Advanced Palladium-Catalyzed Synthesis of 1,5-Dihydro-2H-Pyrrole-2-Ketone Compounds for Commercial Scale-Up
Introduction to Next-Generation Pyrrol-2-One Synthesis
The structural motif of 1,5-dihydro-2H-pyrrol-2-one serves as a critical backbone in the architecture of numerous bioactive natural products and pharmaceutical agents, ranging from antibacterial compounds like Althiomycin to hypoglycemic agents such as Glimepiride. As depicted in the structural diversity of these bioactive molecules, the ability to efficiently construct this heterocyclic core is of paramount importance to modern medicinal chemistry and process development teams. Patent CN112694430B introduces a groundbreaking preparation method that leverages palladium-catalyzed bis-carbonylation to synthesize these valuable scaffolds in a single, efficient step. This innovation addresses long-standing challenges in heterocycle synthesis by utilizing readily available starting materials—specifically propargylamines and benzyl chlorides—thereby offering a streamlined route that bypasses the complexities of traditional multi-step sequences.
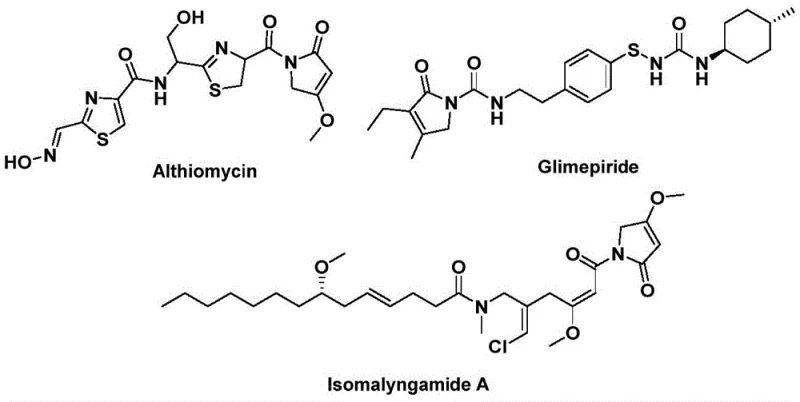
Furthermore, the methodology described in this patent represents a significant leap forward in atom economy and operational simplicity, making it an attractive candidate for industrial adoption. By replacing hazardous gaseous carbon monoxide with a solid surrogate, the process not only enhances laboratory safety but also reduces the engineering barriers associated with high-pressure carbonylation reactions. For R&D directors and process chemists, this translates to a more robust and scalable pathway for generating complex intermediates required for drug discovery pipelines. The broad substrate compatibility ensures that diverse analogues can be rapidly accessed, accelerating the structure-activity relationship (SAR) studies essential for identifying lead candidates in therapeutic areas such as oncology and metabolic disorders.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,5-dihydro-2H-pyrrol-2-one ring system has often relied on cumbersome multi-step synthetic routes that suffer from low overall yields and poor atom economy. Traditional carbonylation strategies frequently necessitate the use of toxic carbon monoxide gas under high pressure, which imposes severe safety constraints and requires specialized, expensive reactor infrastructure that many contract manufacturing organizations lack. Additionally, conventional methods often exhibit limited functional group tolerance, forcing chemists to employ protecting group strategies that add unnecessary steps, increase waste generation, and drive up the cost of goods sold (COGS). These inefficiencies create bottlenecks in the supply chain, particularly when rapid iteration of chemical structures is required during the lead optimization phase of drug development.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a palladium-catalyzed cascade reaction that integrates two carbonylation events and a cyclization step into a single pot. By employing 1,3,5-tricarboxylic acid phenol ester (TFBen) as a safe and convenient solid carbon monoxide source, the method eliminates the need for handling dangerous CO gas, thereby drastically simplifying the operational protocol. The reaction proceeds under relatively mild thermal conditions (100-120°C) in common organic solvents like acetonitrile, utilizing inexpensive and commercially available catalysts such as palladium acetate and DPPF. This one-pot transformation not only reduces the number of isolation and purification steps but also significantly improves the overall process mass intensity (PMI), aligning perfectly with the principles of green chemistry and sustainable manufacturing practices demanded by modern regulatory bodies.
Mechanistic Insights into Pd-Catalyzed Bis-Carbonylation
The catalytic cycle begins with the oxidative addition of the benzyl chloride substrate to the active palladium(0) species generated in situ from palladium acetate and the bidentate phosphine ligand DPPF. This crucial step forms a benzyl-palladium intermediate, which subsequently undergoes migratory insertion of carbon monoxide released from the thermal decomposition of the TFBen additive. The resulting acyl-palladium species then reacts with the propargylamine component, initiating the formation of the five-membered ring framework through a nucleophilic attack or insertion mechanism. This sequence is followed by a second carbon monoxide insertion event, expanding the coordination sphere to form a six-membered palladacycle intermediate that sets the stage for the final ring contraction and product release.
Finally, reductive elimination from the six-membered palladium intermediate releases the desired 1,5-dihydro-2H-pyrrol-2-one product and regenerates the active palladium(0) catalyst to close the cycle. From an impurity control perspective, the use of a well-defined ligand system like DPPF helps stabilize the palladium center, minimizing the formation of side products such as homocoupling derivatives or incomplete carbonylation byproducts. The high selectivity observed across various substrates, including those with electron-withdrawing trifluoromethyl groups or electron-donating methoxy substituents, suggests a robust catalytic manifold that is less sensitive to steric and electronic variations. This mechanistic understanding allows process chemists to fine-tune reaction parameters, such as temperature and stoichiometry, to maximize yield and purity, ensuring that the final API intermediate meets stringent quality specifications required for clinical trials.
