Advanced Palladium-Catalyzed Synthesis of 1,5-Dihydro-2H-Pyrrole-2-Ketone Scaffolds for Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis of 1,5-Dihydro-2H-Pyrrole-2-Ketone Scaffolds for Commercial Scale-Up
The development of efficient synthetic routes for bioactive heterocyclic scaffolds remains a critical priority for the global pharmaceutical industry. As detailed in the recent patent CN112694430B, a novel preparation method for 1,5-dihydro-2H-pyrrole-2-ketone compounds has been established, offering a robust solution for constructing this privileged structural backbone. This specific heterocyclic motif is not merely an academic curiosity but serves as the core framework for several high-value therapeutic agents, including the antibiotic althiomycin, the hypoglycemic agent glimepiride, and the anticancer candidate isomalyngamide A. The ability to access these complex structures through a streamlined, one-step process represents a significant technological leap forward for manufacturers seeking reliable pharmaceutical intermediate supplier partnerships. By leveraging palladium-catalyzed bis-carbonylation, this technology addresses long-standing challenges in yield optimization and operational safety, positioning it as a cornerstone for next-generation drug synthesis.
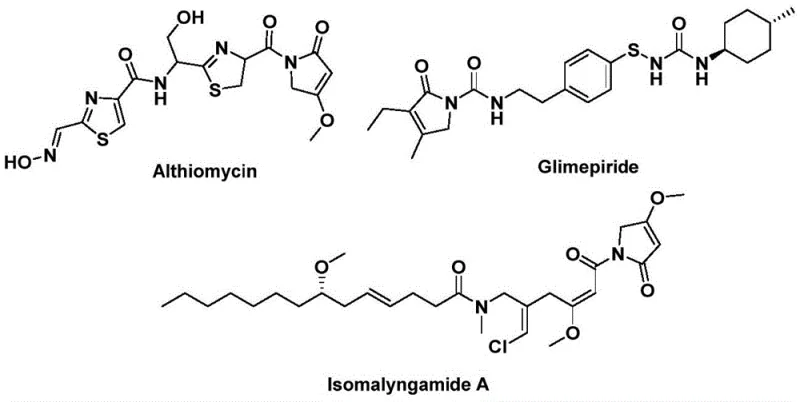
Historically, the construction of carbonyl-containing heterocycles often relied on traditional carbonylation techniques that utilized gaseous carbon monoxide. While effective in theory, these conventional methods present substantial logistical and safety hurdles for industrial application, particularly regarding the handling of toxic, high-pressure gases and the requirement for specialized autoclave reactors. Furthermore, older synthetic pathways frequently suffered from limited substrate scope, harsh reaction conditions that degraded sensitive functional groups, and multi-step sequences that eroded overall process efficiency. These limitations created bottlenecks in cost reduction in API manufacturing, as the need for extensive safety protocols and complex purification steps drove up production expenses. In contrast, the novel approach disclosed in the patent utilizes a solid carbon monoxide surrogate, specifically 1,3,5-tricarboxylic acid phenol ester (TFBen), which releases CO in situ under mild thermal conditions. This strategic shift from gas to solid reagents fundamentally transforms the risk profile of the synthesis, enabling safer operations without sacrificing chemical efficiency.
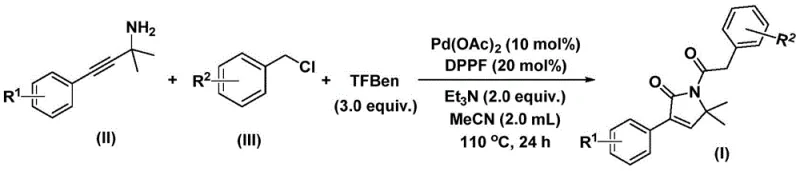
The mechanistic elegance of this palladium-catalyzed bis-carbonylation lies in its precise orchestration of organometallic steps to form the five-membered lactam ring. The catalytic cycle initiates with the oxidative addition of the palladium catalyst into the benzyl chloride bond, generating a reactive benzyl-palladium intermediate. Subsequently, carbon monoxide liberated from the phenol ester surrogate inserts into this intermediate to form an acyl-palladium species. This acyl complex then undergoes nucleophilic attack by the propargyl amine, facilitating the formation of a five-membered ring palladium intermediate. A second insertion of carbon monoxide expands the coordination sphere, eventually leading to reductive elimination that releases the final 1,5-dihydro-2H-pyrrole-2-ketone product and regenerates the active catalyst. This intricate dance of ligands and substrates ensures high regioselectivity and minimizes the formation of unwanted byproducts, thereby simplifying the impurity profile. For R&D teams, understanding this mechanism is crucial for troubleshooting and optimizing reaction parameters to ensure consistent quality across different batches.
