Advanced Palladium-Catalyzed Carbonylation for Scalable 2,3-Dihydroquinolone Pharmaceutical Intermediates
Advanced Palladium-Catalyzed Carbonylation for Scalable 2,3-Dihydroquinolone Pharmaceutical Intermediates
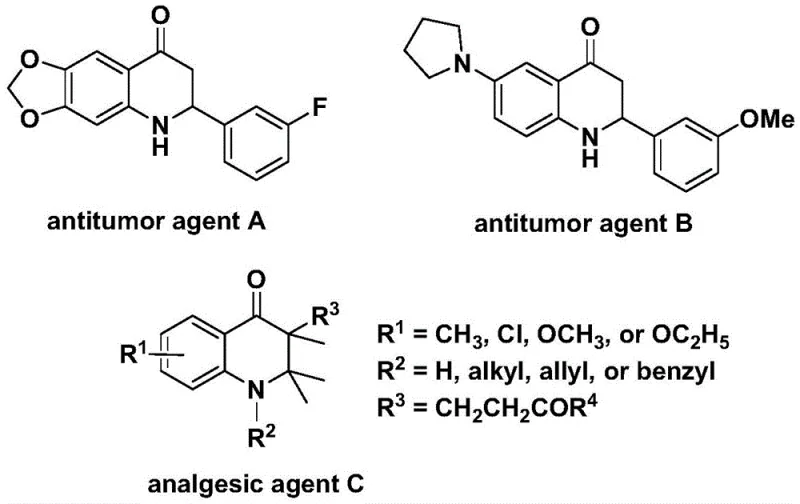
The landscape of heterocyclic chemistry is continuously evolving, driven by the demand for more efficient and safer synthetic routes to bioactive scaffolds. A pivotal development in this domain is detailed in patent CN112239456B, which discloses a robust preparation method for substituted 2,3-dihydroquinolone compounds. These nitrogen-containing carbonyl heterocycles are not merely academic curiosities; they form the core skeleton of numerous molecules with profound biological activities, ranging from potent antitumor agents to analgesics. As illustrated in the structural diversity of known bioactive molecules, the ability to access these scaffolds efficiently is critical for drug discovery pipelines. The disclosed technology leverages a transition metal palladium-catalyzed carbonylation reaction, utilizing N-pyridine sulfonyl-o-iodoaniline and olefins as key starting materials. This approach represents a significant leap forward in synthetic methodology, addressing long-standing challenges regarding safety, substrate scope, and operational simplicity that have historically plagued the industrial production of these valuable intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of carbonyl-containing heterocycles like 2,3-dihydroquinolones has often relied on direct carbonylation using carbon monoxide gas. While chemically effective, the use of gaseous CO presents severe logistical and safety hurdles for large-scale manufacturing. The toxicity of carbon monoxide requires specialized high-pressure equipment, rigorous leak detection systems, and extensive safety protocols, all of which drive up capital expenditure and operational costs. Furthermore, conventional methods often suffer from limited substrate tolerance, where sensitive functional groups on the aromatic ring or the olefin component may degrade under harsh reaction conditions. This lack of versatility forces process chemists to employ protecting group strategies, adding unnecessary steps, reducing overall atom economy, and generating excessive waste. Additionally, older catalytic systems frequently struggle with regioselectivity, leading to complex mixtures of isomers that are difficult and expensive to separate, ultimately compromising the purity required for pharmaceutical applications.
The Novel Approach
The methodology described in the patent offers a transformative solution by replacing hazardous carbon monoxide gas with a solid CO surrogate, specifically 1,3,5-trimesic acid phenol ester (TFBen). This substitution fundamentally alters the safety profile of the reaction, allowing it to be conducted in standard laboratory glassware or reactors without the need for high-pressure gas lines. The process utilizes a palladium catalyst system, specifically bis(acetylacetone)palladium paired with a dppp ligand, which facilitates the smooth insertion of the carbonyl group under relatively mild thermal conditions (100-120°C). This novel approach not only enhances operator safety but also dramatically simplifies the reaction setup, making it highly amenable to scale-up. The method exhibits remarkable versatility, successfully synthesizing both 2-aryl and 3-alkyl substituted derivatives through simple substrate design. By enabling the direct coupling of diverse olefins with iodoaniline derivatives, this route provides a streamlined, one-pot strategy that bypasses the multi-step sequences typically required in legacy syntheses, thereby accelerating the timeline from benchtop discovery to commercial production.
Mechanistic Insights into Palladium-Catalyzed Carbonylative Cyclization
Understanding the mechanistic underpinnings of this transformation is essential for R&D directors aiming to optimize the process for specific API candidates. The reaction initiates with the oxidative addition of the palladium(0) species into the carbon-iodine bond of the N-pyridine sulfonyl-o-iodoaniline substrate. This step generates a crucial aryl-palladium intermediate, setting the stage for the subsequent carbonylation event. Unlike traditional methods where CO gas coordinates directly from the headspace, here the carbon monoxide is released in situ from the decomposition of the phenol ester surrogate. This controlled release ensures a steady concentration of CO at the catalytic center, promoting efficient insertion into the aryl-palladium bond to form an acyl-palladium intermediate. This precise control over CO availability is a key factor in minimizing side reactions such as homocoupling of the aryl halide, which is a common impurity in carbonylation chemistry.
