Advanced Palladium-Catalyzed Carbonylation Strategy for Scalable Thioester Manufacturing
Advanced Palladium-Catalyzed Carbonylation Strategy for Scalable Thioester Manufacturing
The landscape of organic synthesis is constantly evolving to meet the rigorous demands of modern pharmaceutical and fine chemical production, particularly regarding safety, efficiency, and scalability. A significant breakthrough in this domain is detailed in patent CN113004181B, which discloses a novel method for preparing thioester compounds through a transition metal-catalyzed carbonylation reaction. This technology represents a paradigm shift by replacing traditional, hazardous sulfur sources with stable sulfonyl chlorides, thereby addressing long-standing issues related to odor, toxicity, and catalyst deactivation. By leveraging a sophisticated catalytic system comprising palladium acetate, Xantphos ligand, and tungsten hexacarbonyl, this process achieves high reaction efficiency under relatively mild conditions. For R&D directors and procurement managers seeking a reliable thioester supplier, this methodology offers a compelling value proposition, combining robust chemical performance with streamlined operational protocols that are essential for cost reduction in pharmaceutical intermediate manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of thioester compounds has relied heavily on the direct acylation of thiols with carboxylic acids or their derivatives, a pathway fraught with significant practical and safety challenges. Thiols are notoriously malodorous, posing severe health risks and requiring specialized containment infrastructure to prevent environmental contamination and worker exposure. Furthermore, sulfur-containing compounds like thiols are potent catalyst poisons; they frequently coordinate strongly with transition metal centers, leading to rapid deactivation of expensive catalysts and necessitating higher loadings or frequent replenishment. Alternative methods involving the oxidative coupling of aldehydes or substitution reactions of halogenated alkanes often suffer from poor atom economy, harsh reaction conditions, or limited substrate scope. These conventional routes frequently generate complex impurity profiles that are difficult to separate, complicating downstream purification and increasing the overall cost of goods sold. Consequently, the industry has long sought a safer, more efficient alternative that maintains high yield while eliminating the logistical burden of handling volatile sulfur species.
The Novel Approach
The innovative strategy outlined in the patent data circumvents these traditional bottlenecks by utilizing sulfonyl chlorides as the sulfur source instead of thiols. This substitution is transformative because sulfonyl chlorides are stable, odorless solids that are commercially available and easy to handle, drastically improving the safety profile of the manufacturing process. The reaction employs a unique catalytic system where tungsten hexacarbonyl acts dually as the carbonyl source and the reducing agent, eliminating the need for external carbon monoxide gas cylinders or additional stoichiometric reductants. This integration simplifies the reactor setup and reduces capital expenditure on high-pressure gas handling equipment. Moreover, the use of benzyl chloride compounds as the electrophilic partners expands the synthetic utility beyond simple aryl halides, allowing for the incorporation of diverse functional groups. The result is a highly designable synthetic route that tolerates a wide range of substituents, including alkyl, alkoxy, and halogen groups, ensuring that complex molecular architectures can be accessed without compromising yield or purity.
Mechanistic Insights into Pd-Catalyzed Carbonylative Thioesterification
The core of this technological advancement lies in the intricate interplay between the palladium catalyst and the tungsten carbonyl species within the reaction medium. The mechanism initiates with the oxidative addition of the benzyl chloride substrate to the active palladium(0) species, generated in situ from palladium acetate and the bidentate phosphine ligand Xantphos. This step is critical as it activates the carbon-chlorine bond, setting the stage for carbonyl insertion. Subsequently, carbon monoxide is released from the tungsten hexacarbonyl complex and inserts into the palladium-carbon bond to form an acyl-palladium intermediate. Unlike traditional carbonylations that require external CO pressure, this system generates CO internally, maintaining a steady, low concentration that favors the desired transformation while minimizing side reactions. The sulfonyl chloride then participates in the cycle, likely undergoing reduction and sulfur transfer to intercept the acyl-palladium species, ultimately releasing the thioester product and regenerating the catalyst. This elegant cascade ensures that the sulfur atom is incorporated efficiently without the formation of disulfide byproducts commonly associated with thiol oxidation.
From an impurity control perspective, this mechanism offers distinct advantages over thiol-based routes. In conventional syntheses, the oxidation of thiols to disulfides is a pervasive side reaction that consumes raw materials and creates impurities with similar polarity to the product, making purification arduous. By employing sulfonyl chlorides, the reaction pathway bypasses the free thiol intermediate entirely, thereby suppressing disulfide formation at the source. Additionally, the specific choice of Xantphos as a ligand provides a wide bite angle that stabilizes the palladium center against aggregation and decomposition, which is crucial for maintaining catalytic activity over the extended reaction time of 24 hours. The presence of water and triethylamine further modulates the reaction environment, facilitating the reduction of the sulfonyl chloride and neutralizing acidic byproducts like HCl. This precise control over the reaction microenvironment results in a cleaner crude reaction mixture, significantly reducing the burden on downstream purification processes and enhancing the overall mass balance of the operation.
