Advanced Copper-Catalyzed Synthesis of 2-Hydroxy-Indole-3-Ketone Pharmaceutical Intermediates for Commercial Scale-Up
Advanced Copper-Catalyzed Synthesis of 2-Hydroxy-Indole-3-Ketone Pharmaceutical Intermediates for Commercial Scale-Up
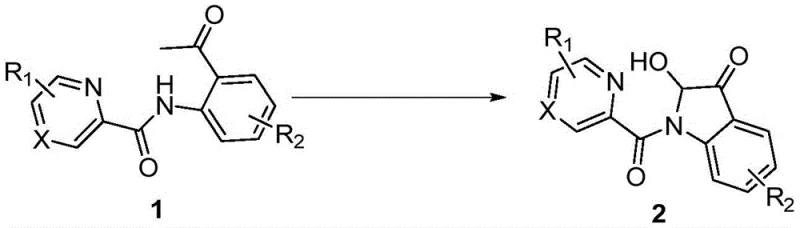
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and economically viable pathways for constructing complex heterocyclic scaffolds that serve as critical building blocks for drug discovery. A significant breakthrough in this domain is detailed in Chinese Patent CN113278007A, which discloses a highly efficient synthetic method for 2-hydroxy-indole-3-ketone compounds. These privileged structures are ubiquitous in natural products and biologically active molecules, ranging from alkaloids like notoamide O to potential anticancer agents. The patent outlines a transformative approach utilizing N-(2-acetylphenyl)picolinamide derivatives as starting materials, employing copper salts such as cuprous iodide as catalysts, and leveraging molecular oxygen as the terminal oxidant. This methodology represents a paradigm shift from traditional, labor-intensive syntheses, offering a streamlined one-step protocol that operates under relatively mild thermal conditions between 80°C and 110°C. For R&D directors and procurement specialists alike, this innovation promises not only enhanced chemical efficiency but also substantial reductions in raw material costs and waste generation, positioning it as a cornerstone technology for the next generation of indole-based API intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 2-hydroxy-indol-3-one skeleton, particularly those possessing a quaternary carbon center at the C2 position, has presented formidable challenges to organic chemists. Early methodologies, such as those proposed by the Foote group, relied heavily on the use of dimethyl dioxirane (DMD) as a potent oxidant to convert 2-methyl-1H-indoles. However, this approach suffered from critically low yields, often falling below 10%, rendering it practically useless for any meaningful commercial application or extensive biological screening. Furthermore, alternative strategies reported by groups such as Dash necessitated the use of 3-hydroxy-indol-2-one as a specialized starting material, which itself requires multi-step preparation, thereby increasing the overall step count and cost. These conventional routes frequently demanded the use of sensitive Grignard reagents, which impose severe constraints on reaction conditions, requiring strict anhydrous environments and cryogenic temperatures that are difficult and expensive to maintain on a large industrial scale. The reliance on such hazardous reagents and the poor atom economy of these legacy processes create significant bottlenecks in the supply chain, leading to high production costs and inconsistent batch-to-batch quality.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the novel strategy disclosed in patent CN113278007A introduces a direct, oxidative cyclization pathway that bypasses the need for pre-functionalized substrates or exotic oxidants. By utilizing readily available N-(2-acetylphenyl)picolinamide derivatives, the process achieves ring closure through a copper-catalyzed mechanism driven by atmospheric oxygen. This approach eliminates the necessity for preparing unstable intermediates like 3-hydroxy-indol-2-one, effectively collapsing multiple synthetic steps into a single, high-yielding transformation. The reaction conditions are remarkably benign, proceeding efficiently in common polar aprotic solvents such as dimethyl sulfoxide (DMSO) or N,N-dimethylformamide (DMF) at temperatures around 100°C. This simplicity translates directly into operational ease, allowing for safer handling and reduced energy consumption compared to cryogenic Grignard reactions. Moreover, the use of inexpensive copper salts and acetic acid additives ensures that the reagent costs remain minimal, addressing a primary concern for procurement managers focused on cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Copper-Catalyzed Oxidative Cyclization
