Advanced Zinc-Mediated Cyclization for Scalable N-Arylindole-3-Carbonitrile Manufacturing
Advanced Zinc-Mediated Cyclization for Scalable N-Arylindole-3-Carbonitrile Manufacturing
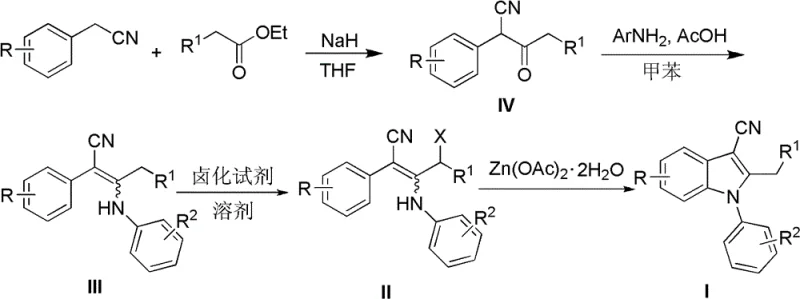
The pharmaceutical industry continuously seeks robust, scalable, and cost-effective methodologies for constructing privileged heterocyclic scaffolds, among which the N-arylindole core stands out as a critical structural motif found in numerous bioactive molecules, including the antipsychotic agent Sertindole. Patent CN102020601B introduces a transformative synthetic strategy for accessing N-arylindole-3-carbonitrile derivatives, addressing long-standing challenges associated with traditional indole synthesis. This proprietary technology leverages a tandem sequence involving allylic halogenation followed by a zinc acetate-mediated intramolecular cyclization, offering a distinct alternative to precious metal-catalyzed processes. By shifting away from expensive palladium or ruthenium catalysts towards earth-abundant zinc species, this method not only enhances the economic viability of the process but also simplifies the impurity profile, a factor of paramount importance for regulatory compliance in active pharmaceutical ingredient (API) manufacturing. The versatility of this approach allows for the introduction of diverse functional groups at the 3-position of the indole ring via the nitrile handle, providing medicinal chemists with a powerful vector for late-stage diversification.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the N-arylindole scaffold has relied heavily on classical methodologies such as the Fischer indole synthesis or transition metal-catalyzed cross-coupling reactions, both of which present significant drawbacks in a modern commercial context. The Fischer indole synthesis, while conceptually simple, often necessitates harsh acidic conditions and high temperatures that can lead to the degradation of sensitive functional groups and the formation of complex regioisomeric mixtures that are difficult to separate. Furthermore, strategies involving the direct arylation of indoles or the cyclization of o-haloanilines typically depend on palladium or copper catalysis, introducing the risk of toxic metal contamination that requires extensive and costly purification steps to meet strict International Council for Harmonisation (ICH) guidelines. These conventional routes frequently suffer from limited substrate scope, poor atom economy, and the generation of substantial chemical waste, creating bottlenecks in the supply chain for high-purity pharmaceutical intermediates. Additionally, the reliance on specialized ligands and inert atmosphere conditions for many metal-catalyzed variants increases operational complexity and capital expenditure, rendering them less attractive for large-scale production.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN102020601B offers a streamlined, operationally simple pathway that circumvents the need for precious metals entirely. The novel approach utilizes a readily accessible 2-phenyl-3-anilino-2-enenitrile precursor, which undergoes a selective allylic halogenation using reagents like N-bromosuccinimide (NBS) or N-chlorosuccinimide (NCS) under mild conditions. This intermediate is then subjected to an intramolecular cyclization promoted by zinc acetate dihydrate, a cheap and non-toxic Lewis acid, to forge the pyrrole ring efficiently. This methodology eliminates the requirement for cryogenic temperatures or high-pressure equipment, operating instead at ambient or reflux temperatures in common aprotic solvents such as 1,2-dichloroethane or acetonitrile. The result is a process that delivers ideal yields with exceptional regioselectivity, ensuring that the nitrile group remains intact at the 3-position for further functionalization. By decoupling the synthesis from volatile precious metal markets and complex catalytic systems, this route provides a reliable foundation for cost reduction in pharmaceutical intermediate manufacturing while maintaining high standards of chemical purity.
