Scalable Visible Light Photocatalysis for High-Purity 3-Aryl-2H-Indazole Manufacturing
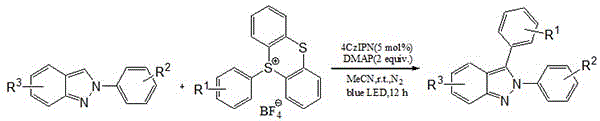
The pharmaceutical and fine chemical industries are constantly seeking robust, sustainable methodologies for constructing complex heterocyclic scaffolds essential for drug discovery. Patent CN114805209B introduces a groundbreaking visible light-induced protocol for the synthesis of 3-aryl-2H-indazole compounds, addressing critical limitations associated with traditional cross-coupling reactions. This innovative approach utilizes an organic photocatalyst, specifically 2,4,5,6-tetra(9-carbazolyl)-isophthalonitrile (4CzIPN), to facilitate the C3-arylation of 2-aryl-2H-indazoles using aryl sulfonium salts as radical precursors. By shifting away from noble metal catalysis, this technology offers a greener, more cost-effective pathway that aligns perfectly with modern green chemistry principles and stringent regulatory requirements for residual metal limits in active pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the functionalization of the C3 position on the indazole core has relied heavily on transition metal-catalyzed cross-coupling reactions, which present significant operational and economic challenges for large-scale manufacturing. These classical strategies typically necessitate the use of expensive palladium or copper catalysts, specialized ligands, and often require harsh reaction conditions such as elevated temperatures or strong bases to drive the transformation to completion. Furthermore, the inevitable presence of trace heavy metals in the final product necessitates rigorous and costly purification steps to meet pharmacopeial standards, creating a bottleneck in the supply chain for high-purity pharmaceutical intermediates. The reliance on stoichiometric oxidants in some variations also generates substantial chemical waste, complicating environmental compliance and increasing the overall carbon footprint of the synthesis.
The Novel Approach
In stark contrast, the methodology disclosed in the patent leverages visible light photoredox catalysis to achieve the same structural transformation under remarkably mild and benign conditions. By employing aryl sulfonium salts as stable and versatile aryl radical sources, the reaction proceeds efficiently at room temperature without the need for external heating or aggressive reagents. The use of the metal-free organic photocatalyst 4CzIPN not only eliminates the risk of heavy metal contamination but also simplifies the reaction setup, allowing for straightforward execution in standard laboratory or plant equipment equipped with blue LED arrays. This paradigm shift represents a significant advancement in synthetic efficiency, offering a streamlined route that reduces both the complexity of the operation and the environmental impact associated with the production of valuable indazole derivatives.
Mechanistic Insights into 4CzIPN-Catalyzed Radical Arylation
The core of this transformative synthesis lies in the unique photophysical properties of the 4CzIPN catalyst, which acts as a potent single-electron transfer agent upon excitation by blue light. When irradiated, the photocatalyst enters an excited state capable of reducing the aryl sulfonium salt substrate through a single-electron transfer process, thereby generating a highly reactive aryl radical species alongside a sulfur-centered byproduct. This aryl radical subsequently attacks the electron-rich C3 position of the 2-aryl-2H-indazole nucleus, forming a new carbon-carbon bond and establishing the desired 3-aryl substitution pattern with high regioselectivity. The presence of DMAP as an additive plays a crucial role in facilitating the reaction kinetics, likely by assisting in the deprotonation or stabilization of intermediate species, ensuring the catalytic cycle turns over efficiently to deliver the target molecule in high yields ranging from 71% to 85%.
From an impurity control perspective, this radical mechanism offers distinct advantages over ionic pathways that might suffer from competing side reactions or rearrangements. The specificity of the radical addition to the indazole ring minimizes the formation of regioisomers, while the mild conditions prevent the degradation of sensitive functional groups that might be present on either the indazole or the aryl sulfonium coupling partner. This inherent selectivity translates directly into a cleaner crude reaction profile, reducing the burden on downstream purification processes such as column chromatography or recrystallization. For process chemists, understanding this mechanism is vital for optimizing reaction parameters such as light intensity and concentration to maximize throughput while maintaining the high purity standards required for clinical and commercial applications.
How to Synthesize 3-Aryl-2H-Indazole Efficiently
Implementing this photocatalytic protocol requires careful attention to the specific molar ratios and reaction environment detailed in the patent to ensure reproducibility and optimal yield. The standard procedure involves dissolving the 2-aryl-2H-indazole substrate and the aryl sulfonium salt in acetonitrile, followed by the addition of the 4CzIPN catalyst and DMAP base under an inert nitrogen atmosphere to prevent oxygen quenching of the excited state. While the general concept is straightforward, precise control over the stoichiometry—specifically using a 1:1.5 ratio of indazole to sulfonium salt and 5 mol% catalyst loading—is critical for driving the reaction to completion within the standard 12-hour timeframe. Detailed standardized synthesis steps for replicating this high-efficiency transformation are provided in the technical guide below.
