemically straightforward, this conventional pathway suffers from severe practical drawbacks that hinder its application in large-scale manufacturing. The primary issue lies in the nature of thiol reagents, which possess extremely unpleasant, pervasive odors that pose significant health and safety risks in industrial settings. Furthermore, thiols are potent catalyst poisons; they readily coordinate with transition metals, deactivating the catalysts required for many sophisticated transformations and leading to inconsistent reaction yields. Alternative methods involving the oxidative coupling of aldehydes or substitution reactions of halogenated alkanes often require harsh conditions or generate substantial waste. These limitations create bottlenecks in cost reduction in fine chemical manufacturing, as extensive safety measures and waste treatment protocols become necessary to handle toxic sulfur sources.
The Novel Approach
The methodology disclosed in the patent offers a transformative solution by replacing problematic thiols with stable and odorless sulfonyl chlorides. This strategic shift not only eliminates the olfactory hazards associated with thiols but also prevents catalyst poisoning, thereby enhancing the overall robustness of the synthetic route. The reaction employs a palladium catalyst system supported by a Xantphos ligand, which facilitates the coupling of benzyl chlorides with sulfonyl chlorides under carbonylation conditions. Crucially, the use of tungsten carbonyl [W(CO)6] simplifies the reagent profile by serving a dual purpose, removing the need for separate carbon monoxide gas sources or additional reducing agents. This streamlined approach results in a cleaner reaction profile with fewer byproducts. As illustrated in the general reaction scheme below, the transformation proceeds efficiently to yield the target thioester structure.
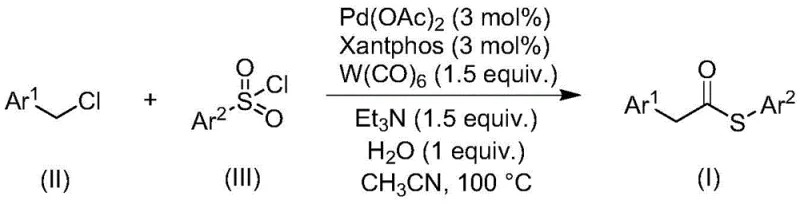
The versatility of this novel approach is further evidenced by its compatibility with a wide range of substrates. The reaction tolerates various substituents on both the benzyl chloride and the sulfonyl chloride components, including electron-donating and electron-withdrawing groups. This broad substrate scope allows chemists to access a diverse library of thioester derivatives without needing to redesign the core synthetic strategy for each new analog. Such flexibility is paramount for medicinal chemistry campaigns where rapid iteration of molecular structures is required. By avoiding the use of gaseous CO and toxic thiols, the process inherently reduces the regulatory burden and operational complexity typically associated with carbonylation reactions.
Mechanistic Insights into Palladium-Catalyzed Carbonylation
The success of this transformation relies on a sophisticated catalytic cycle driven by the palladium-Xantphos complex. The mechanism likely initiates with the oxidative addition of the benzyl chloride to the active Pd(0) species, forming a benzyl-palladium(II) intermediate. Subsequently, the insertion of carbon monoxide, released in situ from the decomposition of tungsten carbonyl, generates an acyl-palladium species. The presence of the bulky and electron-rich Xantphos ligand is critical here, as it stabilizes the palladium center and promotes the reductive elimination step. Simultaneously, the sulfonyl chloride undergoes reduction, facilitated by the tungsten carbonyl which acts as the oxygen acceptor and reducing equivalent, eventually generating the sulfur nucleophile in situ. This nucleophile then attacks the acyl-palladium complex or intercepts the acyl intermediate to form the final thioester bond. The precise interplay between the palladium catalyst and the tungsten reductant ensures high turnover numbers and minimizes side reactions such as homocoupling of the benzyl halide.
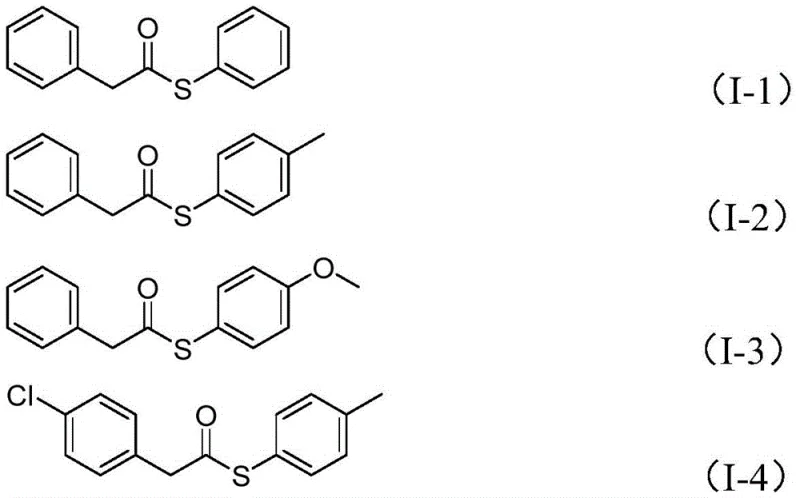
From an impurity control perspective, this mechanism offers distinct advantages over thiol-based routes. In traditional methods, the oxidation of thiols to disulfides is a common and difficult-to-remove side reaction that compromises product purity. By generating the sulfur component from a sulfonyl chloride in a controlled manner within the coordination sphere of the metal, the formation of disulfide impurities is significantly suppressed. Furthermore, the use of water as a co-reagent in specific stoichiometric ratios helps to moderate the reaction environment, potentially hydrolyzing reactive intermediates that could lead to polymeric byproducts. The result is a crude reaction mixture that is easier to purify, often requiring only standard silica gel chromatography to achieve high-purity specifications. This level of control over the impurity profile is essential for meeting the stringent quality standards required for API intermediates.
