Advanced Ruthenium-Catalyzed Silylation for Scalable Pharmaceutical Intermediate Manufacturing
Advanced Ruthenium-Catalyzed Silylation for Scalable Pharmaceutical Intermediate Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking more efficient and cost-effective pathways to synthesize complex heterocyclic scaffolds, particularly those containing silicon-carbon bonds which are pivotal in modern drug design and material science. Patent CN109651421B introduces a groundbreaking synthesis method for 2-aryl ortho-substituted triethyl silicon pyridine compounds, addressing critical bottlenecks in traditional C-H activation strategies. This innovation leverages a relatively inexpensive ruthenium complex catalyst to facilitate the direct ortho-silylation of 2-arylpyridine derivatives with triethylsilane in a streamlined one-pot process. By eliminating the reliance on precious metals like rhodium or iridium and avoiding multi-step purification of intermediates, this technology offers a compelling value proposition for manufacturers aiming to optimize their supply chains for high-purity pharmaceutical intermediates.
From a strategic sourcing perspective, the adoption of this ruthenium-catalyzed protocol represents a significant shift towards sustainable and economically viable manufacturing practices. The ability to produce these silicon-containing pyridine derivatives, which possess notable biological activities including antiviral and antitumor properties, using abundant base metals is a major technical breakthrough. For R&D directors and procurement managers alike, understanding the mechanistic nuances and operational simplicity of this patent is essential for evaluating its potential integration into existing production lines. The following analysis details how this method outperforms conventional electrophilic substitution and noble-metal catalysis, providing a robust foundation for the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of silicon-containing pyridine derivatives has relied heavily on electrophilic substitution reactions involving reagents such as trimethylsilyl trifluoromethanesulfonate (TMSOTf) or trimethylchlorosilane (TMSCl). These traditional pathways are fraught with significant disadvantages, including multi-step synthesis requirements, poor chemical selectivity, and limited functional group compatibility. Furthermore, these methods often generate substantial quantities of inorganic salt byproducts, complicating downstream purification and waste management processes. While recent advancements have introduced noble metal catalysts based on rhodium and iridium to activate carbon-hydrogen bonds, the prohibitive cost and scarcity of these metals severely hinder their widespread industrial adoption. The economic burden of using such expensive catalysts, combined with the operational complexity of multi-step sequences, creates a barrier to entry for cost-sensitive manufacturing sectors.
The Novel Approach
In stark contrast, the method disclosed in CN109651421B utilizes a cost-effective ruthenium catalyst system to achieve direct ortho-silylation with remarkable efficiency. By employing triethylsilane as the silylating agent in the presence of an inorganic base and an unsaturated olefin, the reaction proceeds smoothly under heating conditions to yield the target 2-aryl ortho-substituted triethyl silicon pyridine compounds. A prime example of this efficiency is demonstrated in the synthesis of specific derivatives where yields reach up to 85%, showcasing the robustness of the catalytic system.

This novel approach not only simplifies the reaction setup to a single vessel but also ensures that intermediates formed during the conversion process do not require isolation or purification. The use of readily available ruthenium complexes, such as tris(triphenylphosphine)carbonyl ruthenium hydrochloride, significantly lowers the capital investment required for catalyst procurement. This transition from expensive noble metals to base metals, coupled with a one-pot operational mode, directly translates to reduced labor inputs and lower overall production costs, making it an ideal solution for cost reduction in fine chemical manufacturing.
Mechanistic Insights into Ruthenium-Catalyzed C-H Silylation
The core of this technological advancement lies in the unique catalytic cycle facilitated by the ruthenium complex, which enables the selective activation of the ortho C-H bond on the aryl ring directed by the pyridine nitrogen atom. The general reaction scheme illustrates how the 2-arylpyridine substrate coordinates with the ruthenium center, positioning the ortho C-H bond for activation.
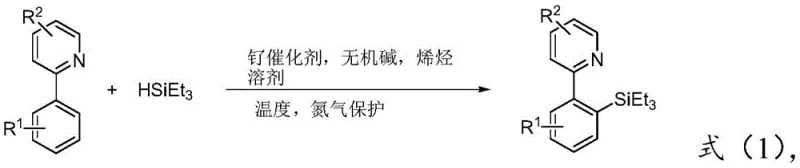
In this mechanism, the unsaturated olefin, preferably norbornene, acts as a crucial transient mediator or hydrogen acceptor that facilitates the turnover of the catalytic cycle. The inorganic base, such as potassium acetate, plays a vital role in neutralizing acidic byproducts and maintaining the active state of the catalyst. This synergistic interaction between the ruthenium catalyst, the olefin additive, and the base allows for the direct insertion of the triethylsilyl group into the C-H bond with high regioselectivity. Understanding this mechanism is critical for R&D teams, as it highlights the importance of precise stoichiometric control—specifically the molar ratios of substrate to silane to catalyst—to maximize yield and minimize side reactions.