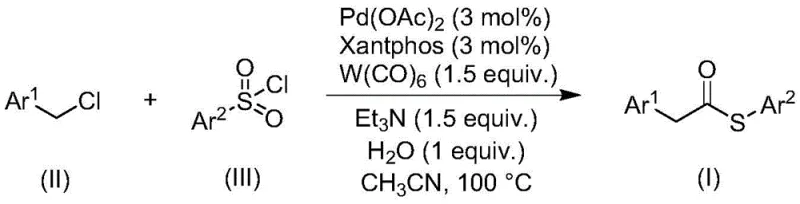
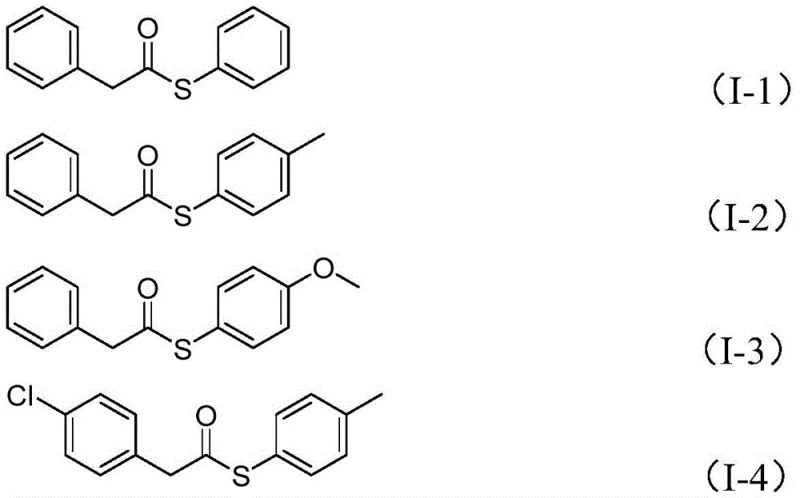
To illustrate the versatility of this catalytic system, the patent data provides several specific examples demonstrating the successful synthesis of various thioester derivatives. As shown in the structural examples below, the method accommodates a broad spectrum of electronic and steric environments on both the benzyl and sulfonyl components. For instance, electron-rich substrates bearing methoxy groups and electron-deficient substrates with chloro or fluoro substituents both proceed to give the corresponding thioesters in good yields. The tolerance extends to polycyclic aromatic systems like naphthalene, indicating that the catalyst system is robust enough to handle sterically demanding substrates. This breadth of scope is vital for medicinal chemists who require rapid access to diverse libraries of thioester intermediates for structure-activity relationship (SAR) studies. The ability to tune the Ar1 and Ar2 groups independently allows for the fine-tuning of the physicochemical properties of the final molecule, making this method a powerful tool for the late-stage functionalization of complex drug candidates.
How to Synthesize Thioester Compounds Efficiently
Implementing this carbonylation protocol in a laboratory or pilot plant setting requires careful attention to reagent ratios and reaction parameters to maximize yield and reproducibility. The standard procedure involves charging a reaction vessel with the benzyl chloride and sulfonyl chloride substrates alongside the catalytic system consisting of palladium acetate and Xantphos. Crucially, tungsten hexacarbonyl is added as the solid CO source, along with triethylamine as the base and a stoichiometric amount of water to facilitate the reduction process. The reaction is typically conducted in acetonitrile, which has been identified as the optimal solvent for solubilizing the diverse range of reactants while promoting high conversion rates. Heating the mixture to 100 °C for 24 hours allows the slow release of CO and the subsequent catalytic turnover to reach completion. Following the reaction, the workup is remarkably straightforward, involving simple filtration to remove metal residues followed by silica gel treatment and column chromatography. For a detailed, step-by-step guide on optimizing these parameters for your specific substrate, please refer to the standardized synthesis protocol below.
- Charge a reaction vessel with palladium acetate, Xantphos ligand, tungsten hexacarbonyl, triethylamine, water, benzyl chloride, and sulfonyl chloride in an organic solvent like acetonitrile.
