Pioneering Palladium-Catalyzed Synthesis of Carbonyl-Bridged Biheterocyclic Compounds for Scalable Pharmaceutical Intermediate Production
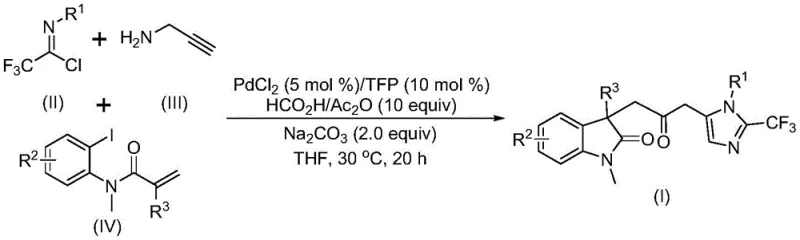
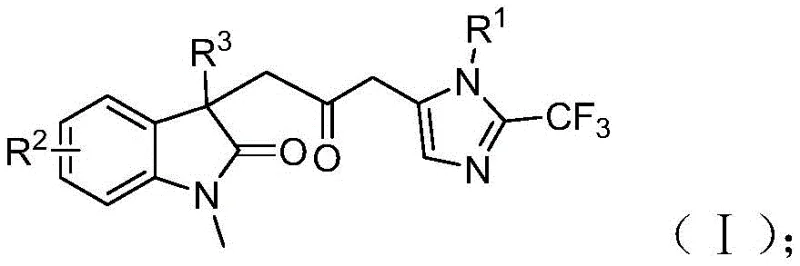
Patent CN115353511A, published on November 18, 2022, introduces a groundbreaking multi-component synthesis method for carbonyl-bridged biheterocyclic compounds, representing a significant advancement in the field of organic synthesis for pharmaceutical intermediates. This innovative approach eliminates the need for toxic carbon monoxide gas while utilizing readily available starting materials, including trifluoroethylimidoyl chloride, propargylamine, and acrylamide, under mild reaction conditions at 30°C. The method demonstrates exceptional substrate compatibility and functional group tolerance, enabling the synthesis of diverse substituted biheterocyclic compounds with trifluoromethyl and carbonyl moieties through strategic substrate design. With its simple operational procedure and scalability to gram-level reactions, this patented technology offers substantial potential for industrial implementation in the production of complex pharmaceutical intermediates. The elimination of hazardous gases and use of cost-effective catalysts position this methodology as a sustainable and economically viable solution for modern pharmaceutical manufacturing processes, addressing critical challenges faced by R&D teams in developing efficient synthetic routes for bioactive molecules.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing biheterocyclic compounds have been plagued by significant limitations that hinder their practical application in industrial settings. Conventional methods often require harsh reaction conditions, including high temperatures and pressures, which increase operational complexity and safety concerns, particularly when handling toxic gases like carbon monoxide in carbonylation reactions. Many existing protocols suffer from low substrate compatibility, with narrow functional group tolerance that restricts the diversity of accessible molecular structures, thereby limiting their utility in pharmaceutical development where structural variation is crucial for optimizing biological activity. The multi-step nature of many conventional syntheses leads to reduced overall yields and increased production costs due to multiple purification steps and intermediate isolations. Furthermore, the reliance on expensive transition metal catalysts without efficient recovery systems creates economic barriers for large-scale implementation, while the generation of hazardous byproducts complicates waste management and increases environmental compliance costs. These limitations collectively result in extended development timelines, higher production costs, and reduced flexibility in meeting the evolving demands of pharmaceutical research and development teams seeking diverse compound libraries for structure-activity relationship studies.
