Advanced Zinc-Mediated Cyclization for Scalable N-Arylindole-3-Carbonitrile Derivatives Production
Advanced Zinc-Mediated Cyclization for Scalable N-Arylindole-3-Carbonitrile Derivatives Production
The pharmaceutical industry continuously seeks robust and cost-effective methodologies for constructing complex heterocyclic scaffolds, particularly the N-arylindole core which is prevalent in numerous bioactive molecules such as Sertindole. Patent CN102020601B introduces a transformative synthetic strategy that addresses the longstanding challenges associated with traditional indole synthesis, specifically focusing on the efficient construction of N-arylindole-3-carbonitrile derivatives. This technology leverages a mild, zinc-mediated intramolecular cyclization pathway that bypasses the need for precious metal catalysts, thereby offering a distinct advantage in terms of both economic feasibility and environmental compliance for large-scale manufacturing. By utilizing readily available halogenating agents and zinc acetate dihydrate, the process ensures high purity profiles while maintaining operational simplicity, making it an attractive option for reliable pharmaceutical intermediate supplier networks aiming to optimize their production pipelines. The versatility of this method allows for significant structural diversity, enabling the introduction of various functional groups at the 3-position of the indole ring before the final cyclization step occurs. This strategic flexibility is crucial for medicinal chemists who require rapid access to diverse analog libraries for structure-activity relationship studies without compromising on yield or scalability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of N-arylindole rings has relied heavily on methodologies that present substantial hurdles for industrial application, often involving harsh reaction conditions or prohibitively expensive reagents. Traditional approaches such as Fischer indole synthesis frequently require strong acidic conditions and high temperatures, which can lead to decomposition of sensitive functional groups and generate significant amounts of hazardous waste. Furthermore, transition metal-catalyzed coupling strategies, while effective for specific substrates, often depend on palladium or ruthenium catalysts that are not only costly but also pose severe challenges regarding residual metal contamination in the final active pharmaceutical ingredient. These heavy metal residues necessitate additional purification steps, such as scavenging or extensive chromatography, which drastically increase processing time and overall manufacturing costs. Additionally, many conventional routes lack the flexibility to introduce diverse substituents on the indole benzene ring late in the synthesis, limiting the ability to explore chemical space efficiently. The reliance on specialized reagents like hypervalent iodides or specific halo-arenes further complicates the supply chain, creating vulnerabilities in procurement and potential delays in project timelines for drug development programs.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes a streamlined two-step sequence involving allylic halogenation followed by zinc-mediated cyclization to achieve the desired N-arylindole-3-carbonitrile structure. This methodology operates under significantly milder conditions, typically employing room temperature for the halogenation step and reflux in common aprotic solvents for the cyclization, thereby minimizing energy consumption and thermal stress on the molecules. The use of zinc acetate dihydrate as a promoter is a game-changer, as zinc is abundant, inexpensive, and generally recognized as safe, eliminating the regulatory burden associated with heavy metal clearance. 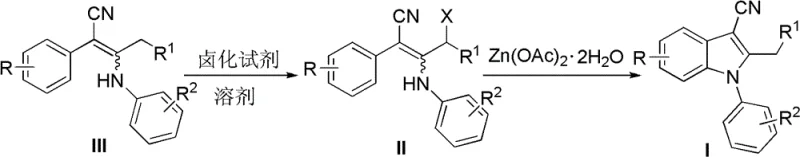 As illustrated in the reaction scheme, the transformation proceeds from the 2-phenyl-3-anilino-2-enenitrile precursor through a halogenated intermediate directly to the cyclized product without the need for isolating unstable intermediates. This telescoped nature of the reaction reduces unit operations, solvent usage, and overall processing time, contributing to substantial cost reduction in API manufacturing. Moreover, the compatibility of this method with a wide range of substituents on both the aniline and phenylacetonitrile moieties ensures that it can be adapted for the commercial scale-up of complex polymer additives or specialty chemicals beyond just pharmaceutical applications.
As illustrated in the reaction scheme, the transformation proceeds from the 2-phenyl-3-anilino-2-enenitrile precursor through a halogenated intermediate directly to the cyclized product without the need for isolating unstable intermediates. This telescoped nature of the reaction reduces unit operations, solvent usage, and overall processing time, contributing to substantial cost reduction in API manufacturing. Moreover, the compatibility of this method with a wide range of substituents on both the aniline and phenylacetonitrile moieties ensures that it can be adapted for the commercial scale-up of complex polymer additives or specialty chemicals beyond just pharmaceutical applications.