- Dissolve 2-aryl-2H-indazole and aryl sulfonium salt in acetonitrile solvent within a reaction vessel.
- Add the organic photocatalyst 4CzIPN (5 mol%) and the base DMAP (2 equivalents) to the mixture under a nitrogen atmosphere.
- Irradiate the reaction mixture with blue LEDs at room temperature for 12 hours, followed by extraction and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this visible light-induced synthesis route presents a compelling value proposition centered on cost optimization and supply security. By eliminating the dependency on volatile precious metal markets for palladium or rhodium catalysts, manufacturers can stabilize their raw material costs and mitigate the financial risks associated with metal price fluctuations. The simplified operational requirements, which do not demand high-pressure reactors or extreme thermal conditions, allow for the utilization of existing glass-lined or stainless steel infrastructure with minimal modification, thereby accelerating the timeline from process development to commercial production. This accessibility ensures a more reliable supply of critical indazole intermediates, reducing the lead time for high-purity pharmaceutical intermediates needed for urgent drug development programs.
- Cost Reduction in Manufacturing: The most significant economic driver of this technology is the complete removal of expensive transition metal catalysts and the associated ligands, which traditionally account for a substantial portion of the bill of materials for complex heterocyclic synthesis. Without the need for costly metal scavenging resins or extensive purification protocols to remove trace metals to ppm levels, the overall processing costs are drastically simplified, leading to substantial cost savings in pharmaceutical intermediate manufacturing. Furthermore, the use of commodity chemicals like acetonitrile and commercially available organic photocatalysts ensures that the input costs remain low and predictable, enhancing the overall margin profile for the final active ingredient.
- Enhanced Supply Chain Reliability: The robustness of the aryl sulfonium salt precursors and the stability of the organic photocatalyst contribute to a more resilient supply chain that is less susceptible to disruptions common with specialized metal reagents. Since the reaction operates at room temperature and atmospheric pressure, the safety profile is significantly improved, reducing the regulatory hurdles and insurance costs associated with hazardous high-energy processes. This operational safety allows for continuous or semi-continuous manufacturing setups, ensuring a steady flow of materials and enhancing the reliability of the supply chain for downstream API producers who depend on consistent quality and delivery schedules.
- Scalability and Environmental Compliance: Scaling this photochemical process is increasingly feasible with modern flow chemistry technologies and large-scale LED reactor arrays, allowing for the commercial scale-up of complex pharmaceutical intermediates without the linear increase in waste generation seen in batch thermal processes. The absence of heavy metals and stoichiometric oxidants means that the waste stream is significantly less toxic and easier to treat, aligning with strict environmental regulations and corporate sustainability goals. This green chemistry advantage not only facilitates faster regulatory approval but also positions the manufacturer as a preferred partner for global pharmaceutical companies prioritizing environmentally responsible sourcing strategies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this visible light-induced arylation technology, based on the specific data and embodiments disclosed in the patent literature. Understanding these nuances is essential for R&D teams evaluating the feasibility of integrating this route into their existing synthetic portfolios for indazole-based drug candidates. The answers provided reflect the proven capabilities of the method as demonstrated in the experimental examples, offering clarity on substrate scope and operational parameters.
Q: What are the primary advantages of this photocatalytic method over traditional transition metal catalysis?
A: This method eliminates the need for expensive and toxic transition metal catalysts like palladium or copper, significantly reducing heavy metal impurity risks and downstream purification costs while operating under mild room temperature conditions.
Q: What is the substrate scope for the aryl sulfonium salts in this reaction?
A: The process demonstrates excellent tolerance for various functional groups on the aryl sulfonium salt, including electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups such as cyano and ester functionalities.
Q: Is this process suitable for large-scale commercial production of pharmaceutical intermediates?
A: Yes, the use of stable organic photocatalysts, ambient temperature operation, and simple workup procedures makes this methodology highly amenable to scale-up for industrial manufacturing of complex indazole derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Aryl-2H-Indazole Supplier
As the demand for sophisticated heterocyclic building blocks continues to grow, NINGBO INNO PHARMCHEM stands ready to leverage advanced technologies like this visible light-induced protocol to deliver superior value to our global partners. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 3-aryl-2H-indazole meets the exacting standards required for clinical trials and commercial drug manufacturing, providing you with a secure and high-quality source of critical intermediates.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs and cost targets. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this metal-free methodology for your supply chain. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to being your trusted partner in the development and supply of next-generation pharmaceutical intermediates.