The Novel Approach
The patented methodology described in CN115353511A presents a transformative solution to these longstanding challenges through an elegant palladium-catalyzed multi-component reaction that operates under remarkably mild conditions at 30°C without requiring toxic carbon monoxide gas. By utilizing a carefully designed combination of palladium chloride (5 mol%), trifuryl phosphine (10 mol%), and a formic acid/acetic anhydride mixture as a safe CO surrogate, this approach achieves high efficiency while maintaining excellent functional group tolerance across diverse substrates. The reaction demonstrates exceptional versatility through strategic substrate design, enabling the synthesis of various substituted biheterocyclic compounds with trifluoromethyl and carbonyl functionalities that are highly valuable in pharmaceutical applications. The simplified operational procedure eliminates complex gas handling systems, significantly enhancing workplace safety while reducing capital investment requirements for manufacturing facilities. With reaction times ranging from 12 to 20 hours and compatibility with common organic solvents like THF, this method offers practical advantages for process chemists seeking scalable solutions that balance efficiency, safety, and cost-effectiveness in intermediate production.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade
The reaction mechanism involves a sophisticated cascade process that begins with zero-valent palladium insertion into the carbon-iodine bond of the iodoaniline derivative, followed by an intramolecular Heck reaction that generates a divalent alkyl palladium intermediate. This key intermediate then undergoes carbonylation through carbon monoxide released from the formic acid/acetic anhydride mixture, forming an acyl palladium species that serves as the crucial electrophile for subsequent transformations. Simultaneously, the base-promoted reaction between trifluoroethylimidoyl chloride and propargylamine forms a trifluoroacetamidine intermediate through intermolecular carbon-nitrogen bond formation, which subsequently undergoes isomerization to generate the nucleophilic species required for the final cyclization step. The acyl palladium intermediate activates the trifluoroacetamidine compound, facilitating an intramolecular cyclization that ultimately yields the target carbonyl-bridged biheterocyclic structure with high regioselectivity and efficiency. This elegant mechanistic pathway demonstrates how careful catalyst selection and reaction design can overcome traditional limitations in carbonylation chemistry while maintaining excellent control over product formation.
The method exhibits exceptional control over impurity profiles through its well-defined reaction pathway and mild operating conditions, which minimize side reactions commonly associated with harsher synthetic approaches. The use of sodium carbonate as a base promotes clean conversion while preventing unwanted decomposition pathways that could lead to impurity formation. The compatibility with various functional groups across different substrates (as demonstrated in the patent examples) indicates robustness against potential side reactions that might generate impurities in more sensitive systems. The chromatographic purification process described (involving filtration, silica gel mixing, and column chromatography) provides an effective means to achieve high purity levels required for pharmaceutical applications, with the patent demonstrating consistent production of compounds meeting stringent quality specifications as evidenced by comprehensive NMR and HRMS characterization data. This level of impurity control is particularly valuable for pharmaceutical intermediates where regulatory requirements demand rigorous quality standards throughout the manufacturing process.
How to Synthesize Carbonyl-Bridged Biheterocyclic Compounds Efficiently
This innovative synthetic route represents a significant advancement in the preparation of complex biheterocyclic structures essential for pharmaceutical development. The patented methodology offers a streamlined approach that eliminates hazardous gas handling while maintaining high efficiency and substrate flexibility. Detailed standardized synthesis procedures have been developed based on the patent specifications, with optimized parameters for catalyst loading, solvent selection, and reaction timing to ensure consistent product quality at scale. The following section provides a step-by-step guide for implementing this technology in laboratory or production settings, with specific attention to critical process parameters that influence yield and purity outcomes.
