Advanced Visible Light Catalytic Synthesis of 3-Trifluoroalkylquinoxalinone for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more efficient and sustainable pathways to access complex heterocyclic scaffolds, particularly those incorporating fluorine atoms which are critical for enhancing metabolic stability and bioavailability in drug candidates. Patent CN112142680B introduces a groundbreaking methodology for the synthesis of 3-trifluoroalkylquinoxalinone compounds utilizing visible light catalysis. This technology represents a significant paradigm shift from traditional thermal or stoichiometric oxidation methods, offering a mild, metal-free, and environmentally benign route. By leveraging organic photocatalysts and air as the terminal oxidant, this process operates at room temperature in a mixed solvent system containing water, drastically reducing the energy footprint and hazardous waste generation associated with conventional synthetic routes. For R&D directors and procurement managers, this patent outlines a robust strategy for producing high-purity pharmaceutical intermediates with improved cost-efficiency and supply chain reliability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoroalkylated quinoxalinone skeletons has been fraught with operational challenges and safety concerns. Early methodologies, such as those reported by the Studer group, relied heavily on the use of trifluoroiodomethane gas as the trifluoromethyl source. As illustrated in the reaction scheme below, this approach necessitates the handling of hazardous gaseous reagents and requires strict inert gas protection, typically using argon, to prevent side reactions and ensure safety. Furthermore, these reactions often demand strong bases like DBU in stoichiometric amounts, which complicates the workup procedure and generates significant chemical waste. The requirement for specialized gas handling infrastructure and the inherent risks associated with pressurized gases make such methods less attractive for large-scale commercial manufacturing.
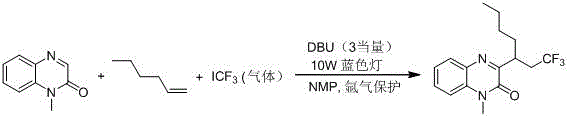
Subsequent developments attempted to address the gas handling issues by employing solid sodium trifluoromethyl sulfinate as an alternative trifluoromethyl source. However, as depicted in the following reaction scheme, these methods introduced new limitations, primarily the reliance on stoichiometric amounts of strong inorganic oxidants such as potassium persulfate or hypervalent iodine reagents. These reactions typically require elevated temperatures, often around 80°C, to proceed effectively. The use of excess oxidants not only increases the raw material costs but also leads to the formation of inorganic salt byproducts that complicate purification and pose environmental disposal challenges. The combination of heating requirements and heavy oxidant usage results in a process that is energy-intensive and generates a higher E-factor, making it less suitable for green chemistry initiatives and cost-sensitive production environments.
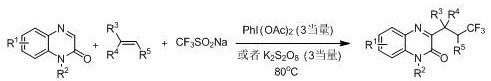
The Novel Approach
In stark contrast to the aforementioned limitations, the method disclosed in patent CN112142680B offers a streamlined and sustainable solution. The core innovation lies in the use of visible light to drive the reaction at room temperature using air as the sole oxidant. The general reaction scheme demonstrates the convergence of quinoxalinone, various olefins, and sodium trifluoromethyl sulfinate in the presence of a catalytic amount of an organic dye or metal-free photocatalyst. This approach eliminates the need for hazardous gases, strong bases, and stoichiometric oxidants. The ability to run the reaction in a mixture of organic solvent and water further enhances the green credentials of the process. By operating under ambient conditions, the method significantly reduces energy consumption related to heating and cooling, while the use of air avoids the cost and logistics of supplying inert gases. This creates a much more manageable and scalable process for the production of diverse 3-trifluoroalkylquinoxalinone derivatives.
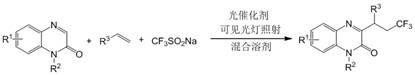
Mechanistic Insights into Visible Light Photocatalytic Trifluoroalkylation
The success of this transformation relies on the intricate interplay between the photocatalyst, the light source, and the substrates to generate reactive radical species under mild conditions. Upon irradiation with visible light, the organic photocatalyst, such as 2,4,5,6-tetra(9-carbazolyl)-isophthalonitrile (4CzIPN), enters an excited state capable of engaging in single electron transfer (SET) processes. The excited photocatalyst facilitates the oxidation of the sodium trifluoromethyl sulfinate, leading to the fragmentation of the sulfinate group and the release of the trifluoromethyl radical. This radical species is highly reactive and adds selectively to the double bond of the olefin substrate, generating a new carbon-centered radical intermediate. This intermediate subsequently undergoes cyclization with the quinoxalinone ring, followed by oxidation and deprotonation steps mediated by molecular oxygen from the air to restore aromaticity and yield the final trifluoroalkylated product. The catalytic cycle is closed by the regeneration of the ground-state photocatalyst, allowing the reaction to proceed with high atom economy.
From an impurity control perspective, the mild nature of this photocatalytic mechanism offers distinct advantages over thermal radical initiators. High-temperature conditions often promote non-selective radical generation, leading to polymerization of the olefin or over-oxidation of the sensitive quinoxalinone core. By maintaining the reaction at room temperature and utilizing the specific redox potential of the photocatalyst, the process ensures that radical generation is controlled and synchronized with the substrate availability. This selectivity minimizes the formation of side products such as bis-trifluoromethylated species or oligomers, resulting in a cleaner crude reaction profile. Consequently, the downstream purification burden is significantly reduced, facilitating the isolation of high-purity intermediates that meet the stringent quality requirements of the pharmaceutical industry without the need for extensive recrystallization or multiple chromatographic steps.
