Advanced Visible Light Catalysis for Scalable 3-Trifluoroalkylquinoxalinone Production
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to introduce trifluoroalkyl groups into heterocyclic scaffolds, driven by the profound impact these moieties have on the physicochemical properties of drug candidates. Patent CN112142680B introduces a groundbreaking visible light catalytic protocol for the synthesis of 3-trifluoroalkylquinoxalinone compounds, addressing critical limitations in existing synthetic routes. This innovation leverages organic dye photocatalysts to facilitate a three-component coupling reaction between quinoxalinones, olefins, and sodium trifluoromethyl sulfinate under mild, aerobic conditions. For R&D directors and procurement managers alike, this technology represents a significant leap forward in sustainable manufacturing, offering a pathway to high-purity intermediates without the burden of toxic metal residues or complex gas handling infrastructure. The ability to perform these transformations at room temperature using air as the terminal oxidant not only reduces energy consumption but also streamlines the workflow for commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 3-trifluoroalkylquinoxalinone skeletons has been fraught with operational challenges and safety hazards that hinder efficient production. Early methodologies, such as those reported by the Studer group, relied heavily on the use of gaseous trifluoroiodomethane (CF3I) as the trifluoromethylating agent. This approach necessitates specialized gas handling equipment and strict inert atmosphere protection using argon, creating significant bottlenecks in both laboratory optimization and industrial deployment. Furthermore, the requirement for strong bases like DBU and the inherent toxicity of gaseous reagents pose serious safety risks to operators and complicate waste disposal protocols. Subsequent improvements by other research groups attempted to replace the gas with solid sodium trifluoromethyl sulfinate; however, these variations often demanded stoichiometric amounts of inorganic oxidants such as potassium persulfate or hypervalent iodine reagents, coupled with elevated reaction temperatures. These harsh conditions frequently lead to the generation of substantial inorganic salt waste, complicating downstream purification and increasing the environmental footprint of the manufacturing process.
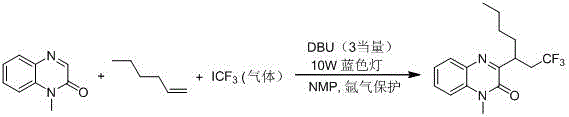
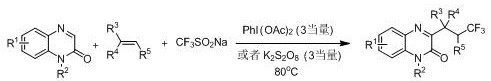
The Novel Approach
In stark contrast to these cumbersome precedents, the methodology disclosed in patent CN112142680B utilizes a visible light-driven catalytic cycle that operates under exceptionally mild conditions. By employing commercially available organic dyes such as 2,4,5,6-tetra(9-carbazolyl)-isophthalonitrile (4CzIPN) as photocatalysts, the reaction proceeds efficiently at room temperature in the presence of air, eliminating the need for inert gas protection. The use of sodium trifluoromethyl sulfinate as a stable, solid source of trifluoromethyl radicals avoids the logistical nightmares associated with gaseous reagents. Moreover, the inclusion of water as a co-solvent in the reaction medium enhances the green chemistry profile of the process, reducing the reliance on pure organic solvents. This novel approach not only simplifies the experimental setup but also ensures a cleaner reaction profile, making it an ideal candidate for reliable pharmaceutical intermediate supplier networks aiming to minimize production costs and environmental impact.
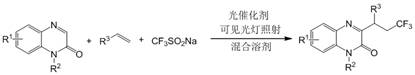
Mechanistic Insights into Visible Light Photocatalytic Radical Addition
The core of this transformative synthesis lies in the photoredox catalytic cycle initiated by the excitation of the organic dye under visible light irradiation. Upon absorption of photons from the LED source, the photocatalyst transitions to an excited state capable of engaging in single electron transfer (SET) processes. In this specific transformation, the excited photocatalyst facilitates the oxidation of sodium trifluoromethyl sulfinate, generating the highly reactive trifluoromethyl radical species along with sulfur dioxide and a sulfinate anion. This radical addition mechanism is pivotal for the functionalization of the olefin component, where the trifluoromethyl radical adds across the double bond to form a new carbon-centered radical intermediate. This intermediate subsequently undergoes radical addition to the C3 position of the quinoxalinone ring, followed by oxidation and deprotonation steps to restore aromaticity and yield the final 3-trifluoroalkylated product. The elegance of this mechanism is its reliance on molecular oxygen from the air to close the catalytic cycle, regenerating the ground state photocatalyst without the need for external chemical oxidants.
From an impurity control perspective, the mildness of this radical pathway offers distinct advantages over thermal or metal-catalyzed alternatives. The absence of transition metals eliminates the risk of heavy metal contamination, a critical parameter for API intermediates destined for clinical use. Furthermore, the room temperature conditions suppress side reactions such as polymerization of the olefin or decomposition of sensitive functional groups that might occur under heating. The use of water as a co-solvent also aids in quenching certain polar byproducts, facilitating a cleaner workup procedure. For quality assurance teams, this translates to a more consistent impurity profile and reduced burden on analytical validation, ensuring that the high-purity OLED material or pharmaceutical precursors produced meet stringent regulatory specifications with minimal downstream processing.