How to Synthesize 1,5-Dihydro-2H-Pyrrole-2-Ketone Efficiently
Implementing this synthesis requires careful attention to reagent stoichiometry and thermal control to maximize the benefits of the bis-carbonylation pathway. The process is designed to be operationally simple, utilizing common laboratory solvents like acetonitrile and standard heating equipment, which facilitates easy translation from bench scale to pilot plant operations. The protocol involves mixing the palladium source, phosphine ligand, base, solid CO surrogate, and the two primary organic substrates in a single vessel, followed by heating to promote the cascade reaction. Detailed standardized synthesis steps see the guide below, which outlines the precise molar ratios and temperature profiles required to achieve the high yields reported in the patent data.
- Combine palladium acetate catalyst, DPPP ligand, triethylamine base, solid CO surrogate (TFBen), propargyl amine, and benzyl chloride in acetonitrile solvent.
- Heat the reaction mixture to 110°C and stir for 24 hours under inert atmosphere to facilitate the bis-carbonylation cyclization.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the target 1,5-dihydro-2H-pyrrole-2-ketone product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic methodology offers tangible benefits that extend beyond mere chemical yield. The transition to a solid carbon monoxide surrogate eliminates the dependency on hazardous gas infrastructure, which drastically simplifies facility requirements and reduces the regulatory burden associated with storing and transporting toxic gases. This shift not only enhances workplace safety but also streamlines the supply chain by allowing for the use of standard shipping containers for reagents rather than specialized pressure vessels. Consequently, the lead time for raw material acquisition is potentially reduced, and the overall cost of goods sold is optimized through the removal of expensive safety mitigation systems. The robustness of the reaction conditions further ensures that production schedules remain uninterrupted by technical failures or safety incidents.
- Cost Reduction in Manufacturing: The utilization of cheap and readily available starting materials, such as substituted benzyl chlorides and propargyl amines, combined with the elimination of high-pressure gas equipment, leads to substantial cost savings. The one-pot nature of the reaction minimizes solvent usage and labor hours associated with intermediate isolation, directly impacting the bottom line by reducing the operational expenditure required for high-purity pharmaceutical intermediate production.
- Enhanced Supply Chain Reliability: Since the key reagents including palladium acetate, DPPP ligand, and the solid CO surrogate are commercially available commodities, the risk of supply disruption is significantly mitigated. The method's tolerance for a wide range of functional groups means that diverse derivatives can be produced using a unified platform, allowing manufacturers to respond rapidly to changing market demands for specific analogues without retooling entire production lines.
- Scalability and Environmental Compliance: The post-treatment process is remarkably straightforward, involving simple filtration and standard column chromatography, which generates less hazardous waste compared to traditional multi-step syntheses. This simplicity supports easier scale-up from kilograms to metric tons while maintaining strict environmental compliance, as the reduced solvent load and absence of toxic gas emissions align with modern green chemistry principles and regulatory standards.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented technology. These insights are derived directly from the experimental data and mechanistic studies presented in the patent documentation, providing clarity on reaction conditions and substrate limitations. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the key advantages of using a solid CO surrogate over gaseous CO?
A: Using 1,3,5-tricarboxylic acid phenol ester (TFBen) as a solid carbon monoxide surrogate eliminates the need for high-pressure gas cylinders and specialized containment equipment, significantly enhancing operational safety and simplifying logistics for large-scale manufacturing.
Q: What is the substrate compatibility of this palladium-catalyzed method?
A: The method demonstrates excellent functional group tolerance, successfully accommodating electron-donating groups like methoxy and methyl, as well as electron-withdrawing groups such as halogens (F, Cl, Br) and trifluoromethyl substituents on both the propargyl amine and benzyl chloride starting materials.
Q: How does this process impact purification and downstream processing?
A: The reaction proceeds with high conversion rates in acetonitrile, allowing for a straightforward workup procedure involving simple filtration followed by standard silica gel column chromatography, which minimizes solvent waste and reduces the complexity of isolating high-purity intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,5-Dihydro-2H-Pyrrole-2-Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the palladium-catalyzed bis-carbonylation described in CN112694430B. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to market supply is seamless. Our state-of-the-art facilities are equipped to handle complex organometallic reactions with stringent purity specifications, supported by rigorous QC labs that guarantee every batch meets the highest international standards. We are committed to delivering high-purity 1,5-dihydro-2H-pyrrole-2-ketone derivatives that empower your drug development programs with reliable, high-quality intermediates.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of adopting this route for your supply chain. We encourage potential partners to contact us directly to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your next generation of bioactive molecules is built on a foundation of chemical excellence and supply chain security.
