Advanced Palladium-Catalyzed Synthesis of Carbonyl-Bridged Biheterocyclic Compounds: Enabling Commercial Scale-Up for Pharmaceutical Manufacturing
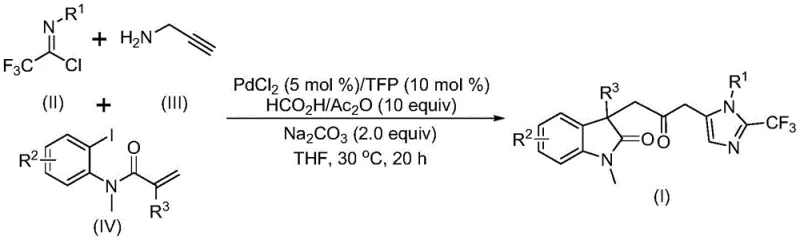
Chinese patent CN115353511A introduces a groundbreaking multi-component methodology for synthesizing carbonyl-bridged biheterocyclic compounds through a palladium-catalyzed carbonylation cascade reaction that eliminates the need for toxic carbon monoxide gas while maintaining high reaction efficiency and substrate compatibility. This innovative approach represents a significant advancement in heterocyclic chemistry by enabling the construction of complex molecular architectures through a one-pot process that combines trifluoroethylimidoyl chloride, propargylamine, and acrylamide substrates under mild conditions at 30°C. The methodology demonstrates exceptional versatility across diverse functional groups, allowing for the synthesis of various substituted biheterocyclic compounds containing trifluoromethyl and carbonyl moieties that are crucial for pharmaceutical applications. By avoiding hazardous gases and utilizing readily available starting materials, this process addresses critical safety and environmental concerns while offering substantial operational advantages for industrial scale-up. The patent specifically highlights the method's scalability to gram-level reactions, providing a clear pathway toward commercial implementation in fine chemical manufacturing. This development is particularly valuable for pharmaceutical companies seeking efficient routes to bioactive heterocyclic scaffolds that form the backbone of numerous drug molecules.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing biheterocyclic compounds typically rely on either direct coupling of two heterocycle substrates or transition metal-catalyzed tandem cyclization reactions that often require harsh reaction conditions, specialized equipment, and toxic reagents such as carbon monoxide gas, which presents significant safety hazards and necessitates expensive containment systems in manufacturing facilities. These conventional methods frequently suffer from limited substrate scope, poor functional group tolerance, and low yields due to competing side reactions or incomplete conversions, particularly when attempting to incorporate carbonyl groups into complex heterocyclic frameworks. The requirement for high temperatures or pressures in many existing protocols increases energy consumption and operational costs while reducing overall process efficiency, making large-scale production economically challenging for pharmaceutical manufacturers seeking cost-effective routes to bioactive compounds. Furthermore, the use of toxic gases like carbon monoxide creates substantial regulatory compliance burdens and requires extensive safety protocols that slow down production timelines and increase facility requirements, ultimately impacting supply chain reliability for critical pharmaceutical intermediates. Many established methodologies also lack the flexibility to accommodate diverse substitution patterns needed for structure-activity relationship studies in drug discovery programs, limiting their utility in developing novel therapeutic agents with optimized biological profiles.
The Novel Approach
The patented methodology overcomes these limitations through an innovative palladium-catalyzed carbonylation cascade reaction that utilizes formic acid/acetic anhydride as a safe carbon monoxide surrogate, eliminating the need for handling toxic CO gas while maintaining excellent reaction efficiency under mild conditions at 30°C with reaction times of 12-20 hours. This multi-component process combines trifluoroethylimidoyl chloride, propargylamine, and acrylamide substrates in a single reaction vessel using readily available palladium chloride (5 mol%) and trifuryl phosphine (10 mol%) as catalysts with sodium carbonate as base in THF solvent, creating a streamlined synthetic pathway that significantly reduces operational complexity and safety concerns. The method demonstrates exceptional substrate compatibility across a wide range of functional groups, enabling the synthesis of diverse substituted biheterocyclic compounds with trifluoromethyl groups that are valuable building blocks for pharmaceutical applications, as evidenced by the successful preparation of fifteen different derivatives with yields ranging from 40% to 92%. By employing inexpensive and commercially available starting materials along with simple workup procedures involving filtration and column chromatography, this approach offers substantial cost advantages while maintaining high product purity essential for pharmaceutical manufacturing standards. The demonstrated scalability to gram-level reactions provides a clear foundation for industrial implementation, addressing both technical feasibility and commercial viability concerns for potential adopters in the fine chemical industry.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade Reaction
The reaction mechanism begins with zero-valent palladium insertion into the carbon-iodine bond of the aryl iodide substrate followed by intramolecular Heck reaction to generate a divalent alkyl palladium intermediate, which then undergoes carbonylation using carbon monoxide released from the formic acid/acetic anhydride mixture to form an acyl palladium species that serves as a key electrophilic intermediate in the cascade process. Concurrently, base-promoted intermolecular carbon-nitrogen bond formation between trifluoroethylimidoyl chloride and propargylamine yields trifluoroacetamidine compounds that subsequently undergo isomerization to generate nucleophilic species capable of reacting with the acyl palladium intermediate. The final step involves palladium-catalyzed intramolecular cyclization where the activated trifluoroacetamidine undergoes nucleophilic attack on the acyl palladium species followed by reductive elimination to form the carbonyl-bridged biheterocyclic product with simultaneous regeneration of the palladium catalyst for subsequent catalytic cycles. This elegant cascade mechanism efficiently constructs multiple chemical bonds in a single operation while maintaining excellent regioselectivity and stereochemical control through precise coordination of the palladium catalyst with various functional groups present in the substrates.
