Advanced Palladium-Catalyzed Carbonylation for Scalable 2,3-Dihydroquinolone Production
Advanced Palladium-Catalyzed Carbonylation for Scalable 2,3-Dihydroquinolone Production
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct nitrogen-containing heterocycles, particularly the 2,3-dihydroquinolone scaffold, which serves as a critical pharmacophore in numerous bioactive molecules. As detailed in the groundbreaking patent CN112239456B, a novel preparation method has been established that leverages transition metal palladium catalysis to efficiently synthesize substituted 2,3-dihydroquinolone compounds. This technology represents a significant leap forward in synthetic organic chemistry, addressing long-standing challenges regarding safety, substrate compatibility, and operational simplicity. The core innovation lies in the utilization of N-pyridine sulfonyl-o-iodoaniline and various olefins as starting materials, reacting under controlled conditions to yield high-purity intermediates essential for drug discovery and development. For R&D directors and procurement specialists alike, understanding the nuances of this patented process is vital for securing a reliable pharmaceutical intermediates supplier capable of delivering complex scaffolds with consistency.
The biological significance of the 2,3-dihydroquinolone skeleton cannot be overstated, as it forms the structural backbone of compounds exhibiting potent anti-cancer and analgesic activities. Historical data indicates that specific derivatives within this class have demonstrated significant efficacy against human cancer cell lines, validating the therapeutic potential of this chemical space. 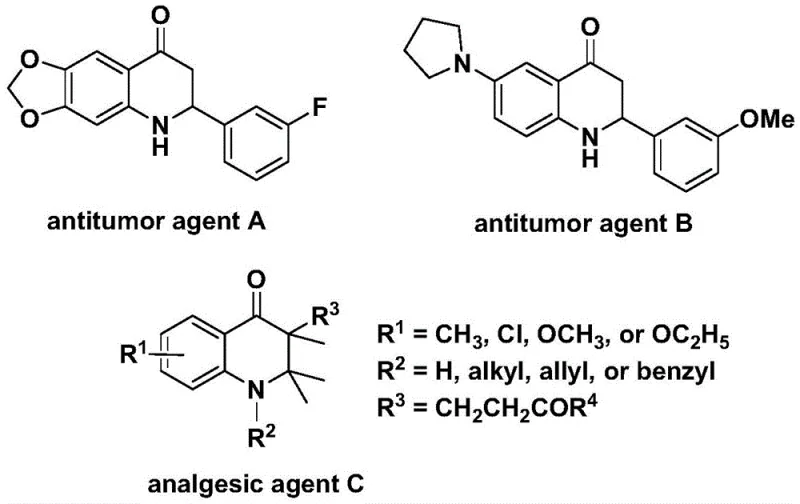 Consequently, the ability to access diverse analogs through efficient synthetic routes is a strategic priority for any organization focused on oncology or pain management therapeutics. The method described in CN112239456B not only provides access to these core structures but does so with a level of modularity that allows for the rapid generation of structure-activity relationship (SAR) libraries. This capability is indispensable for modern medicinal chemistry teams aiming to optimize lead compounds while maintaining strict control over impurity profiles and synthetic feasibility.
