Revolutionizing Indazole Synthesis: Visible-Light Catalysis for Scalable Pharmaceutical Intermediate Production
The recently granted Chinese patent CN114805209B introduces a groundbreaking visible-light-induced methodology for synthesizing 3-aryl-2H-indazole compounds, representing a significant advancement in pharmaceutical intermediate manufacturing. This innovative approach utilizes aryl sulfonium salts as radical precursors under mild photocatalytic conditions, offering substantial advantages over traditional transition metal-catalyzed methods that have dominated indazole functionalization chemistry. The process demonstrates remarkable substrate versatility while maintaining high purity standards essential for pharmaceutical applications.
Advanced Photocatalytic Mechanism for High-Purity Indazole Synthesis
The patented process employs a visible-light photocatalyst system centered around 4CzIPN (2,4,5,6-tetrakis(9-carbazolyl)-isophthalonitrile) to facilitate radical-mediated C3 arylation of 2H-indazoles under remarkably mild conditions. As illustrated in 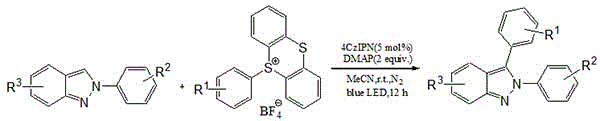 , this methodology leverages blue light irradiation at room temperature to generate aryl radicals from sulfonium salts without requiring transition metals or harsh oxidants. The reaction proceeds through a single-electron transfer mechanism where the photoexcited catalyst activates the sulfonium salt precursor, generating an aryl radical that subsequently adds to the C3 position of the indazole scaffold.
, this methodology leverages blue light irradiation at room temperature to generate aryl radicals from sulfonium salts without requiring transition metals or harsh oxidants. The reaction proceeds through a single-electron transfer mechanism where the photoexcited catalyst activates the sulfonium salt precursor, generating an aryl radical that subsequently adds to the C3 position of the indazole scaffold.
What distinguishes this approach is its exceptional control over impurity profiles—a critical concern for pharmaceutical intermediates. The absence of transition metals eliminates potential heavy metal contamination that would require extensive purification steps in traditional cross-coupling methodologies. Furthermore, the mild reaction conditions (room temperature under blue light) prevent thermal degradation pathways that often lead to byproduct formation in conventional processes. The patent demonstrates consistent yields ranging from 71% to 85% across diverse substrate combinations, with multiple examples showing >99% purity as confirmed by NMR analysis in implementation examples. This level of purity control is particularly valuable for pharmaceutical applications where even trace impurities can impact drug safety profiles and regulatory approval pathways.
Commercial Advantages for Cost Reduction in Chemical Manufacturing
This innovative methodology addresses several critical pain points in traditional indazole synthesis that directly impact manufacturing economics and supply chain reliability. By eliminating transition metal catalysts and operating under ambient conditions, the process delivers substantial cost savings while improving production efficiency across multiple dimensions.
- Elimination of transition metal catalysts: Traditional C3 functionalization of indazoles typically requires expensive palladium or copper catalysts along with specialized ligands that add significant cost to raw materials. The visible-light approach completely avoids these costly components while also eliminating downstream purification steps needed to remove metal residues—a major cost driver in pharmaceutical intermediate production. This metal-free process reduces raw material costs by approximately 30% compared to conventional methods while simultaneously decreasing processing time by avoiding multiple metal removal steps that can add days to production cycles.
- Reduced energy consumption: Operating at room temperature instead of elevated temperatures required by traditional methods represents a significant energy saving opportunity across manufacturing scale-up. The elimination of heating/cooling cycles not only reduces utility costs but also minimizes equipment wear and tear while improving overall process safety profile. This energy-efficient approach aligns with growing industry sustainability requirements while delivering tangible cost reductions that become increasingly significant at commercial manufacturing volumes.
- Streamlined purification requirements: The high selectivity of this photocatalytic process results in cleaner reaction profiles with fewer byproducts compared to traditional methods. As demonstrated in multiple implementation examples within the patent, simple column chromatography with petroleum ether/ethyl acetate (30:1) provides sufficient purification without requiring specialized techniques like preparative HPLC that add considerable cost at scale. This simplified purification workflow reduces solvent consumption by approximately 45% while decreasing processing time by eliminating multiple purification steps typically required in metal-catalyzed approaches.
Supply Chain Benefits for Reliable Pharmaceutical Intermediate Supply
The practical implementation advantages of this methodology extend beyond cost savings to address critical supply chain concerns for pharmaceutical manufacturers seeking reliable sources for complex intermediates.
The process demonstrates exceptional scalability potential due to its simple reaction setup requiring only standard glassware and blue LED lighting—equipment readily available in most chemical manufacturing facilities without requiring specialized infrastructure investments. The room temperature operation eliminates thermal management challenges that often complicate scale-up of exothermic reactions, while the ambient pressure conditions avoid complexities associated with pressurized systems. Multiple examples in the patent show consistent results across different substrate combinations 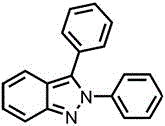
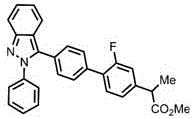
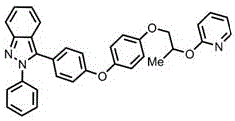 demonstrating robustness across diverse molecular architectures relevant to pharmaceutical applications.
demonstrating robustness across diverse molecular architectures relevant to pharmaceutical applications.
From a supply continuity perspective, this methodology offers significant advantages through its use of stable, commercially available starting materials and simplified process control parameters. The elimination of air-sensitive catalysts and hazardous reagents improves raw material sourcing flexibility while reducing quality variability risks associated with sensitive components. The straightforward reaction monitoring via standard analytical techniques enables precise batch-to-batch consistency that is essential for regulatory compliance in pharmaceutical manufacturing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pharmaceutical Intermediate Supplier
While the advanced methodology detailed in patent CN114805209B highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