How to Synthesize 3-Trifluoroalkylquinoxalinone Efficiently
The practical implementation of this synthesis is designed to be straightforward and adaptable to standard laboratory and pilot plant equipment. The protocol involves mixing the quinoxalinone substrate, the chosen olefin, and sodium trifluoromethyl sulfinate in a solvent system comprising an organic solvent like 1,4-dioxane and water. A catalytic amount of the photocatalyst is added, and the mixture is stirred under visible light irradiation. The reaction progress is monitored via TLC, and upon completion, the product is isolated through a simple extraction and concentration sequence. The detailed standardized synthesis steps, including specific molar ratios and workup procedures optimized for maximum yield and purity, are outlined in the guide below.
- Prepare the reaction mixture by combining the quinoxalinone substrate, olefin, sodium trifluoromethyl sulfinate, and an organic photocatalyst in a mixed solvent system of organic solvent and water.
- Irradiate the homogeneous mixture with a visible light source (such as a 3W blue LED lamp) while stirring in air at room temperature for approximately 12 hours.
- Upon completion, extract the reaction liquid with ethyl acetate, concentrate under reduced pressure, and purify the crude product via silica gel column chromatography to obtain the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this visible light catalytic technology presents compelling economic and logistical benefits. The shift from hazardous gaseous reagents to stable solid salts simplifies the sourcing and storage of raw materials, reducing the regulatory burden and safety costs associated with handling compressed gases. Furthermore, the elimination of stoichiometric oxidants and transition metal catalysts removes significant cost drivers from the bill of materials. The absence of heavy metals means that expensive and time-consuming metal scavenging steps are no longer required, streamlining the production timeline and reducing the consumption of specialized purification resins. This translates directly into a more cost-effective manufacturing process with a lower overall cost of goods sold (COGS).
- Cost Reduction in Manufacturing: The process achieves cost optimization through multiple vectors, primarily by replacing expensive and hazardous reagents with commodity chemicals. The use of air as a free and abundant oxidant eliminates the need for purchasing chemical oxidants like persulfates or hypervalent iodine compounds. Additionally, the metal-free nature of the catalysis avoids the high costs associated with palladium or other precious metal catalysts, as well as the downstream costs of removing metal residues to meet regulatory limits. The energy efficiency of running reactions at room temperature rather than heating to 80°C or higher further contributes to substantial utility cost savings over long production campaigns.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly improved by the use of readily available and stable starting materials. Sodium trifluoromethyl sulfinate is a commercially available solid that does not require special transportation protocols compared to toxic gases. The robustness of the reaction conditions, which tolerate air and moisture to a certain extent due to the aqueous co-solvent system, reduces the risk of batch failures caused by minor deviations in atmospheric control. This reliability ensures consistent delivery schedules and reduces the likelihood of production delays, making it easier to maintain steady inventory levels for critical pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling this process to commercial volumes is facilitated by the mild reaction conditions and the use of flow chemistry-compatible parameters. The low viscosity of the reaction mixture and the absence of exothermic hazards associated with strong oxidants allow for safer scale-up in larger reactors. From an environmental standpoint, the reduction in chemical waste and the use of water as a co-solvent align with increasingly strict global environmental regulations. This green chemistry profile not only minimizes waste disposal costs but also enhances the corporate sustainability metrics of the manufacturing entity, which is becoming a key factor in supplier selection for major pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this visible light catalytic technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the operational feasibility and advantages of the method. Understanding these details is crucial for technical teams evaluating the integration of this process into existing manufacturing workflows.
Q: What are the primary advantages of using sodium trifluoromethyl sulfinate over trifluoroiodomethane gas in this synthesis?
A: Sodium trifluoromethyl sulfinate is a stable solid reagent that is significantly safer and easier to handle than trifluoroiodomethane gas. It eliminates the need for complex gas handling equipment and inert gas protection, simplifying the operational workflow and enhancing safety profiles for industrial applications.
Q: Does this photocatalytic method require expensive transition metal catalysts?
A: No, the method utilizes non-metallic organic photocatalysts such as 2,4,5,6-tetra(9-carbazolyl)-isophthalonitrile or organic dyes like Rose Bengal. This avoids the contamination issues associated with transition metals, thereby reducing the cost and complexity of downstream purification processes required to meet pharmaceutical purity standards.
Q: What are the typical reaction conditions regarding temperature and atmosphere for this process?
A: The reaction proceeds efficiently at room temperature under an air atmosphere. This contrasts sharply with traditional methods that often require heating or strict inert gas protection, resulting in substantial energy savings and a more environmentally friendly manufacturing process suitable for large-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoroalkylquinoxalinone Supplier
The technological advancements detailed in patent CN112142680B underscore the potential for producing high-value fluorinated heterocycles with unprecedented efficiency and sustainability. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate such innovative laboratory methodologies into robust commercial processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this visible light catalysis can be realized at an industrial level. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify the absence of impurities and residual solvents.
We invite potential partners to engage with our technical procurement team to discuss how this technology can be tailored to your specific project needs. By collaborating with us, you can leverage our capabilities to conduct a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this greener synthetic route. We encourage you to request specific COA data and route feasibility assessments to validate the performance of our 3-trifluoroalkylquinoxalinone intermediates against your internal standards. Let us help you optimize your supply chain and accelerate your drug development timelines with our superior manufacturing solutions.