The efficacy of this synthetic route lies in its elegant mechanistic design, which leverages the coordinating ability of the pyridine moiety to direct the copper catalyst towards the specific activation of the acetyl group. The proposed mechanism involves the initial chelation of the copper species by the nitrogen atom of the picolinamide group, forming a stable metallacycle intermediate that positions the metal center in close proximity to the reactive acetyl functionality. Under an oxygen atmosphere, the copper catalyst facilitates the activation of molecular oxygen, generating reactive oxygen species that promote the oxidative cleavage and subsequent cyclization required to form the indole-3-one core. This pyridine-directed C-H activation and functionalization strategy ensures high regioselectivity, minimizing the formation of unwanted byproducts and simplifying the downstream purification process. The involvement of acetic acid as an additive likely plays a crucial role in proton transfer steps and stabilizing the catalytic cycle, further enhancing the turnover number of the copper catalyst. For technical teams, understanding this mechanism highlights the robustness of the process, as the directing group strategy is known to tolerate a wide array of electronic and steric environments on the aromatic rings.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based oxidations that often lead to complex mixtures of over-oxidized products. The controlled nature of the copper-oxygen interaction allows for the selective formation of the hydroxyl group at the C2 position without compromising the integrity of the ketone at the C3 position. The patent data indicates that various substituents, including electron-donating alkyl groups and electron-withdrawing halogens, are well-tolerated, suggesting that the electronic properties of the substrate do not drastically alter the fundamental catalytic cycle. This broad substrate compatibility is critical for medicinal chemists who need to rapidly generate libraries of analogs for structure-activity relationship (SAR) studies. The high yields reported across diverse examples, ranging from simple phenyl derivatives to more complex heterocyclic fused systems, underscore the reliability of this mechanistic pathway. Consequently, the risk of batch failure due to substrate sensitivity is significantly mitigated, ensuring a consistent supply of high-purity intermediates for downstream drug synthesis.
How to Synthesize 2-Hydroxy-Indole-3-Ketone Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and purity. The general procedure involves charging a reaction vessel with the N-(2-acetylphenyl)picolinamide substrate, a catalytic amount of cuprous iodide, and an excess of acetic acid in a solvent like DMSO. The system must be purged and maintained under an oxygen atmosphere to drive the oxidative cyclization to completion. While the patent specifies a temperature range of 80-110°C, optimization data suggests that maintaining the reaction at 100°C for approximately 4 to 6 hours provides an optimal balance between reaction rate and product stability. Following the reaction, standard workup procedures involving extraction with ethyl acetate and purification via column chromatography are employed to isolate the target 2-hydroxy-indole-3-ketone. The detailed standardized synthesis steps, including precise stoichiometric ratios and safety precautions for scaling, are outlined in the guide below.
- Prepare the reaction mixture by combining N-(2-acetylphenyl)picolinamide substrate, cuprous iodide catalyst (25-40 mol%), and acetic acid additive in a polar aprotic solvent like DMSO.
- Establish an oxygen atmosphere within the reaction vessel and heat the mixture to a temperature range of 80-110°C, preferably maintaining 100°C for optimal conversion.
- Monitor reaction progress via TLC, then proceed to workup involving ethyl acetate extraction, saline washing, drying, and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this copper-catalyzed methodology offers compelling economic and logistical benefits that extend far beyond simple yield improvements. The transition from expensive, hazardous reagents like Grignard reagents or DMD to commodity chemicals such as cuprous iodide and acetic acid results in a drastic simplification of the raw material portfolio. This shift not only lowers the direct cost of goods sold (COGS) but also reduces the regulatory burden associated with storing and handling pyrophoric or highly explosive materials. Furthermore, the elimination of multi-step precursor synthesis means that the lead time for producing the final intermediate is significantly shortened, allowing for more responsive inventory management and faster time-to-market for new drug candidates. The use of oxygen as the oxidant is particularly advantageous from a green chemistry standpoint, as it generates water as the primary byproduct, thereby minimizing the volume of toxic waste that requires costly disposal. These factors collectively contribute to a more resilient and cost-effective supply chain capable of supporting long-term commercial production.