Consequently, the ability to access diverse analogs through efficient synthetic routes is a strategic priority for any organization focused on oncology or pain management therapeutics. The method described in CN112239456B not only provides access to these core structures but does so with a level of modularity that allows for the rapid generation of structure-activity relationship (SAR) libraries. This capability is indispensable for modern medicinal chemistry teams aiming to optimize lead compounds while maintaining strict control over impurity profiles and synthetic feasibility.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of carbonyl-containing heterocycles often relied on direct carbonylation using carbon monoxide gas, a reagent that poses severe safety hazards and logistical challenges in a production environment. Handling high-pressure CO gas requires specialized equipment, rigorous safety protocols, and often limits the scalability of the reaction due to mass transfer limitations between the gas and liquid phases. Furthermore, conventional methods frequently suffer from poor functional group tolerance, necessitating extensive protection and deprotection sequences that drive up costs and extend lead times. Many existing protocols also struggle with regioselectivity issues when dealing with unsymmetrical olefins, leading to complex mixtures that are difficult to purify. These inefficiencies create bottlenecks in the supply chain, making it difficult for procurement managers to secure cost reduction in API manufacturing without compromising on quality or safety standards. The reliance on hazardous gases also complicates regulatory compliance and environmental reporting, adding another layer of administrative burden to chemical production.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN112239456B introduces a paradigm shift by employing a solid carbon monoxide surrogate, specifically 1,3,5-trimesic acid phenyl ester, which releases CO in situ under thermal conditions. This innovation effectively mitigates the risks associated with gaseous CO, allowing the reaction to be performed in standard laboratory glassware or reactors without the need for high-pressure autoclaves. The process operates at a moderate temperature range of 100-120°C, typically around 110°C, using dioxane as a preferred solvent to ensure complete dissolution of reactants and optimal catalytic activity. 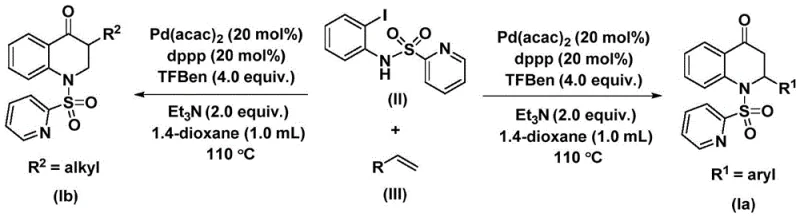 This approach significantly simplifies the operational workflow, enabling chemists to focus on substrate design rather than engineering constraints. The compatibility with a wide array of olefins, including those bearing aryl, alkyl, or silyl groups, underscores the versatility of this system. By eliminating the need for hazardous gas handling, this novel approach facilitates the commercial scale-up of complex organic syntheses, offering a safer and more sustainable pathway for producing high-value intermediates.
This approach significantly simplifies the operational workflow, enabling chemists to focus on substrate design rather than engineering constraints. The compatibility with a wide array of olefins, including those bearing aryl, alkyl, or silyl groups, underscores the versatility of this system. By eliminating the need for hazardous gas handling, this novel approach facilitates the commercial scale-up of complex organic syntheses, offering a safer and more sustainable pathway for producing high-value intermediates.
Mechanistic Insights into Pd-Catalyzed Carbonylative Cyclization
To fully appreciate the technical robustness of this synthesis, one must delve into the mechanistic intricacies of the palladium-catalyzed cycle. The reaction initiates with the oxidative addition of the palladium catalyst, specifically bis(acetylacetone)palladium or Pd(acac)2, into the carbon-iodine bond of the N-pyridine sulfonyl-o-iodoaniline substrate. This step generates a reactive aryl-palladium intermediate, which is stabilized by the bidentate phosphine ligand, 1,3-bis(diphenylphosphino)propane (dppp). The choice of ligand is critical, as it modulates the electronic and steric environment around the metal center, facilitating the subsequent insertion steps. Following oxidative addition, the carbon monoxide released from the phenyl ester surrogate inserts into the palladium-carbon bond, forming an acyl-palladium species. This migratory insertion is the key carbonylation step that introduces the ketone functionality into the final heterocyclic ring. The precision of this step ensures that the carbonyl group is installed at the exact position required for the 2,3-dihydroquinolone architecture, minimizing the formation of regioisomeric byproducts.