Impurity control in this synthetic route is achieved through careful optimization of reaction parameters including catalyst loading (5 mol% PdCl₂), ligand selection (trifuryl phosphine at 10 mol%), base concentration (2.0 equivalents Na₂CO₃), and solvent choice (THF), which collectively minimize side reactions such as homocoupling or premature decomposition of sensitive intermediates. The mild reaction temperature of 30°C prevents thermal degradation pathways that could lead to impurity formation while ensuring sufficient reactivity for the multi-step cascade process to proceed efficiently. Substrate design plays a crucial role in impurity management, as the specific combination of trifluoroethylimidoyl chloride with propargylamine creates a well-defined reaction trajectory that avoids common side products associated with alternative coupling methodologies. The workup procedure involving filtration followed by silica gel mixing and column chromatography provides an additional purification step that effectively removes residual catalysts, ligands, and minor byproducts, resulting in high-purity final products suitable for pharmaceutical applications as confirmed by HRMS and NMR characterization data presented in the patent examples.
How to Synthesize Carbonyl-Bridged Biheterocyclic Compounds Efficiently
This patented methodology represents a significant advancement in heterocyclic synthesis by providing a safe, efficient route to valuable carbonyl-bridged biheterocyclic compounds through a carefully designed palladium-catalyzed cascade reaction that eliminates hazardous reagents while maintaining excellent yields and substrate flexibility. The process leverages readily available starting materials including trifluoroethylimidoyl chloride, propargylamine, and acrylamide in combination with an optimized catalytic system featuring palladium chloride and trifuryl phosphine under mild thermal conditions to construct complex molecular architectures in a single operation. Detailed standardized synthesis procedures have been developed based on extensive experimental optimization documented in the patent, ensuring consistent product quality and process reliability across different production scales. The following step-by-step guide outlines the precise methodology for implementing this innovative synthetic route in laboratory or manufacturing settings.
- Prepare the reaction mixture by adding palladium chloride (5 mol%), trifuryl phosphine (10 mol%), sodium carbonate (2.0 equiv), and formic acid/acetic anhydride mixture (10 equiv) to THF solvent under inert atmosphere.
- Introduce trifluoroethylimidoyl chloride, propargylamine, and acrylamide substrates into the reaction system with precise stoichiometric ratios to ensure optimal conversion.
- Conduct the reaction at controlled temperature of 30°C for 16-20 hours followed by standard workup including filtration, silica gel mixing, and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthetic methodology addresses critical pain points in pharmaceutical intermediate manufacturing by providing a safer, more efficient route to complex heterocyclic compounds that are essential building blocks for numerous drug development programs. The elimination of toxic carbon monoxide gas from the synthesis process significantly reduces safety risks and regulatory compliance burdens while lowering capital investment requirements for specialized containment equipment, making this approach particularly attractive for manufacturers seeking to improve operational efficiency without compromising product quality or worker safety.
- Cost Reduction in Manufacturing: The use of inexpensive commercial starting materials combined with simplified processing requirements eliminates expensive reagent costs associated with traditional carbonylation methods while reducing capital expenditure through avoidance of specialized gas handling infrastructure required when using toxic carbon monoxide gas directly.
- Enhanced Supply Chain Reliability: Reliance on widely available raw materials ensures consistent access to starting components while minimizing vulnerability to supply chain disruptions that often affect specialized chemical reagents requiring complex logistics networks or restricted distribution channels.
- Scalability and Environmental Compliance: The demonstrated scalability from laboratory to gram-scale production provides clear commercial implementation pathways while meeting increasingly stringent environmental regulations through inherently safer chemistry principles that eliminate hazardous gas usage without requiring additional end-of-pipe treatment systems.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial concerns regarding implementation of this patented synthetic methodology based on detailed analysis of technical specifications and experimental results documented in CN115353511A patent documentation.
Q: How does this novel method eliminate the need for toxic carbon monoxide gas while maintaining high reaction efficiency?
A: The patented process utilizes formic acid/acetic anhydride as an in situ carbon monoxide surrogate that safely generates CO under mild reaction conditions without requiring specialized gas handling equipment or containment systems typically needed for gaseous CO.
Q: What are the key advantages of using readily available starting materials in this synthesis?
A: The methodology employs commercially accessible substrates including propargylamine and acrylamide which demonstrate excellent supply chain stability while eliminating dependency on specialized or hazardous reagents that often create procurement bottlenecks in pharmaceutical manufacturing.
Q: How does the catalytic system ensure excellent substrate compatibility across diverse functional groups?
A: The optimized palladium/trifuryl phosphine catalyst system maintains high activity across various substitution patterns while preventing common side reactions through precise control of reaction parameters including temperature, concentration, and stoichiometry.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocyclic Compounds Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications required by global pharmaceutical standards through our state-of-the-art manufacturing facilities equipped with rigorous QC labs capable of comprehensive analytical characterization. As a leading CDMO specializing in complex heterocyclic chemistry, we have successfully implemented similar multi-component catalytic processes across various therapeutic areas, demonstrating our capability to translate innovative academic discoveries into robust industrial manufacturing solutions that meet the highest quality requirements of global pharmaceutical clients.
We invite you to contact our technical procurement team to request specific COA data and route feasibility assessments for your particular application needs through our Customized Cost-Saving Analysis service that evaluates potential efficiency improvements tailored to your specific manufacturing requirements.