Mechanistic Insights into Zinc-Mediated Intramolecular Cyclization
The core innovation of this synthetic route lies in the mechanistic elegance of the zinc-mediated cyclization step, which facilitates the construction of the indole nucleus through a concerted electrophilic aromatic substitution-like pathway. Upon treatment of the enenitrile precursor with a halogenating agent, an allylic halide intermediate is generated in situ, activating the carbon chain for nucleophilic attack. The subsequent addition of zinc acetate dihydrate serves a dual purpose: it acts as a mild Lewis acid to coordinate with the nitrile nitrogen and the halogen leaving group, thereby increasing the electrophilicity of the allylic carbon, and simultaneously promotes the deprotonation of the aniline nitrogen to enhance its nucleophilicity. This coordination environment lowers the activation energy for the intramolecular ring closure, allowing the reaction to proceed smoothly under thermal conditions without the need for strong bases that might hydrolyze the sensitive nitrile functionality. The zinc species effectively templates the cyclization, ensuring that the reaction proceeds through the energetically favorable 5-exo-trig pathway to form the five-membered pyrrole ring exclusively.
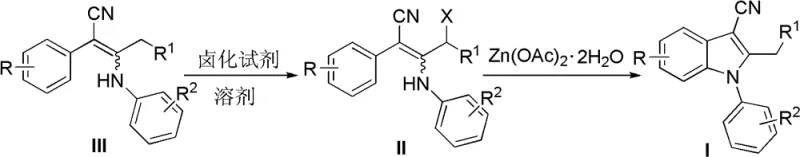
From an impurity control perspective, this mechanism offers significant advantages over radical-based or high-energy thermal cyclizations. The mild nature of the zinc acetate promoter minimizes side reactions such as polymerization of the enenitrile or over-halogenation of the aromatic rings, which are common pitfalls in aggressive halogenation protocols. The specificity of the zinc coordination ensures that the cyclization occurs selectively at the ortho-position of the aniline ring, preventing the formation of regioisomers that could complicate downstream purification. Furthermore, because the reaction does not involve transition metals with variable oxidation states like palladium or copper, there is no risk of generating metal-associated organic impurities or colored byproducts that are notoriously difficult to remove by crystallization. The resulting crude product typically exhibits high purity, often requiring only a simple recrystallization or flash chromatography to achieve pharmaceutical grade specifications. This clean reaction profile is critical for R&D directors focused on minimizing the genotoxic impurity burden and ensuring robust process validation for regulatory filings.
How to Synthesize N-Arylindole-3-Carbonitrile Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to solvent dryness and stoichiometry to maximize the efficiency of the halogenation-cyclization cascade. The process begins with the dissolution of the 2-phenyl-3-anilino-2-enenitrile starting material in an anhydrous aprotic solvent, followed by the controlled addition of the halogenating agent to generate the reactive intermediate. Once halogenation is confirmed complete via thin-layer chromatography, the zinc salt is introduced directly to the same pot, eliminating the need for intermediate isolation and reducing solvent consumption. The detailed standardized synthesis steps, including specific molar ratios, temperature profiles, and workup procedures optimized for reproducibility, are outlined below for technical reference.
- Dissolve the 2-phenyl-3-anilino-2-enenitrile precursor in an aprotic solvent such as 1,2-dichloroethane or acetonitrile.
- Add a halogenating agent like N-bromosuccinimide (NBS) at room temperature to effect allylic halogenation, forming the halo-intermediate in situ.
