Scalable Ruthenium-Catalyzed Synthesis of 2-Aryl Ortho-Substituted Triethyl Silicon Pyridine Compounds
Introduction to Advanced Silylation Technology
The landscape of organic synthesis for heterocyclic compounds is constantly evolving, driven by the need for more sustainable and cost-effective manufacturing processes. Patent CN109651421B introduces a significant breakthrough in the preparation of 2-aryl ortho-substituted triethyl silicon pyridine compounds, a class of molecules that serves as critical building blocks in the development of novel materials and bioactive agents. These silicon-containing pyridine derivatives are increasingly recognized for their potential applications in antiviral and antitumor therapies, making their efficient production a priority for the global pharmaceutical industry. The disclosed technology addresses the longstanding challenges associated with traditional silylation methods by leveraging a robust ruthenium-catalyzed system that operates under relatively mild heating conditions. By shifting away from ultra-expensive noble metals to more abundant transition metals, this invention paves the way for a more economically viable supply chain for high-purity pharmaceutical intermediates.
Furthermore, the methodology described in the patent emphasizes a streamlined one-pot synthesis approach that eliminates the need for isolating unstable intermediates, thereby reducing both processing time and waste generation. This is particularly relevant for procurement managers and supply chain directors who are tasked with minimizing the environmental footprint and operational costs of chemical manufacturing. The ability to synthesize these complex ortho-substituted structures in a single reaction vessel represents a paradigm shift from multi-step electrophilic substitutions that often suffer from poor regioselectivity. As we delve deeper into the technical specifics, it becomes clear that this process offers a reliable pathway for the commercial scale-up of complex pharmaceutical intermediates, ensuring consistent quality and availability for downstream drug development projects.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of silicon-functionalized pyridine derivatives has relied heavily on electrophilic substitution reactions involving reagents such as trimethylsilyl trifluoromethanesulfonate (TMSOTf) or trimethylchlorosilane (TMSCl). These traditional routes are fraught with significant drawbacks, including multi-step synthetic sequences that drastically increase production lead times and operational complexity. Moreover, electrophilic substitutions often exhibit low chemical selectivity, leading to mixtures of regioisomers that require extensive and costly purification efforts to separate the desired ortho-substituted product from para- or meta-substituted byproducts. The generation of large quantities of inorganic salt waste during these processes also poses substantial environmental compliance challenges, complicating waste disposal and increasing the overall cost of goods sold. Additionally, while recent advancements have introduced noble metal catalysts based on rhodium and iridium to activate carbon-hydrogen bonds, the exorbitant price and scarcity of these metals render them impractical for large-scale industrial applications, limiting their utility to small-batch laboratory research.
The Novel Approach
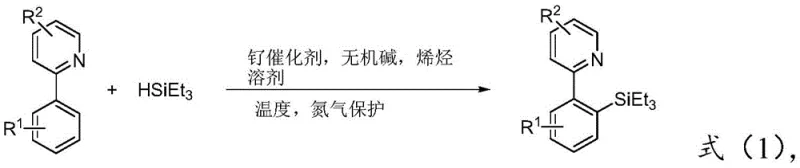
In stark contrast to these legacy methods, the innovative process detailed in patent CN109651421B utilizes a cost-effective ruthenium complex to catalyze the direct C-H silylation of 2-arylpyridines with triethylsilane. This novel approach creatively incorporates an inorganic base and an unsaturated olefin, specifically norbornene, to facilitate the catalytic cycle under heating conditions, typically around 120°C. The result is a highly efficient one-pot synthesis that delivers the target 2-aryl ortho-substituted triethyl silicon pyridine compounds with impressive yields, reaching up to 85% in optimized examples. By eliminating the need for intermediate isolation and purification, this method significantly simplifies the workflow, reducing labor inputs and solvent consumption. The use of a relatively inexpensive ruthenium catalyst, such as tris(triphenylphosphine)carbonyl ruthenium hydrochloride, further enhances the economic feasibility of the process, making it an attractive option for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Ruthenium-Catalyzed C-H Silylation
The core of this technological advancement lies in the unique mechanistic pathway enabled by the ruthenium catalyst in the presence of norbornene and an inorganic base. The reaction initiates with the coordination of the ruthenium center to the nitrogen atom of the pyridine ring, which acts as a directing group to position the metal in proximity to the ortho-carbon-hydrogen bond. This coordination facilitates the activation of the inert C-H bond through a concerted metalation-deprotonation mechanism, assisted by the acetate base. The unsaturated olefin, norbornene, plays a crucial role in this catalytic cycle, likely acting as a hydrogen acceptor or facilitating the reductive elimination step that forms the new carbon-silicon bond. This synergistic interaction between the ruthenium catalyst, the base, and the olefin additive allows for the selective functionalization of the ortho-position with high precision, avoiding the random substitution patterns typical of electrophilic aromatic substitution. The robustness of this catalytic system ensures that the reaction proceeds smoothly even in the presence of various substituents on the aryl ring, maintaining high turnover numbers and minimizing catalyst deactivation.