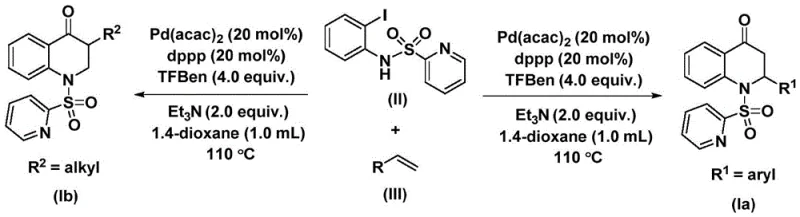
Following the formation of the acyl-palladium species, the olefin substrate coordinates to the metal center and undergoes migratory insertion. This step is critical for establishing the stereochemistry and regiochemistry of the final dihydroquinolone ring. The resulting alkyl-palladium intermediate then undergoes an intramolecular nucleophilic attack or reductive elimination, facilitated by the proximal nitrogen atom, to close the six-membered heterocyclic ring and regenerate the active palladium catalyst. The choice of the pyridine sulfonyl protecting group on the nitrogen is not arbitrary; it serves to activate the nitrogen for the cyclization step while remaining stable enough to withstand the reaction conditions. This mechanistic pathway ensures high conversion rates and excellent selectivity, as evidenced by the broad range of substrates tolerated, including those with electron-withdrawing halogens and electron-donating alkoxy groups. The robustness of this catalytic cycle allows for the synthesis of complex architectures with high fidelity, ensuring that the impurity profile remains manageable even when scaling to multi-kilogram batches.
How to Synthesize Substituted 2,3-Dihydroquinolone Efficiently
The practical implementation of this synthesis is designed for ease of execution, requiring standard organic synthesis techniques without the need for exotic equipment. The process involves charging a reaction vessel with the palladium catalyst, the dppp ligand, the solid CO surrogate, a base such as triethylamine, the iodoaniline derivative, and the chosen olefin in an aprotic solvent like dioxane. The mixture is then heated to promote the catalytic cycle. Detailed standard operating procedures for optimizing stoichiometry and workup are critical for maximizing yield and purity. For the complete, step-by-step standardized synthesis protocol including specific molar ratios and purification details, please refer to the technical guide below.
- Combine palladium catalyst, ligand, CO surrogate, additive, N-pyridine sulfonyl-o-iodoaniline, and olefin in an organic solvent.
- Heat the reaction mixture to 100-120°C and stir for 24-48 hours to facilitate the carbonylation cyclization.
- Filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the shift from gas-phase carbonylation to this solid-surrogate method represents a substantial opportunity for risk mitigation and cost optimization. The elimination of carbon monoxide gas removes a major regulatory and safety bottleneck, simplifying the permitting process for manufacturing facilities and reducing the insurance liabilities associated with toxic gas storage. This inherently safer process translates directly into lower operational overheads and a more resilient supply chain, as production is less susceptible to shutdowns caused by safety audits or gas supply disruptions. Furthermore, the use of commercially available starting materials, such as readily sourced olefins and iodoanilines, ensures a stable raw material supply base. The ability to source these precursors from multiple vendors reduces dependency on single-source suppliers, enhancing the overall security of supply for critical pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the simplification of the reaction infrastructure. By avoiding high-pressure reactors and specialized gas handling systems, capital investment for new production lines is significantly lowered. Additionally, the high reaction efficiency and substrate compatibility mean that fewer purification steps are required to remove complex byproducts, leading to reduced solvent consumption and waste disposal costs. The use of a solid CO surrogate also improves atom economy compared to methods that require excess gas pressure, ensuring that raw material costs are optimized. These factors combine to deliver a manufacturing process that is not only cheaper to operate but also more environmentally sustainable, aligning with modern green chemistry initiatives.
- Enhanced Supply Chain Reliability: The robustness of the catalytic system ensures consistent batch-to-batch quality, which is paramount for maintaining uninterrupted supply to downstream API manufacturers. The method's tolerance for various functional groups means that a single platform technology can be adapted to produce a wide library of analogues without requalifying the entire process for each new derivative. This flexibility allows for rapid response to changing market demands or clinical trial requirements. Moreover, the scalability of the reaction from gram to kilogram levels has been demonstrated, providing confidence that the supply can be ramped up quickly to meet commercial launch timelines without the typical teething problems associated with scaling novel chemistries.
- Scalability and Environmental Compliance: From an environmental perspective, the replacement of toxic CO gas with a solid ester significantly reduces the potential for atmospheric emissions and workplace exposure hazards. The reaction conditions are mild enough to minimize energy consumption for heating and cooling, contributing to a lower carbon footprint for the manufacturing process. The post-treatment procedure, involving simple filtration and column chromatography, generates waste streams that are easier to manage and treat compared to those from heavy metal-catalyzed processes that require extensive scavenging. This alignment with environmental, social, and governance (ESG) goals makes the technology attractive for partnerships with major pharmaceutical companies that have strict sustainability mandates for their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed carbonylation technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of the method's capabilities and limitations for potential partners.
Q: What is the primary advantage of using 1,3,5-trimesic acid phenol ester in this synthesis?
A: It acts as a solid carbon monoxide surrogate, eliminating the need for handling toxic and hazardous CO gas, thereby significantly improving operational safety and simplifying equipment requirements for scale-up.
Q: What types of substituents are compatible with this palladium-catalyzed protocol?
A: The method demonstrates excellent substrate compatibility, tolerating various functional groups including methyl, tert-butyl, methoxy, fluorine, and chlorine on the aryl ring, as well as linear, branched alkyl, and silyl groups.
Q: How does this method impact the purity profile of the final API intermediate?
A: The use of specific ligands like dppp and mild reaction conditions minimizes side reactions and byproduct formation, resulting in a cleaner crude reaction mixture that simplifies downstream purification and ensures high-purity specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Substituted 2,3-Dihydroquinolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the one described in CN112239456B for accelerating drug development. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to industrial manufacturing is seamless. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our facility is designed to handle complex organometallic reactions safely and efficiently, leveraging the latest advancements in catalytic chemistry to drive down costs and improve sustainability for our global partners.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific molecule. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your supply chain resilience and reduce your overall time to market.