Mechanistic Insights into Zinc-Mediated Intramolecular Cyclization
The core of this synthetic breakthrough lies in the precise mechanistic pathway where the initial allylic halogenation activates the substrate for subsequent nucleophilic attack by the aniline nitrogen. In the first stage, the 2-phenyl-3-anilino-2-enenitrile compound is treated with a halogenating reagent such as N-bromosuccinimide (NBS) or N-chlorosuccinimide (NCS) in an aprotic solvent like 1,2-dichloroethane or acetonitrile. This step selectively introduces a halogen atom at the allylic position, generating a highly reactive electrophilic center that is primed for intramolecular displacement. The choice of solvent is critical here, as aprotic environments prevent side reactions such as hydrolysis of the nitrile group or solvolysis of the halogenated intermediate, ensuring high fidelity in the transformation. Following halogenation, the addition of zinc acetate dihydrate facilitates the cyclization by coordinating with the nitrogen lone pair and potentially activating the nitrile group, lowering the activation energy for the ring-closing step. This coordination effect promotes the nucleophilic attack of the aniline nitrogen onto the activated allylic carbon, resulting in the formation of the five-membered pyrrole ring characteristic of the indole scaffold. The entire process is driven by thermodynamic stability gained upon aromatization of the indole system, which serves as a powerful driving force for the reaction to proceed to completion even under relatively mild thermal conditions.
From an impurity control perspective, this mechanism offers distinct advantages by minimizing the formation of regioisomers and polymeric byproducts that often plague radical-based or acid-catalyzed indole syntheses. The specificity of the allylic halogenation ensures that the reactive site is well-defined, reducing the likelihood of random substitution on the aromatic rings which could lead to difficult-to-separate impurities. Furthermore, the use of zinc salts helps to buffer the reaction medium, preventing the accumulation of acidic byproducts like succinimide or hydrogen halides that could otherwise catalyze decomposition pathways. 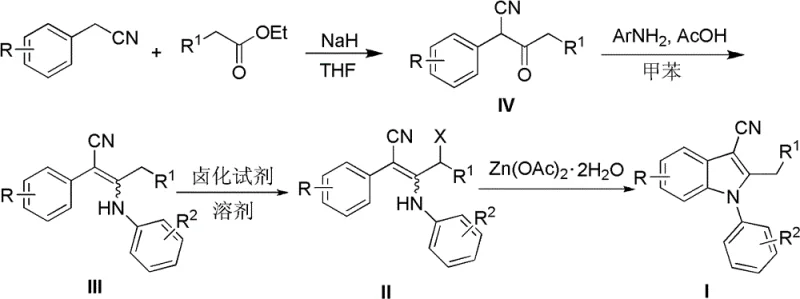 As shown in the comprehensive synthesis pathway, the method tolerates a variety of electron-donating and electron-withdrawing groups, such as methoxy, chloro, and methyl substituents, without significant loss in efficiency. This robustness implies that the impurity profile remains consistent and manageable across different batches, which is a critical requirement for meeting stringent quality standards in the production of high-purity OLED material or pharmaceutical intermediates. The ability to control the stereochemistry and regiochemistry of the cyclization ensures that the final product possesses the correct structural integrity required for downstream biological activity, thereby reducing the risk of batch rejection during quality control testing.
As shown in the comprehensive synthesis pathway, the method tolerates a variety of electron-donating and electron-withdrawing groups, such as methoxy, chloro, and methyl substituents, without significant loss in efficiency. This robustness implies that the impurity profile remains consistent and manageable across different batches, which is a critical requirement for meeting stringent quality standards in the production of high-purity OLED material or pharmaceutical intermediates. The ability to control the stereochemistry and regiochemistry of the cyclization ensures that the final product possesses the correct structural integrity required for downstream biological activity, thereby reducing the risk of batch rejection during quality control testing.
How to Synthesize N-Arylindole-3-Carbonitrile Efficiently
To implement this synthesis effectively, operators must adhere to strict anhydrous conditions during the halogenation phase to prevent the degradation of the reactive intermediate and ensure optimal conversion rates. The process begins with dissolving the enenitrile precursor in a dry aprotic solvent, followed by the controlled addition of the halogenating agent at room temperature to manage exothermicity. Once the halogenation is complete as monitored by TLC, the zinc acetate dihydrate is introduced directly into the same vessel, and the mixture is heated to reflux to drive the cyclization to completion. This streamlined workflow minimizes material handling and exposure to air, which is essential for maintaining the stability of the intermediates and achieving the reported high yields. By following these standardized protocols, manufacturing teams can reliably produce multi-kilogram quantities of the target indole derivatives with consistent quality attributes suitable for clinical trial material supply.
- Dissolve the 2-phenyl-3-anilino-2-enenitrile precursor in an aprotic solvent such as 1,2-dichloroethane and treat with a halogenating agent like N-bromosuccinimide at room temperature.