- Cost Reduction in Manufacturing: The replacement of noble metal catalysts or stoichiometric oxidants with inexpensive copper salts and molecular oxygen fundamentally alters the cost structure of the synthesis. By avoiding the need for specialized, pre-functionalized starting materials, the process reduces the number of unit operations required, which in turn lowers labor, energy, and equipment depreciation costs. The high atom economy of the reaction ensures that a greater proportion of the input mass is converted into the desired product, minimizing waste and maximizing resource efficiency. Additionally, the mild reaction conditions reduce the energy load on heating and cooling systems, contributing to further operational savings. These cumulative effects result in substantial cost savings that can be passed down the supply chain, making the final API more competitive in the global market.
- Enhanced Supply Chain Reliability: The reliance on widely available, commodity-grade reagents ensures that the supply chain is not vulnerable to the shortages or price volatility often associated with specialized fine chemicals. Cuprous iodide and acetic acid are produced on a massive industrial scale, guaranteeing a steady and secure supply for continuous manufacturing operations. The robustness of the reaction against varying substrate electronics means that suppliers can source a wider range of starting materials without risking process failure, adding flexibility to the procurement strategy. This reliability is crucial for maintaining uninterrupted production schedules, especially when scaling up to meet the demands of clinical trials or commercial launch. The simplified process flow also reduces the number of potential failure points, enhancing the overall dependability of the manufacturing line.
- Scalability and Environmental Compliance: The operational simplicity of running the reaction at atmospheric pressure and moderate temperatures makes this process inherently scalable from gram to ton quantities without the need for specialized high-pressure reactors. The use of DMSO or DMF as solvents is compatible with existing recovery and recycling infrastructure in most chemical plants, facilitating solvent reuse and reducing environmental impact. The generation of minimal hazardous waste aligns with increasingly stringent environmental regulations, reducing the risk of compliance issues and associated fines. The ability to scale this process efficiently allows manufacturers to respond quickly to market demand surges, ensuring that the supply of critical pharmaceutical intermediates remains stable and secure. This scalability is a key enabler for the commercial success of drugs relying on this indole scaffold.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their specific projects. The responses cover aspects ranging from substrate tolerance to operational safety, ensuring a comprehensive overview of the technology's value proposition.
Q: What are the primary advantages of this copper-catalyzed method over traditional synthesis routes?
A: Unlike traditional methods requiring harsh oxidants like dimethyl dioxirane or sensitive Grignard reagents, this protocol utilizes molecular oxygen as a green oxidant and inexpensive copper salts, significantly improving atom economy and operational safety.
Q: What is the substrate scope for this oxidative cyclization reaction?
A: The method demonstrates excellent substrate applicability, tolerating various substituents including alkyl groups, halogens (chloro, bromo, iodo), and different heterocyclic systems such as pyridine, pyrazine, and quinoline derivatives.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the reaction operates under mild conditions (80-110°C) using commercially available solvents and catalysts, avoiding cryogenic temperatures or ultra-high pressures, which facilitates straightforward commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Hydroxy-Indole-3-Ketone Supplier
As the demand for complex heterocyclic intermediates continues to grow in the pharmaceutical sector, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM is essential for navigating the complexities of process development and scale-up. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the laboratory bench to full-scale manufacturing. We are committed to delivering high-purity 2-hydroxy-indole-3-ketone derivatives that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our expertise in copper-catalyzed transformations allows us to optimize this specific patent-protected route for maximum efficiency and cost-effectiveness, tailored to your unique supply chain requirements.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis can benefit your pipeline. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic advantages of switching to this copper-catalyzed method. We encourage you to contact us today to obtain specific COA data for our reference standards and to receive detailed route feasibility assessments for your target molecules. Let us help you secure a reliable, cost-effective, and sustainable supply of these critical pharmaceutical intermediates.