- Combine palladium chloride catalyst (5 mol%), trifuryl phosphine ligand (10 mol%), sodium carbonate base (2.0 equiv), formic acid/acetic anhydride mixture (10 equiv), trifluoroethylimidoyl chloride, propargylamine, acrylamide derivative, and THF solvent in a reaction vessel under inert atmosphere
- Stir the reaction mixture at precisely 30°C for 16-20 hours while monitoring conversion through standard analytical techniques to ensure complete transformation without over-reaction
- Perform post-reaction processing including filtration through silica gel followed by column chromatography purification using appropriate eluent systems to isolate the target carbonyl-bridged biheterocyclic compound with high purity
Commercial Advantages for Procurement and Supply Chain Teams
This novel synthetic methodology addresses critical pain points in pharmaceutical intermediate supply chains by offering a more sustainable and economically viable production pathway that aligns with modern manufacturing requirements. The elimination of toxic carbon monoxide gas handling reduces safety infrastructure costs while improving workplace conditions, making this approach particularly attractive for manufacturers seeking to enhance their environmental, social, and governance (ESG) profiles without compromising production efficiency. The use of readily available starting materials from established chemical suppliers ensures reliable sourcing with minimal supply chain disruption risk, while the simplified reaction protocol reduces equipment complexity and maintenance requirements compared to traditional high-pressure carbonylation processes.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and specialized high-pressure equipment significantly reduces capital expenditure requirements for manufacturing facilities. By utilizing safe CO surrogates instead of toxic carbon monoxide gas, the process avoids costly gas handling infrastructure and associated safety systems while maintaining high reaction efficiency. The simplified operational procedure reduces labor costs through shorter training requirements and fewer specialized personnel needs, while the excellent substrate compatibility minimizes raw material waste through higher conversion rates and reduced need for extensive purification steps.
- Enhanced Supply Chain Reliability: The reliance on commercially available starting materials with established global supply chains ensures consistent access to key reagents without dependency on specialized or single-source suppliers. The robust nature of the reaction across diverse substrates provides flexibility to adapt to potential supply constraints by substituting alternative feedstocks while maintaining product quality. The mild reaction conditions (30°C) reduce energy consumption compared to high-temperature processes, making production less vulnerable to energy price fluctuations while improving overall process stability and consistency across different manufacturing sites.
- Scalability and Environmental Compliance: The demonstrated scalability from laboratory to gram-scale reactions provides confidence in the method's potential for commercial implementation without requiring significant process re-engineering. The elimination of hazardous gases reduces environmental compliance costs while improving worker safety profiles, aligning with increasingly stringent global regulations on chemical manufacturing processes. The simplified waste stream resulting from fewer side reactions and byproducts lowers disposal costs and environmental impact, supporting sustainable manufacturing initiatives that are becoming increasingly important in pharmaceutical supply chain decisions.
Frequently Asked Questions (FAQ)
The following questions address common concerns from procurement and technical teams regarding implementation of this patented technology for pharmaceutical intermediate production. Each response is grounded in the specific technical details provided in patent CN115353511A, ensuring accurate information for decision-making purposes regarding process adoption and scale-up considerations.
Q: How does this method eliminate toxic carbon monoxide gas while maintaining high reaction efficiency?
A: The patented process utilizes a formic acid/acetic anhydride mixture as a safe carbon monoxide surrogate that releases CO in situ under mild conditions (30°C), eliminating the need for hazardous gas handling systems while maintaining excellent reaction efficiency through controlled release kinetics.
Q: What are the key advantages of this methodology compared to conventional biheterocyclic compound synthesis approaches?
A: This method offers superior substrate compatibility across diverse functional groups, operates under mild temperature conditions without high-pressure equipment requirements, uses cost-effective catalysts with simple workup procedures, and demonstrates excellent scalability from laboratory to commercial production volumes.
Q: How does the substrate flexibility benefit pharmaceutical development programs?
A: The broad functional group tolerance allows medicinal chemists to rapidly generate diverse compound libraries through strategic substrate design by varying R groups on trifluoroethylimidoyl chloride, iodoaniline derivatives, and acrylamide components while maintaining consistent reaction performance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocyclic Compounds Supplier
NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring seamless transition from laboratory development to full-scale manufacturing of these complex biheterocyclic intermediates. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required for pharmaceutical applications, with comprehensive analytical capabilities to verify product quality at every stage of production. By leveraging our deep expertise in palladium-catalyzed transformations and multi-component reactions, we provide reliable supply solutions that address both current manufacturing needs and future scalability requirements for our global pharmaceutical partners.
For organizations seeking to optimize their intermediate supply chain while maintaining high quality standards, we invite you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your requirements. Take advantage of our Customized Cost-Saving Analysis service to evaluate how this innovative synthetic route can enhance your production economics while ensuring reliable supply of these critical pharmaceutical intermediates.