- Introduce zinc acetate dihydrate to the reaction mixture and heat to reflux to promote intramolecular cyclization, yielding the target N-arylindole derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this zinc-mediated technology translates into tangible strategic benefits that extend far beyond simple reaction yield. The primary driver of value is the drastic simplification of the raw material portfolio; by replacing expensive palladium catalysts and specialized phosphine ligands with commodity-grade zinc acetate and succinimide derivatives, the direct material cost of goods sold (COGS) is significantly reduced. This shift also mitigates supply chain risk, as zinc salts are produced in massive volumes for diverse industrial applications, ensuring consistent availability and price stability compared to precious metals which are subject to geopolitical volatility and mining constraints. Furthermore, the operational simplicity of the process—operating at atmospheric pressure and moderate temperatures—reduces the energy footprint of the manufacturing campaign and lowers the barrier for technology transfer between different production sites. The elimination of heavy metal scavenging resins and complex filtration steps shortens the overall cycle time, enhancing throughput capacity without the need for additional capital investment in specialized reactor hardware.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally superior due to the replacement of high-value transition metal catalysts with low-cost zinc reagents. In traditional palladium-catalyzed indole syntheses, the cost of the metal and ligand system can constitute a significant portion of the batch cost, and the subsequent removal of trace metals often requires expensive scavenging agents or multiple recrystallizations. By utilizing zinc acetate, which is orders of magnitude cheaper and easier to handle, the process eliminates these specific cost centers entirely. Additionally, the high atom economy of the cyclization step means that less raw material is wasted as byproduct, further driving down the effective cost per kilogram of the final API intermediate. This lean manufacturing approach allows for substantial margin improvement in the final drug product pricing.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of universally available reagents that do not rely on single-source suppliers or constrained mining outputs. Precious metal catalysts often face long lead times and allocation issues during periods of high demand, whereas zinc acetate and common halogenating agents like NBS are stocked by multiple global chemical distributors. This redundancy ensures that production schedules are not disrupted by raw material shortages. Moreover, the robustness of the reaction conditions means that the process is less sensitive to minor variations in utility quality or operator technique, leading to higher first-pass success rates and more predictable delivery timelines for downstream customers. This reliability is crucial for maintaining continuous manufacturing operations and meeting just-in-time delivery commitments.
- Scalability and Environmental Compliance: Scaling this chemistry from gram to ton scale is straightforward due to the absence of exothermic hazards associated with strong bases or pyrophoric reagents. The use of standard solvents like dichloroethane or acetonitrile allows for easy solvent recovery and recycling, aligning with green chemistry principles and reducing waste disposal costs. From a regulatory standpoint, the low toxicity profile of zinc compared to heavy metals simplifies the environmental impact assessment and wastewater treatment requirements. The process generates minimal hazardous waste, and the aqueous waste streams are easier to treat, facilitating compliance with increasingly stringent environmental regulations. This environmental compatibility not only reduces operational risk but also enhances the corporate sustainability profile of the manufacturing organization.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this N-arylindole synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent literature, ensuring accuracy and relevance for process development teams. Understanding these nuances is essential for evaluating the fit of this methodology within your existing manufacturing infrastructure and quality systems.
Q: How does this zinc-mediated method compare to palladium-catalyzed routes regarding metal residues?
A: Unlike palladium-catalyzed cross-couplings which require stringent and costly metal scavenging to meet ppm-level specifications, this zinc-mediated protocol utilizes inexpensive zinc salts that are significantly easier to remove during aqueous workup, simplifying purification and reducing downstream processing costs.
Q: Is this synthetic route suitable for large-scale commercial production?
A: Yes, the process utilizes commodity chemicals like N-bromosuccinimide and zinc acetate, operates under mild reflux conditions without cryogenic requirements, and avoids hazardous high-pressure hydrogenation, making it highly amenable to multi-kilogram and ton-scale manufacturing.
Q: What is the substrate scope for functional group tolerance on the indole ring?
A: The methodology demonstrates robust tolerance for various substituents including halogens (chloro, bromo), alkoxy groups (methoxy, ethoxy), and alkyl chains on both the aniline and phenylacetonitrile moieties, allowing for diverse derivatization prior to the final ring closure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Arylindole-3-Carbonitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceutical intermediates requires more than just a viable reaction scheme; it demands a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the laboratory bench to full-scale industrial manufacture. Our facilities are equipped with state-of-the-art rigorous QC labs capable of detecting impurities at trace levels, guaranteeing that every batch meets stringent purity specifications required for global regulatory submissions. We understand the critical nature of the N-arylindole scaffold in CNS drug development and are committed to delivering high-quality intermediates that accelerate your drug discovery and development timelines.
We invite you to engage with our technical team to explore how this zinc-mediated cyclization technology can be adapted to your specific molecular targets. By leveraging our process optimization capabilities, we can provide a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this greener, more efficient route. We encourage you to contact our technical procurement team today to request specific COA data for similar indole derivatives and to discuss route feasibility assessments tailored to your project's unique requirements. Let us collaborate to build a resilient and cost-effective supply chain for your next-generation therapeutics.