Furthermore, the impurity profile of the final product is significantly improved compared to electrophilic substitution methods. Since the reaction avoids the formation of large amounts of inorganic salts and relies on a highly selective catalytic pathway, the resulting crude mixture is cleaner and easier to purify. The patent data indicates that simple column chromatography is sufficient to isolate the pure product, suggesting that the formation of regioisomers or over-silylated byproducts is minimal. This high level of selectivity ensures that the final API intermediate meets stringent purity specifications, reducing the risk of toxic impurities carrying over into the final drug substance.
How to Synthesize 2-Aryl Ortho-Substituted Triethyl Silicon Pyridine Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to ensure reproducibility and safety on a larger scale. The process involves combining the 2-arylpyridine substrate with a slight excess of triethylsilane, a catalytic amount of the ruthenium complex, and the necessary additives in a suitable solvent like toluene. The reaction is typically conducted under an inert atmosphere, such as nitrogen protection, to prevent oxidation of the sensitive catalyst or reagents. Heating the mixture to approximately 120°C for a duration of 16 hours allows the transformation to proceed to completion. Detailed standardized synthesis steps see the guide below.
- Combine 2-arylpyridine, triethylsilane, inorganic base (e.g., potassium acetate), unsaturated olefin (e.g., norbornene), and ruthenium catalyst in a solvent like toluene.
- Heat the reaction mixture to approximately 120°C under nitrogen protection with electromagnetic stirring for 16 hours.
- Remove the solvent via rotary evaporation and purify the crude product using column chromatography to isolate the target silylated compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the shift to this ruthenium-catalyzed methodology offers tangible benefits that extend beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the supply chain for raw materials. By replacing scarce and volatile noble metals like rhodium and iridium with abundant and stable ruthenium complexes, manufacturers can secure a more reliable supply of critical catalysts, thereby mitigating the risk of production delays caused by material shortages. Additionally, the one-pot nature of the reaction reduces the number of unit operations required, which directly correlates to lower energy consumption and reduced demand for processing equipment and labor.
- Cost Reduction in Manufacturing: The economic impact of switching to this method is profound, primarily driven by the substitution of expensive catalysts with affordable alternatives. Since the ruthenium catalyst is used in small molar amounts and is significantly cheaper than its rhodium or iridium counterparts, the direct material cost per kilogram of product is substantially lowered. Moreover, the elimination of intermediate isolation steps means that solvents and consumables associated with multiple workups are saved, further driving down the variable costs of production. This efficiency allows for a more competitive pricing structure for the final high-purity pharmaceutical intermediates without sacrificing quality.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to a more resilient supply chain. The tolerance of the method to various functional groups, including electron-donating and electron-withdrawing substituents, means that a single standardized protocol can be adapted for a wide range of substrate derivatives. This flexibility reduces the need for developing unique processes for each new analog, accelerating the timeline from laboratory discovery to commercial supply. Consequently, suppliers can respond more rapidly to fluctuating market demands, ensuring consistent availability of critical building blocks for downstream drug synthesis.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this method aligns well with green chemistry principles. The reduction in waste generation, particularly the avoidance of large quantities of inorganic salts typical of electrophilic substitutions, simplifies waste treatment and disposal procedures. The use of common solvents like toluene and the ability to run the reaction in a single vessel make the process highly scalable from pilot plant to full commercial production. This scalability ensures that the method can meet the volume requirements of global pharmaceutical markets while maintaining strict adherence to environmental regulations regarding hazardous waste.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the method's practical application and advantages over legacy techniques.
Q: Why is the ruthenium catalyst preferred over rhodium or iridium for this silylation?
A: While rhodium and iridium complexes are effective for C-H activation, they are significantly more expensive and less available for large-scale industrial applications. The patented method utilizes relatively inexpensive ruthenium complexes, such as tris(triphenylphosphine)carbonyl ruthenium hydrochloride, which drastically reduces raw material costs without compromising catalytic efficiency.
Q: What are the key advantages of the one-pot synthesis method described in the patent?
A: The one-pot methodology eliminates the need for isolating and purifying intermediates during the conversion process. This simplifies the operational workflow, reduces solvent consumption, minimizes waste generation, and shortens the overall production cycle, leading to substantial improvements in process efficiency and environmental compliance.
Q: Can this method tolerate various functional groups on the aryl ring?
A: Yes, the method demonstrates excellent functional group compatibility. The patent examples show successful silylation of substrates containing electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups like trifluoromethyl, indicating robust applicability for synthesizing diverse pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Aryl Ortho-Substituted Triethyl Silicon Pyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the one described in CN109651421B for the production of next-generation pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs, guaranteeing that every batch of 2-aryl ortho-substituted triethyl silicon pyridine meets the exacting standards required by global regulatory bodies.
We invite forward-thinking pharmaceutical companies and research institutions to collaborate with us to leverage this cost-effective synthesis route. By partnering with our technical procurement team, you can obtain a Customized Cost-Saving Analysis tailored to your specific project needs. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise can accelerate your development timelines and optimize your manufacturing costs for these valuable silicon-containing heterocycles.
