Advanced Copper-Catalyzed Oxidative Cyclization for High-Purity Indole Ketone Manufacturing
Advanced Copper-Catalyzed Oxidative Cyclization for High-Purity Indole Ketone Manufacturing
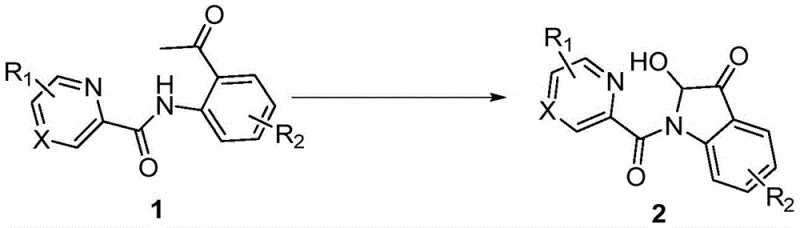
The pharmaceutical industry continuously seeks robust and atom-economical pathways to access complex heterocyclic scaffolds, particularly those found in bioactive natural products and drug candidates. Patent CN113278007A introduces a significant breakthrough in the synthesis of 2-hydroxy-indole-3-ketone compounds, a privileged structural motif prevalent in alkaloids such as notoamide O and brevianamide A. This technology leverages a copper-catalyzed aerobic oxidative cyclization strategy, transforming readily available N-(2-acetylphenyl)picolinamide derivatives into valuable indole ketones in a single operational step. By utilizing molecular oxygen as the terminal oxidant and inexpensive copper salts as catalysts, this method addresses critical pain points in modern medicinal chemistry, including step economy and environmental sustainability. For R&D teams focused on rapid library synthesis and process chemists aiming for scalable routes, this patent offers a compelling alternative to legacy methodologies that often suffer from low yields or harsh reaction conditions.
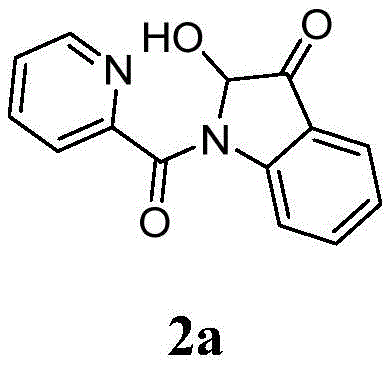
The core innovation lies in the efficient construction of the quaternary carbon center at the C2 position of the indole ring, a challenging transformation that typically requires multi-step sequences. The disclosed method achieves this through a pyridine-directed C-H activation mechanism, where the picolinamide moiety acts as a bidentate directing group to facilitate regioselective cyclization. This approach not only streamlines the synthetic route but also enhances the overall atom economy by incorporating oxygen directly from the air into the final product structure. As a reliable pharmaceutical intermediate supplier, understanding such mechanistic nuances is crucial for optimizing process parameters and ensuring consistent quality in commercial batches. The versatility of this reaction is further evidenced by its tolerance to a wide range of functional groups, including halogens, alkyl chains, and electron-withdrawing substituents, making it a versatile tool for diversifying chemical libraries.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-hydroxy-indol-3-ones has been plagued by significant technical hurdles that hinder their widespread application in drug discovery. Early reports, such as those by the Foote group, relied on the oxidation of 2-methyl-1H-indoles using dimethyl dioxirane (DMD), a reagent that is not only unstable and difficult to handle on scale but also resulted in dismal yields of less than 10%. Such low efficiency renders the process economically unviable for commercial manufacturing, as the cost of goods would be prohibitively high due to massive material loss. Furthermore, alternative strategies involving Grignard reagents, as described by the Dash group, necessitate the use of pre-functionalized 3-hydroxy-indol-2-one starting materials. This requirement adds extra synthetic steps upstream, increasing the cumulative cost and complexity while introducing sensitivity to moisture and air that complicates reactor operations. The reliance on stoichiometric oxidants or cryogenic conditions in these traditional methods also generates substantial chemical waste, posing challenges for environmental compliance and waste treatment in large-scale facilities.