Subsequently, the olefin substrate coordinates to the palladium center and undergoes insertion into the acyl-palladium bond, generating a new alkyl-palladium intermediate. This step dictates the stereochemistry and substitution pattern at the 3-position of the quinolone ring. The system exhibits remarkable tolerance for steric bulk, accommodating substrates with ortho-substituents or bulky alkyl chains without significant loss in efficiency. The catalytic cycle concludes with a reductive elimination step, which releases the final substituted 2,3-dihydroquinolone product and regenerates the active palladium(0) species to continue the cycle. 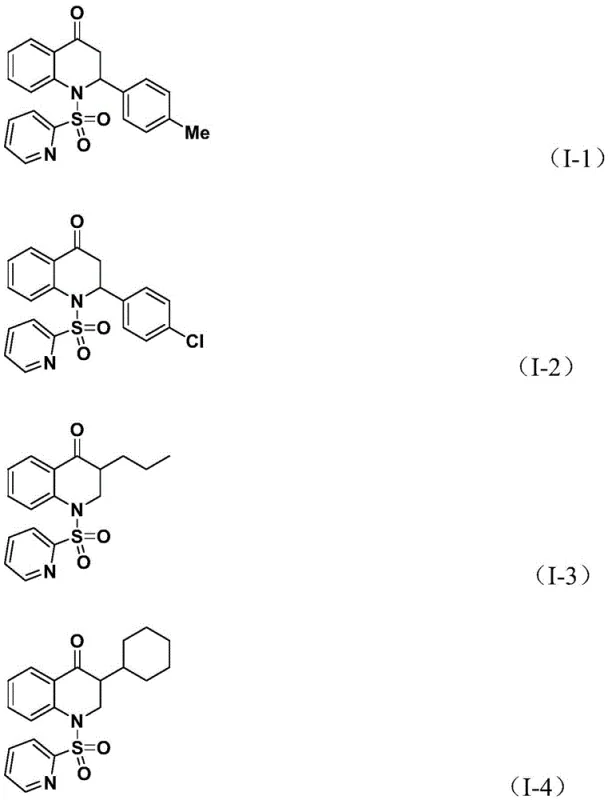 This mechanistic pathway is highly efficient, with reported reaction times ranging from 24 to 48 hours to ensure complete conversion. The use of triethylamine as an additive further aids in neutralizing acidic byproducts and maintaining the catalytic turnover. For R&D teams, understanding this mechanism provides confidence in the reproducibility of the process and the ability to troubleshoot potential deviations during technology transfer.
This mechanistic pathway is highly efficient, with reported reaction times ranging from 24 to 48 hours to ensure complete conversion. The use of triethylamine as an additive further aids in neutralizing acidic byproducts and maintaining the catalytic turnover. For R&D teams, understanding this mechanism provides confidence in the reproducibility of the process and the ability to troubleshoot potential deviations during technology transfer.
How to Synthesize Substituted 2,3-Dihydroquinolone Efficiently
Implementing this synthesis in a practical setting requires adherence to specific operational parameters to maximize yield and purity. The protocol begins with the precise weighing of N-pyridine sulfonyl-o-iodoaniline and the chosen olefin, ensuring a molar ratio that favors product formation, typically with an excess of the olefin to drive the equilibrium forward. The catalyst system, comprising Pd(acac)2 and dppp, is added alongside the solid CO source and the base in anhydrous 1,4-dioxane. It is imperative that the solvent is dry and oxygen-free to prevent catalyst deactivation. The mixture is then heated to 110°C, a temperature that balances reaction kinetics with the stability of the reagents. Detailed standardized synthesis steps are provided below to guide process chemists through the execution of this transformation.
- Prepare the reaction mixture by combining N-pyridine sulfonyl-o-iodoaniline, olefin substrate, Pd(acac)2 catalyst, dppp ligand, and triethylamine in anhydrous dioxane.
- Add the solid carbon monoxide surrogate, 1,3,5-trimesic acid phenyl ester, to the reaction vessel to ensure safe and controlled CO release during heating.