How to Synthesize 3-Trifluoroalkylquinoxalinone Efficiently
Implementing this visible light catalytic protocol requires careful attention to reaction parameters to maximize yield and reproducibility. The standard procedure involves charging a reaction vessel with the quinoxalinone substrate, the desired olefin, sodium trifluoromethyl sulfinate, and the organic photocatalyst in a mixed solvent system typically comprising 1,4-dioxane and water. The detailed standardized synthesis steps below outline the optimal molar ratios, light source specifications, and workup procedures derived from the patent examples to ensure successful execution of this transformation.
- Combine the photocatalyst (e.g., 4CzIPN), quinoxalinone substrate, olefin, and sodium trifluoromethyl sulfinate in a mixed solvent system of organic solvent and water.
- Irradiate the reaction mixture with a visible light source (e.g., 3W Blue LED) while stirring in air at room temperature for approximately 12 hours.
- Extract the reaction mixture with ethyl acetate, concentrate under reduced pressure, and purify the crude product via silica gel column chromatography to obtain the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this visible light catalytic technology offers compelling economic and logistical benefits that directly impact the bottom line. The shift from gaseous reagents to stable solid salts drastically simplifies raw material sourcing and storage, removing the need for specialized gas cylinders and reducing inventory holding costs. Additionally, the elimination of stoichiometric oxidants and the use of catalytic amounts of inexpensive organic dyes significantly lower the material cost per kilogram of product. The ability to run reactions at room temperature without inert gas protection reduces energy consumption associated with heating and nitrogen purging, contributing to substantial cost savings in utility expenses. These factors combined create a more resilient supply chain capable of responding quickly to market demands without being bottlenecked by complex safety protocols or scarce reagents.
- Cost Reduction in Manufacturing: The economic viability of this process is underscored by the replacement of expensive transition metal catalysts and hazardous gaseous reagents with affordable organic dyes and solid sulfinate salts. By operating at room temperature, the process eliminates the energy costs associated with heating reactors to high temperatures, while the use of air as the oxidant removes the expense of purchasing and handling chemical oxidants. This streamlined reagent profile leads to a drastic simplification of the bill of materials, driving down the overall cost of goods sold for high-value trifluoroalkylated intermediates.
- Enhanced Supply Chain Reliability: Supply chain continuity is significantly bolstered by the use of commercially available, shelf-stable reagents that do not require cold chain logistics or special pressure vessels for transport. The robustness of the reaction conditions means that production is less susceptible to delays caused by equipment failure related to high-pressure or high-temperature systems. Furthermore, the simplified operational requirements allow for greater flexibility in manufacturing site selection, enabling decentralized production strategies that mitigate risks associated with single-source dependencies.
- Scalability and Environmental Compliance: Scaling this process to industrial levels is facilitated by the benign nature of the reaction components and the absence of toxic metal waste streams. The use of water as a co-solvent reduces the volume of organic waste generated, aligning with increasingly strict environmental regulations and reducing waste disposal fees. The mild conditions also allow for the use of standard glass-lined or stainless steel reactors without the need for exotic materials of construction, lowering capital expenditure for capacity expansion and ensuring long-term sustainability of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this visible light catalytic synthesis method. These answers are derived directly from the experimental data and beneficial effects described in patent CN112142680B, providing clarity on substrate scope, reaction conditions, and scalability potential for stakeholders evaluating this technology for integration into their production pipelines.
Q: What are the advantages of this visible light method over traditional trifluoromethylation techniques?
A: Unlike traditional methods that require hazardous gaseous reagents like trifluoroiodomethane or stoichiometric amounts of heavy metal oxidants and high temperatures, this patented process utilizes solid sodium trifluoromethyl sulfinate, ambient air as the oxidant, and operates at room temperature, significantly improving safety and environmental compliance.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is highly scalable due to its mild reaction conditions, the use of inexpensive organic dye photocatalysts instead of precious metals, and the elimination of inert gas protection requirements, which simplifies reactor setup and reduces operational costs for industrial production.
Q: What types of substrates are compatible with this synthesis method?
A: The method demonstrates broad substrate scope, accommodating various substituted quinoxalinones and diverse olefins including styrenes, aliphatic alkenes, and functionalized alkenes, allowing for the generation of a wide library of biologically active 3-trifluoroalkylquinoxalinone derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoroalkylquinoxalinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of visible light catalysis in modern medicinal chemistry and are committed to delivering these advanced intermediates to our global partners. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 3-trifluoroalkylquinoxalinone meets the highest standards required for pharmaceutical applications, providing you with a reliable source for your critical drug development programs.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific project needs. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this green chemistry approach can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you accelerate your timeline to market with superior quality intermediates.