The Novel Approach
In stark contrast, the methodology outlined in CN113278007A represents a paradigm shift towards greener and more efficient synthesis. By employing N-(2-acetylphenyl)picolinamide as the starting material, the process bypasses the need for pre-oxidized indole precursors, effectively collapsing multiple synthetic steps into a single pot. The use of cuprous iodide (CuI) as a catalyst, combined with simple carboxylic acid additives like acetic acid, creates a mild yet effective catalytic system that operates under an oxygen atmosphere at moderate temperatures of 80-110°C. This transition from stoichiometric reagents to catalytic turnover significantly reduces the raw material burden and simplifies the workup procedure, as the byproducts are minimal and easily separable. The reaction demonstrates excellent substrate scope, successfully converting various substituted anilines into the corresponding indole ketones with yields often exceeding 70%, as seen in examples like 2d (78%) and 2q (80%). This robustness makes it an ideal candidate for cost reduction in API manufacturing, where yield and operational simplicity are paramount drivers of profitability.
Mechanistic Insights into Cu-Catalyzed Aerobic Oxidative Cyclization
The success of this transformation hinges on a sophisticated interplay between the copper catalyst, the directing group, and molecular oxygen. Mechanistically, the reaction is believed to initiate with the coordination of the copper species to the nitrogen atoms of the picolinamide moiety, forming a stable chelate that positions the metal center in proximity to the ortho-acetyl group. This pyridine-directed C-H activation facilitates the formation of a cyclic organocopper intermediate, which is then susceptible to oxidation by molecular oxygen. The oxygen molecule serves a dual role: it regenerates the active copper catalyst from its reduced state and provides the oxygen atom necessary for the formation of the hydroxyl group at the C2 position. This catalytic cycle ensures that only catalytic amounts of copper are required, minimizing metal contamination in the final product, a critical factor for pharmaceutical intermediates destined for clinical use. The presence of acetic acid as an additive likely assists in proton transfer steps and stabilizes the transition states, thereby lowering the activation energy barrier and allowing the reaction to proceed smoothly at 100°C.
From an impurity control perspective, the mild oxidative conditions offer distinct advantages over aggressive oxidants that often lead to over-oxidation or ring degradation. The selectivity of the copper-catalyzed system ensures that the oxidation is confined to the specific C-H bond activated by the directing group, preserving other sensitive functionalities on the aromatic rings. This high chemoselectivity results in a cleaner reaction profile with fewer side products, which simplifies the downstream purification process and improves the overall recovery of the target material. For quality control teams, this means that meeting stringent purity specifications becomes more achievable without resorting to extensive recrystallization or preparative HPLC. Furthermore, the avoidance of heavy metal oxidants like chromium or manganese salts eliminates the risk of toxic metal residues, aligning the process with modern regulatory guidelines for elemental impurities in drug substances. The ability to tune the electronic properties of the substrate through R1 and R2 substituents allows for fine-tuning of the reaction kinetics, providing process chemists with additional levers to optimize conversion and minimize impurity formation.
How to Synthesize 2-Hydroxy-Indole-3-Ketone Efficiently
To implement this synthesis effectively, precise control over reaction parameters is essential to maximize yield and reproducibility. The patent details a standardized protocol where the substrate, catalyst, and additive are dissolved in a polar aprotic solvent such as DMSO or DMF, creating a homogeneous reaction mixture that ensures efficient heat and mass transfer. The detailed standardized synthesis steps see the guide below for specific operational instructions regarding temperature ramping and oxygen flow rates.
- Charge a reaction vessel with N-(2-acetylphenyl)picolinamide substrate, cuprous iodide catalyst (25-40 mol%), and acetic acid additive in DMSO solvent.
- Evacuate the system and backfill with oxygen gas to establish an oxidative atmosphere, then heat the mixture to 80-110°C with magnetic stirring.