From an impurity control perspective, the high regioselectivity of this ruthenium-catalyzed process is a major advantage for R&D directors focused on purity specifications. The mechanism inherently favors the formation of the ortho-silylated product due to the chelation assistance provided by the pyridine nitrogen, effectively suppressing the formation of unwanted meta- or para-isomers. This intrinsic selectivity reduces the burden on downstream purification processes, such as column chromatography or crystallization, which are often required to remove structural impurities. Furthermore, the compatibility of the reaction conditions with sensitive functional groups, such as methoxy and trifluoromethyl moieties, demonstrates the versatility of the catalyst system. For instance, the successful silylation of substrates containing electron-donating methoxy groups confirms that the catalytic cycle is not inhibited by increased electron density on the aromatic ring, broadening the scope of accessible chemical space for drug discovery.
How to Synthesize 2-Aryl Ortho-Substituted Triethyl Silicon Pyridine Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to maximize yield and reproducibility. The patent outlines a straightforward procedure where the key reagents—2-arylpyridine, triethylsilane, potassium acetate, norbornene, and the ruthenium catalyst—are combined in a solvent such as toluene. The mixture is then subjected to heating under an inert nitrogen atmosphere to prevent oxidation of the catalyst or reagents. Detailed standard operating procedures regarding stoichiometry, temperature ramping, and workup protocols are essential for transferring this laboratory-scale success to pilot and commercial production environments. The following guide summarizes the critical steps derived from the patent data to ensure efficient execution of this transformation.
- Combine 2-arylpyridine, triethylsilane, inorganic base (potassium acetate), unsaturated olefin (norbornene), and ruthenium catalyst in a solvent like toluene.
- Heat the reaction mixture to 120°C under nitrogen protection with electromagnetic stirring for approximately 16 hours.
- Remove the solvent by rotary evaporation and purify the crude product via column chromatography to isolate the target silylated compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ruthenium-catalyzed synthesis offers tangible benefits that extend beyond mere technical feasibility. The primary advantage lies in the substantial cost savings achieved by replacing expensive rhodium or iridium catalysts with a more affordable ruthenium alternative. This switch directly impacts the raw material costs, allowing for a more competitive pricing structure for the final intermediate without compromising on quality or performance. Additionally, the one-pot nature of the reaction eliminates multiple unit operations associated with intermediate isolation, which translates to reduced energy consumption, lower solvent usage, and decreased labor requirements. These operational efficiencies contribute to a leaner manufacturing process that is better suited for high-volume production, ensuring a stable and continuous supply of critical building blocks for the pharmaceutical industry.