How to Synthesize Thioester Compounds Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent ratios and reaction conditions to maximize yield. The protocol involves charging a reaction vessel with the benzyl chloride derivative, the sulfonyl chloride, palladium acetate, Xantphos, tungsten carbonyl, triethylamine, and a stoichiometric amount of water in an aprotic solvent like acetonitrile. The mixture is then heated to 100°C for approximately 24 hours. The detailed standardized synthesis steps, including specific molar ratios and workup procedures validated across multiple examples, are provided in the guide below.
- Charge a reaction vessel with palladium acetate, Xantphos ligand, tungsten carbonyl, triethylamine, water, benzyl chloride derivative, and sulfonyl chloride in an organic solvent such as acetonitrile.
- Heat the reaction mixture to 100°C and maintain stirring for approximately 24 hours to ensure complete conversion of the starting materials into the desired thioester product.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the high-purity thioester compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this carbonylation technology translates into tangible operational benefits that extend beyond mere chemical yield. The shift away from hazardous thiols and gaseous carbon monoxide simplifies the logistics of raw material handling and storage. Solid or liquid reagents like sulfonyl chlorides and tungsten carbonyl are far easier to transport and dose accurately compared to pressurized gases or volatile sulfurs. This stability enhances supply chain reliability, reducing the risk of production delays caused by specialized shipping requirements or regulatory hold-ups on dangerous goods. Furthermore, the simplicity of the post-treatment process, which avoids complex extraction or distillation steps often needed to remove foul-smelling thiols, accelerates the overall manufacturing cycle time.
- Cost Reduction in Manufacturing: The economic impact of this method is driven by the elimination of expensive and specialized reagents. By utilizing tungsten carbonyl as a combined carbonyl source and reducing agent, the process removes the need for purchasing separate reducing agents or installing high-pressure CO infrastructure. Additionally, the starting materials—benzyl chlorides and sulfonyl chlorides—are commodity chemicals available at low cost from multiple global suppliers. The high atom economy and reduced waste generation further contribute to substantial cost savings, as less solvent and energy are required for purification and waste disposal compared to traditional acylation methods.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent output regardless of minor fluctuations in raw material quality. Because the catalyst system is resistant to poisoning by the sulfur source, batch-to-batch variability is minimized. This consistency is crucial for maintaining continuous supply lines for downstream API synthesis. The use of stable, non-volatile starting materials also means that inventory can be held for longer periods without degradation, allowing manufacturers to buffer against market volatility and ensure uninterrupted production schedules for critical pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling this reaction from gram to kilogram or tonne scale is straightforward due to the absence of hazardous gas feeds. The reaction operates at atmospheric pressure (in a closed system) and moderate temperatures, reducing the engineering controls required for high-pressure reactors. From an environmental standpoint, avoiding thiols significantly reduces the emission of volatile organic compounds (VOCs) with objectionable odors, facilitating compliance with strict environmental regulations. The simpler workup procedure generates less aqueous and organic waste, aligning with green chemistry principles and lowering the overall environmental footprint of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this thioester synthesis method. These answers are derived directly from the experimental data and mechanistic understanding provided in the patent documentation. Understanding these nuances is vital for process chemists aiming to adapt this technology to their specific production needs.
Q: Why is sulfonyl chloride preferred over thiols in this synthesis?
A: Sulfonyl chlorides are used as the sulfur source to completely avoid the use of thiol compounds, which are notorious for their unpleasant odor and tendency to poison transition metal catalysts, thereby improving operational safety and catalyst longevity.
Q: What is the dual role of tungsten carbonyl in this reaction?
A: Tungsten carbonyl [W(CO)6] serves a critical dual function: it acts as the carbonyl source for the formation of the ester linkage and simultaneously functions as the reducing agent, eliminating the need for additional external reducing reagents.
Q: Is this carbonylation method suitable for large-scale production?
A: Yes, the method utilizes inexpensive and readily available starting materials like benzyl chlorides and operates under relatively mild conditions (100°C) with simple post-treatment procedures, making it highly amenable to commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thioester Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in accelerating drug development timelines. Our team of expert process chemists has thoroughly evaluated the carbonylation technology described in CN113004181B and is fully prepared to leverage it for your projects. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to manufacturing plant is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of thioester intermediate delivered meets the highest industry standards for pharmaceutical applications.
We invite you to collaborate with us to optimize this route for your specific target molecules. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage potential partners to contact us directly to discuss specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability.