- Monitor reaction progress via TLC until completion, then cool, extract with ethyl acetate, wash, dry, and purify by column chromatography to isolate the target ketone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this copper-catalyzed technology translates into tangible strategic benefits that extend beyond mere chemical efficiency. The shift from expensive, specialized reagents to commodity chemicals like cuprous iodide and acetic acid drastically simplifies the sourcing landscape, reducing dependency on single-source suppliers for exotic catalysts. This democratization of raw materials enhances supply chain resilience, ensuring that production schedules are not disrupted by shortages of critical reagents. Moreover, the elimination of cryogenic conditions and the use of ambient pressure oxygen remove the need for specialized low-temperature reactors or high-pressure autoclaves, allowing the process to be run in standard glass-lined steel reactors commonly available in multipurpose manufacturing plants. This compatibility with existing infrastructure accelerates technology transfer and reduces capital expenditure requirements for scale-up.
- Cost Reduction in Manufacturing: The economic impact of this method is profound, primarily driven by the replacement of stoichiometric oxidants with molecular oxygen, which is essentially free and abundant. By avoiding the purchase of costly reagents like dimethyl dioxirane or Grignard reagents, the direct material costs are significantly lowered. Additionally, the high atom economy of the reaction means that a larger proportion of the starting material ends up in the final product, reducing waste disposal costs and improving the overall mass balance. The simplified workup procedure, which involves standard extraction and chromatography rather than complex quenching and neutralization steps, further reduces labor and utility consumption, contributing to substantial cost savings in the overall manufacturing budget.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to a more predictable and reliable supply chain. Since the reaction tolerates a variety of functional groups and proceeds with good yields across different substrates, the risk of batch failure due to substrate variability is minimized. This consistency allows for more accurate demand forecasting and inventory planning. Furthermore, the use of stable and non-hazardous reagents simplifies logistics and storage requirements, reducing the regulatory burden associated with transporting dangerous goods. This ease of handling ensures that raw materials can be sourced globally without significant logistical bottlenecks, securing the continuity of supply for downstream API production.
- Scalability and Environmental Compliance: From an environmental standpoint, this process aligns perfectly with green chemistry principles, which is increasingly important for maintaining social license to operate. The use of oxygen as the oxidant produces water as the primary byproduct, eliminating the generation of heavy metal waste streams associated with traditional oxidants. This reduction in hazardous waste simplifies effluent treatment and lowers the environmental footprint of the manufacturing site. The mild reaction temperatures and atmospheric pressure conditions also reduce energy consumption compared to high-temperature or high-pressure processes. These factors collectively make the process highly scalable, enabling the seamless transition from kilogram-scale laboratory synthesis to ton-scale commercial production without encountering the safety and engineering challenges typical of more hazardous chemistries.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical disclosures within the patent documentation to provide clarity on feasibility and performance.
Q: What are the key advantages of this copper-catalyzed method over traditional Grignard routes?
A: Unlike traditional methods requiring harsh Grignard reagents and pre-functionalized 3-hydroxy-indol-2-one starting materials, this patent describes a direct oxidative cyclization using molecular oxygen. This eliminates the need for cryogenic conditions and expensive stoichiometric oxidants, significantly simplifying the operational complexity and reducing waste generation.
Q: What is the typical yield and purity profile for these indole ketone derivatives?
A: The patent data demonstrates robust yields ranging from 51% to 80% across various substituted substrates, including halogenated and alkylated derivatives. The use of mild acidic additives and controlled temperatures helps minimize side reactions, resulting in a cleaner crude profile that facilitates easier downstream purification.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the protocol utilizes inexpensive copper salts and common organic solvents like DMSO or DMF under atmospheric oxygen pressure, avoiding high-pressure hydrogenation or exotic catalysts. These factors, combined with the one-pot nature of the reaction, make it highly amenable to scale-up for industrial pharmaceutical intermediate manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Hydroxy-Indole-3-Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of novel therapeutics. Our team of expert process chemists has extensively evaluated the copper-catalyzed oxidative cyclization route described in CN113278007A and confirmed its potential for robust commercial manufacturing. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from clinical trials to market launch. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-hydroxy-indole-3-ketone delivered meets the highest standards of quality and consistency required by global regulatory agencies.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next drug development program. By partnering with our technical procurement team, you can receive a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise in C-H activation chemistry can accelerate your pipeline and optimize your supply chain economics.