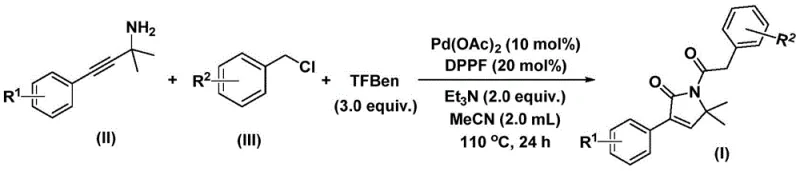
How to Synthesize 1,5-Dihydro-2H-Pyrrole-2-Ketone Efficiently
To implement this synthesis effectively, operators must adhere to precise stoichiometric ratios and thermal profiles as outlined in the experimental data. The process typically involves charging a reaction vessel with the palladium catalyst, ligand, base, solid CO source, and substrates in acetonitrile, followed by heating to 110°C for approximately 24 hours. Detailed standard operating procedures regarding the specific addition order, inert atmosphere maintenance, and workup protocols are critical for reproducibility and safety. For a comprehensive, step-by-step technical guide on executing this transformation, please refer to the standardized synthesis instructions provided below.
- Combine palladium acetate, DPPF ligand, triethylamine, solid CO source (TFBen), propargylamine, and benzyl chloride in acetonitrile solvent.
- Heat the reaction mixture to 110°C and stir for 24 hours under inert atmosphere to facilitate the bis-carbonylation cyclization.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the target 1,5-dihydro-2H-pyrrol-2-one product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement standpoint, this patented methodology offers substantial strategic benefits by leveraging commodity chemicals that are abundant in the global supply chain. The primary starting materials, benzyl chlorides and propargylamines, are widely produced at scale for various industrial applications, ensuring a stable and competitive pricing structure that shields manufacturers from volatile raw material costs. Furthermore, the elimination of high-pressure gas handling equipment reduces the capital expenditure (CAPEX) required for facility upgrades, allowing existing multipurpose reactors to be utilized for production without significant modification. This flexibility enhances supply chain resilience, enabling rapid response to market demands and reducing the lead time associated with sourcing specialized reagents or contracting niche CDMOs.
- Cost Reduction in Manufacturing: The transition from hazardous gaseous carbon monoxide to a solid surrogate significantly lowers operational costs by removing the need for specialized gas containment systems and safety monitoring infrastructure. Additionally, the one-pot nature of the reaction minimizes solvent usage and waste disposal fees, while the high conversion rates reduce the loss of valuable intermediates during purification. By streamlining the synthesis into fewer steps, labor costs are also optimized, contributing to a lower overall cost of goods sold (COGS) for the final pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: Utilizing commercially available catalysts like palladium acetate and DPPF ensures that production is not dependent on proprietary or hard-to-source reagents that could cause supply disruptions. The robustness of the reaction conditions allows for consistent batch-to-batch quality, reducing the risk of production delays caused by failed runs or out-of-specification results. This reliability is crucial for maintaining continuous supply to downstream API manufacturers, particularly in the context of just-in-time inventory models prevalent in the pharmaceutical industry.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on standard heating and stirring operations rather than complex flow chemistry or high-pressure setups. The use of acetonitrile, a common and recyclable solvent, simplifies waste management and aligns with increasingly strict environmental regulations regarding volatile organic compound (VOC) emissions. The simplified workup procedure, involving filtration and standard column chromatography, facilitates easier technology transfer from pilot plant to commercial scale, ensuring a smoother path to market for new drug candidates incorporating this scaffold.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed synthesis route. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on reaction scope, safety, and scalability. Understanding these nuances is essential for project managers evaluating the feasibility of adopting this technology for their specific pipeline needs.
Q: What are the key advantages of this Pd-catalyzed method over traditional carbonylation?
A: This method utilizes a solid carbon monoxide substitute (TFBen) instead of toxic CO gas, significantly improving operational safety and simplifying equipment requirements for scale-up.
Q: What is the substrate scope for the benzyl chloride and propargylamine components?
A: The process demonstrates excellent functional group tolerance, accommodating electron-donating groups like methoxy and methyl, as well as electron-withdrawing groups such as halogens and trifluoromethyl.
Q: Is this synthesis suitable for large-scale pharmaceutical production?
A: Yes, the one-pot nature, use of commercially available catalysts, and simple purification via column chromatography make it highly amenable to commercial scale-up and cost-effective manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,5-Dihydro-2H-Pyrrole-2-Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient intermediate synthesis plays in accelerating drug development timelines. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. We are committed to delivering high-purity 1,5-dihydro-2H-pyrrole-2-ketone compounds that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify identity and assay.
We invite you to engage with our technical procurement team to discuss how this advanced palladium-catalyzed technology can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this greener, more efficient route. Contact us today to obtain specific COA data and route feasibility assessments tailored to your unique molecular requirements, and let us partner with you to optimize your pharmaceutical manufacturing processes.