- Cost Reduction in Manufacturing: The utilization of a ruthenium-based catalyst system significantly lowers the capital expenditure associated with precious metal procurement. Unlike rhodium or iridium complexes, which are subject to volatile market prices and supply constraints, ruthenium catalysts offer a more stable and predictable cost profile. Furthermore, the high atom economy of the direct C-H silylation reaction minimizes waste generation, reducing the costs associated with waste treatment and disposal. The elimination of intermediate purification steps also means less solvent is consumed and recovered, further driving down the variable costs per kilogram of product. This comprehensive approach to cost optimization ensures that the manufacturing process remains economically viable even at large commercial scales.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials, such as 2-arylpyridines and triethylsilane, mitigates the risk of supply chain disruptions. These feedstocks are commodity chemicals with established global supply networks, ensuring consistent availability and reducing the lead time for raw material acquisition. The robustness of the reaction conditions, which tolerate a range of functional groups, also allows for flexibility in sourcing different substituted arylpyridines without needing to re-optimize the entire process. This flexibility is crucial for maintaining production schedules and meeting the dynamic demands of downstream customers in the pharmaceutical and agrochemical sectors. By securing a reliable source of high-quality intermediates, companies can better manage their inventory levels and reduce the safety stock required to buffer against supply volatility.
- Scalability and Environmental Compliance: The simplicity of the one-pot protocol facilitates easy scale-up from laboratory benchtop to industrial reactors. The absence of complex multi-step sequences reduces the potential for equipment bottlenecks and simplifies process control strategies. From an environmental perspective, the reduced generation of inorganic salts and organic waste aligns with increasingly stringent regulatory requirements for green chemistry. The use of toluene as a solvent, while requiring proper handling, is well-understood in industrial settings, and the overall reduction in waste volume simplifies compliance reporting. This alignment with sustainability goals not only reduces regulatory risk but also enhances the corporate social responsibility profile of the manufacturing operation, appealing to environmentally conscious stakeholders and partners.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical and commercial aspects of this synthesis method. These answers are derived directly from the experimental data and technical disclosures within patent CN109651421B, providing clarity on the process capabilities and limitations. Understanding these details is essential for making informed decisions about integrating this technology into existing production workflows or new product development pipelines.
Q: Why is the ruthenium catalyst preferred over rhodium or iridium for this synthesis?
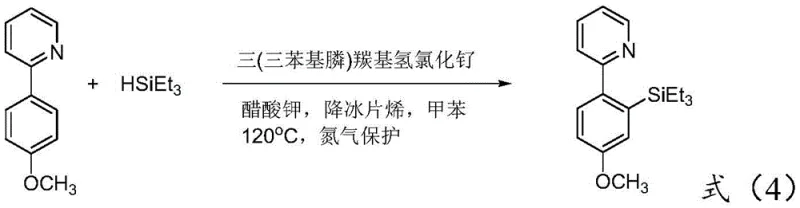
A: While rhodium and iridium complexes are effective for C-H activation, they are significantly more expensive and scarce. The patented method utilizes a ruthenium complex, specifically tris(triphenylphosphine)carbonyl ruthenium hydrochloride, which offers a much lower cost profile suitable for industrial scale-up while maintaining high catalytic efficiency.
Q: What yields can be expected from this synthetic route?
A: According to the experimental data in patent CN109651421B, the optimized conditions using potassium acetate and norbornene in toluene at 120°C can achieve isolated yields up to 85% for the parent 2-phenylpyridine substrate, with comparable efficiency for substituted derivatives.
Q: Does this method tolerate diverse functional groups on the aryl ring?
A: Yes, the protocol demonstrates excellent functional group compatibility. It successfully accommodates electron-donating groups such as methyl and methoxy, as well as electron-withdrawing groups like trifluoromethyl, allowing for the synthesis of a wide variety of downstream pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Aryl Ortho-Substituted Triethyl Silicon Pyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of modern drug development programs. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent literature can be reliably translated into industrial reality. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which employ advanced analytical techniques to verify identity and assess impurity profiles. Our capability to handle complex organometallic transformations, such as the ruthenium-catalyzed silylation described here, positions us as a strategic partner for companies seeking to secure their supply chain for next-generation therapeutic agents.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this ruthenium-catalyzed process for your specific target molecules. We encourage you to contact us to obtain specific COA data for our catalog items or to initiate a dialogue regarding route feasibility assessments for your proprietary compounds. Together, we can accelerate the development of vital pharmaceutical solutions while optimizing manufacturing efficiency and cost.