- Heat the reaction mixture to 100 °C and maintain stirring for approximately 24 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the high-purity thioester product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel carbonylation technology translates into tangible strategic benefits that extend far beyond mere chemical curiosity. The primary driver for cost optimization lies in the elimination of thiols from the supply chain. Thiols are not only expensive due to their specialized handling requirements but also introduce significant hidden costs related to waste disposal and odor control infrastructure. By switching to sulfonyl chlorides, which are commodity chemicals with stable pricing and abundant global supply, manufacturers can achieve substantial cost savings in raw material acquisition. Furthermore, the dual functionality of tungsten hexacarbonyl as both a carbonyl source and a reducing agent simplifies the bill of materials, reducing the number of SKUs that need to be sourced and managed. This consolidation of reagents streamlines inventory management and reduces the risk of supply disruptions caused by the shortage of niche reducing agents. The operational simplicity of the process, which avoids high-pressure gas equipment, also lowers the barrier to entry for contract manufacturing organizations, increasing the pool of potential suppliers and fostering a more competitive pricing environment.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the removal of expensive and hazardous thiol reagents, which often command a premium price due to safety regulations. By utilizing inexpensive benzyl chlorides and sulfonyl chlorides, the direct material cost is significantly lowered. Additionally, the absence of a need for external carbon monoxide gas removes the capital expenditure associated with high-pressure reactors and gas monitoring systems, leading to lower depreciation costs per unit of production. The high atom economy of the reaction, coupled with the efficient use of the palladium catalyst at low loadings (3 mol%), ensures that precious metal costs are kept to a minimum. These factors combine to create a leaner manufacturing process that delivers a superior cost-of-goods-sold profile compared to legacy thiol-based methods.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available starting materials that are produced on a multi-ton scale by numerous chemical vendors globally. Benzyl chlorides and sulfonyl chlorides are staple intermediates in the fine chemical industry, meaning their supply is less susceptible to the volatility seen with specialized sulfur reagents. The robustness of the reaction conditions, which tolerate a wide range of functional groups, means that slight variations in raw material quality are less likely to cause batch failures, thereby improving first-pass yield rates. This reliability reduces the need for safety stock and allows for more agile response to market demand fluctuations. Furthermore, the simplified workup procedure shortens the overall production cycle time, enabling faster turnaround from order to delivery and improving cash flow dynamics for the manufacturing partner.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram and eventually to metric ton quantities is facilitated by the homogeneous nature of the reaction and the absence of gaseous reagents that pose mixing and mass transfer challenges at large scales. The use of acetonitrile, a common industrial solvent with well-established recovery and recycling protocols, aligns with green chemistry principles and simplifies solvent waste management. Avoiding the generation of malodorous thiol waste streams significantly reduces the environmental footprint of the facility, lowering the costs associated with scrubber maintenance and regulatory compliance reporting. The process generates minimal hazardous waste, primarily consisting of spent catalyst and inorganic salts, which can be treated using standard effluent treatment procedures. This environmental compatibility ensures long-term operational sustainability and mitigates the risk of regulatory shutdowns due to emissions violations.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis method is crucial for stakeholders evaluating its potential for integration into existing production lines. The following questions address common inquiries regarding the practical implementation and scope of this technology, derived directly from the experimental data and technical specifications provided in the patent literature. These insights are intended to clarify the operational boundaries and capabilities of the process, ensuring that R&D and production teams have a clear understanding of what to expect during technology transfer. By addressing these points proactively, we aim to facilitate a smoother adoption curve and maximize the value derived from this innovative synthetic route.
Q: What is the primary advantage of using sulfonyl chlorides over thiols in this synthesis?
A: Using sulfonyl chlorides eliminates the handling of unpleasant-smelling and toxic thiol compounds, which often poison catalysts. This significantly improves operational safety and environmental compliance during manufacturing.
Q: Does this process require an external carbon monoxide gas source?
A: No, the process utilizes tungsten hexacarbonyl (W(CO)6) which serves a dual function as both the carbonyl source and the reducing agent, removing the need for hazardous high-pressure CO gas equipment.
Q: What is the substrate scope for the aryl groups in this reaction?
A: The method demonstrates wide functional group tolerance, successfully accommodating substituted phenyl, naphthyl, and heteroaryl groups with substituents such as methyl, methoxy, halogens, and esters.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thioester Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory discovery to commercial reality requires a partner with deep technical expertise and a commitment to excellence. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent CN113004181B can be reliably translated into industrial success. We understand that high-purity thioester compounds are critical intermediates for the synthesis of complex APIs and bioconjugates, and our stringent purity specifications guarantee that every batch meets the rigorous demands of global pharmaceutical clients. Our state-of-the-art rigorous QC labs are equipped to perform comprehensive impurity profiling, ensuring that the unique advantages of this sulfonyl chloride-based route—such as the absence of disulfide contaminants—are fully realized in the final product. We are dedicated to providing a seamless supply chain experience that supports your innovation pipeline from early-stage development through to full-scale commercialization.
We invite you to engage with our technical team to explore how this advanced carbonylation technology can optimize your specific manufacturing needs. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits tailored to your volume requirements and target specifications. Our experts are ready to provide specific COA data and route feasibility assessments to demonstrate the viability of this method for your portfolio. Contact our technical procurement team today to discuss how we can collaborate to enhance your supply chain efficiency and drive down costs while maintaining the highest standards of quality and safety.