- Heat the mixture to 110°C for 24-48 hours under inert atmosphere, followed by filtration and silica gel column chromatography to isolate the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented methodology offers substantial benefits for procurement managers and supply chain heads tasked with optimizing costs and ensuring continuity. The primary advantage lies in the drastic simplification of the reaction infrastructure; by removing the requirement for high-pressure gas lines and specialized containment systems, capital expenditure for manufacturing facilities is significantly reduced. This translates directly into lower overhead costs per kilogram of product produced. Furthermore, the use of commercially available starting materials, such as simple olefins and iodoanilines, ensures a stable and diversified supply base. This reduces the risk of raw material shortages that can plague more exotic synthetic routes. The robustness of the reaction conditions also implies a higher success rate during batch production, minimizing waste and rework. For organizations seeking a reliable agrochemical intermediate supplier or pharma partner, this process reliability is a key differentiator that safeguards production schedules.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous carbon monoxide gas cylinders removes a significant line item from the operational budget, while the high atom economy of the carbonylation step ensures that raw materials are converted into product with minimal waste. The use of a solid CO surrogate also simplifies storage and handling logistics, reducing the need for specialized safety training and monitoring equipment. Additionally, the high yields achieved across a broad substrate scope mean that less starting material is required to produce the same amount of API intermediate, driving down the cost of goods sold. The simplified post-treatment process, involving standard filtration and chromatography, further reduces labor and solvent consumption costs associated with purification.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals like dioxane, triethylamine, and common olefins ensures that the supply chain is resilient to market fluctuations affecting niche reagents. Since the reaction does not depend on custom-synthesized catalysts or unstable intermediates, lead times for raw material procurement are minimized. This stability allows for better forecasting and inventory management, ensuring that production lines can run continuously without interruption. The scalability of the process from gram to multi-kilogram scales means that suppliers can ramp up production quickly in response to increased demand, providing a buffer against supply shocks. This reliability is crucial for maintaining the continuity of downstream drug manufacturing processes.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of toxic gas emissions align perfectly with modern environmental, health, and safety (EHS) standards. The solid waste generated is primarily organic and can be managed through standard disposal protocols, avoiding the complexities associated with hazardous gas scrubbing systems. The process is inherently safer, reducing the risk of workplace accidents and associated liabilities. Moreover, the high selectivity of the reaction reduces the formation of difficult-to-remove impurities, simplifying the wastewater treatment profile. This environmental compatibility makes the process attractive for manufacturing in regions with strict regulatory oversight, facilitating global distribution of the final intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical application of the method. Understanding these details helps stakeholders make informed decisions about integrating this chemistry into their existing portfolios. The responses cover aspects ranging from safety protocols to substrate limitations, ensuring a comprehensive overview of the technology's capabilities.
Q: What are the safety advantages of this carbonylation method compared to traditional CO gas methods?
A: This protocol utilizes 1,3,5-trimesic acid phenyl ester as a solid carbon monoxide surrogate, eliminating the need for high-pressure CO gas cylinders and significantly reducing operational hazards in large-scale manufacturing.
Q: What is the substrate scope for the olefin component in this reaction?
A: The method demonstrates broad compatibility, successfully accommodating both aryl-substituted olefins (such as styrenes with electron-donating or withdrawing groups) and alkyl-substituted olefins, including cyclic and linear chains.
Q: How does the pyridine sulfonyl protecting group influence the reaction outcome?
A: The N-pyridine sulfonyl group acts as a robust directing group that facilitates the palladium insertion and subsequent cyclization, while also allowing for potential downstream deprotection to access free NH quinolone derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-Dihydroquinolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing high-quality heterocyclic building blocks for the development of next-generation therapeutics. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. We are committed to meeting stringent purity specifications through our rigorous QC labs, which utilize advanced analytical techniques to verify the identity and quality of every batch. Our facility is equipped to handle the specific requirements of palladium-catalyzed reactions, including proper metal scavenging to ensure residual catalyst levels meet ICH guidelines. By partnering with us, you gain access to a supply chain that prioritizes both technical excellence and operational reliability.
We invite you to collaborate with our technical procurement team to explore how this innovative synthesis route can benefit your specific project requirements. Whether you need a Customized Cost-Saving Analysis for your current supply chain or require specific COA data to validate our capabilities, we are ready to assist. Please reach out to request route feasibility assessments for your target molecules, and let us demonstrate how our expertise in fine chemical intermediates can accelerate your development timelines. Together, we can overcome synthetic challenges and bring life-saving medicines to market more efficiently.