- Without isolation, add zinc acetate dihydrate directly to the reaction mixture and heat to reflux to induce intramolecular ring closure.
- Upon completion, remove the solvent under reduced pressure and purify the crude N-arylindole-3-carbonitrile product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this zinc-mediated synthesis route presents a compelling value proposition centered around cost efficiency, supply security, and operational scalability. The shift away from precious metal catalysts to commodity chemicals like zinc acetate and NBS fundamentally alters the cost structure of the manufacturing process, removing the volatility associated with palladium and rhodium pricing markets. This transition not only lowers the direct material costs but also simplifies the waste management protocols, as zinc residues are far easier and cheaper to treat than heavy metal contaminants, leading to significant indirect savings in environmental compliance and disposal fees. Furthermore, the reliance on widely available starting materials ensures a resilient supply chain that is less susceptible to geopolitical disruptions or single-source supplier bottlenecks, guaranteeing continuity of supply for critical drug programs. The mild reaction conditions also translate to reduced energy consumption and lower demands on reactor infrastructure, allowing existing facilities to handle production without requiring capital-intensive upgrades or specialized high-pressure equipment.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts such as palladium or ruthenium removes a major cost driver from the bill of materials, while simultaneously reducing the expense associated with metal scavenging resins and analytical testing for residual metals. By utilizing zinc acetate dihydrate, a low-cost and abundant reagent, the process achieves a leaner cost profile that enhances margin potential for high-volume commercial production. Additionally, the telescoped nature of the reaction reduces solvent consumption and labor hours by combining two synthetic steps into a single pot operation, further driving down the cost of goods sold. These cumulative efficiencies result in substantial cost savings that can be passed on to clients or reinvested into R&D initiatives, strengthening the competitive position of the manufacturing entity in the global market.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including substituted phenylacetonitriles, anilines, and N-halosuccinimides, are commodity chemicals produced by multiple vendors worldwide, ensuring a diversified and secure sourcing strategy. This abundance mitigates the risk of supply shortages that often occur with specialized ligands or custom-synthesized catalysts used in alternative methods, providing procurement teams with greater negotiating power and flexibility. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in reagent quality, reducing the likelihood of batch failures due to raw material specifications. Consequently, this reliability supports just-in-time manufacturing models and enables faster response times to fluctuating market demands for key pharmaceutical intermediates.
- Scalability and Environmental Compliance: The mild thermal profile and absence of hazardous reagents make this process inherently safer and easier to scale from laboratory benchtop to multi-ton commercial production without encountering significant engineering hurdles. The use of zinc salts aligns with green chemistry principles by reducing the toxicity of the waste stream, facilitating easier wastewater treatment and lowering the environmental footprint of the manufacturing site. This compliance with increasingly stringent environmental regulations future-proofs the production asset against potential regulatory changes, ensuring long-term operational viability. Moreover, the high atom economy of the cyclization step minimizes waste generation, contributing to a more sustainable manufacturing lifecycle that appeals to environmentally conscious stakeholders and partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method, derived directly from the patent specifications and practical application scenarios. These insights are intended to clarify the operational parameters and strategic benefits for stakeholders evaluating this technology for their own supply chains. Understanding these nuances is essential for making informed decisions about process adoption and partnership opportunities in the fine chemical sector.
Q: What are the primary advantages of this zinc-mediated method over traditional palladium catalysis?
A: This method eliminates the need for expensive transition metal catalysts like palladium or ruthenium, significantly reducing raw material costs and simplifying the removal of heavy metal residues from the final API.
Q: Can the substitution pattern on the indole benzene ring be varied using this protocol?
A: Yes, the protocol allows for diverse substitutions (R groups) on the benzene ring prior to the final ring closure, enabling the synthesis of a wide library of derivatives for SAR studies.
Q: What is the typical yield range for this cyclization process?
A: According to the patent data, the process demonstrates ideal yields ranging from 50% to 93% across various substrates, indicating robust performance for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Arylindole-3-Carbonitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a dependable partner who can translate innovative patent technologies into commercial reality with speed and precision. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from clinical trials to market launch is seamless and uninterrupted. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of N-arylindole-3-carbonitrile meets the highest industry standards for potency and impurity profiles. Our commitment to quality assurance means that you can rely on us for consistent supply of high-purity pharmaceutical intermediates that support your drug development timelines without compromise.
We invite you to engage with our technical procurement team to discuss how this zinc-mediated synthesis route can be tailored to your specific project needs and volume requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits this method offers compared to your current supply sources. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that will demonstrate our capability to deliver value-driven solutions for your complex chemical synthesis challenges.
