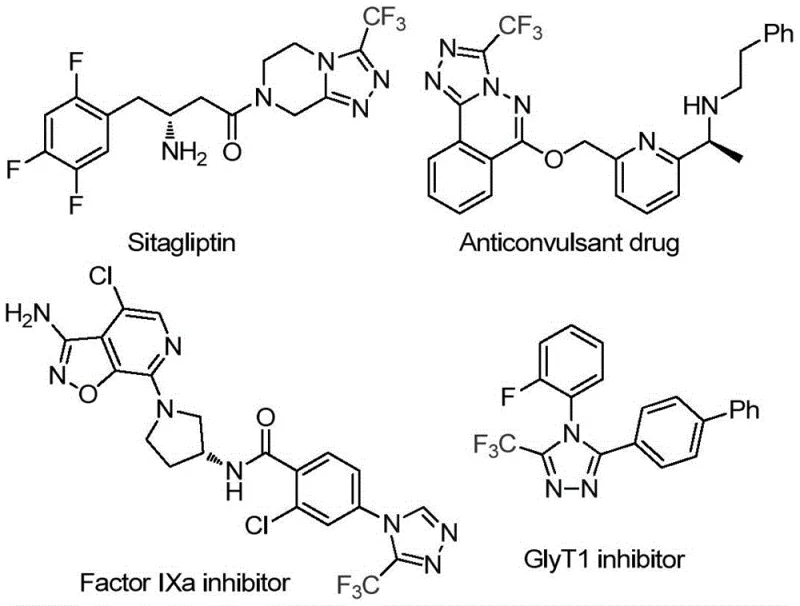
Advanced CF3-Triazole Synthesis: Scalable Green Chemistry for High-Purity Pharmaceutical Intermediates
Patent CN113880781A introduces a groundbreaking method for synthesizing 3-trifluoromethyl-substituted 1,2,4-triazole compounds using glucose as a sustainable carbon source under remarkably mild conditions. This innovation addresses critical challenges in pharmaceutical intermediate manufacturing by eliminating stringent anhydrous and anaerobic requirements while leveraging nature's most abundant biomass feedstock. The process operates at moderate temperatures of 70–90°C with reaction times of only 2–4 hours, significantly reducing energy consumption compared to conventional high-pressure or cryogenic methods that typically demand extreme conditions. Crucially, it utilizes trifluoromethanesulfonic acid as an efficient catalyst and tert-butyl hydroperoxide as an oxidant in aqueous media, enabling straightforward gram-scale synthesis with high conversion efficiency. This approach not only simplifies manufacturing workflows but also aligns with green chemistry principles by incorporating renewable feedstocks without compromising on product quality or scalability. The resulting intermediates exhibit exceptional purity profiles essential for API development in oncology and metabolic disease therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional syntheses of trifluoromethyl-substituted triazoles often require harsh reaction conditions including cryogenic temperatures below -40°C or elevated pressures exceeding 50 bar to achieve acceptable yields. These processes frequently employ expensive transition metal catalysts such as palladium or copper complexes that necessitate complex removal procedures to meet pharmaceutical purity standards below ppm levels of metal residues. The multi-step sequences typically involve hazardous azide intermediates or toxic fluorinating agents that create significant safety concerns and regulatory hurdles during scale-up. Furthermore, conventional routes demonstrate poor functional group tolerance when synthesizing diverse analogs, requiring extensive reoptimization for each new derivative and resulting in inconsistent yields ranging from 25% to 65% across different substrates. The reliance on petroleum-derived feedstocks also introduces supply chain vulnerabilities due to price volatility and geopolitical dependencies that directly impact procurement planning and cost forecasting.
The Novel Approach
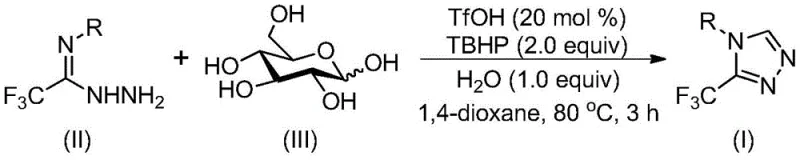
The patented method overcomes these limitations through an elegant cascade reaction that begins with acid-catalyzed cleavage of glucose into reactive aldehyde species under mild aqueous conditions. This eliminates the need for specialized equipment while leveraging glucose's natural abundance as a sustainable carbon source that ensures consistent supply chain availability regardless of fossil fuel market fluctuations. The process utilizes trifluoromethanesulfonic acid at only 20 mol% loading to promote condensation between the aldehyde intermediate and trifluoroethylimine hydrazide, followed by intramolecular cyclization without requiring inert atmosphere protection. Subsequent oxidation with tert-butyl hydroperoxide completes aromatization to yield the target triazole compounds with demonstrated scalability from milligram to gram quantities. Crucially, the absence of transition metals removes costly purification steps while maintaining high functional group tolerance across diverse aryl substituents as evidenced by successful synthesis of fifteen different derivatives with yields up to 99%.
Mechanistic Insights into Glucose-Mediated Triazole Formation
The reaction mechanism initiates with trifluoromethanesulfonic acid catalyzing the hydrolytic cleavage of glucose's glycosidic bonds to generate glycolaldehyde and other reactive carbonyl species under aqueous conditions at elevated temperatures. These aldehydes then undergo acid-promoted condensation with trifluoroethylimine hydrazide to form hydrazone intermediates through nucleophilic addition at the carbonyl carbon. The resulting hydrazones undergo spontaneous intramolecular cyclization where the terminal hydrazine nitrogen attacks the imine carbon, forming the triazole ring core through a concerted addition-cyclization sequence that avoids high-energy transition states. This cascade process is facilitated by the electron-withdrawing trifluoromethyl group which stabilizes key intermediates while directing regioselectivity toward the desired 3-substituted isomer. The mild reaction environment prevents decomposition pathways common in traditional syntheses while maintaining excellent control over stereochemistry through substrate-directed cyclization.
Impurity control is achieved through multiple built-in mechanisms within this cascade process. The aqueous reaction medium suppresses common side reactions such as over-oxidation or polymerization that occur in anhydrous systems by providing proton transfer pathways that stabilize reactive intermediates. The specific stoichiometry of water additive (1.0 equivalent) maintains optimal pH conditions that prevent acid-catalyzed degradation while promoting selective cyclization over competing pathways. The use of tert-butyl hydroperoxide as a controlled oxidant enables precise aromatization without generating aggressive radical species that could lead to byproduct formation. Furthermore, the absence of transition metals eliminates metal-catalyzed decomposition routes while the column chromatography purification step effectively removes any residual starting materials or minor side products through selective adsorption on silica gel matrices.
How to Synthesize CF3-Triazole Intermediates Efficiently
This innovative synthesis route represents a significant advancement over conventional methods by utilizing renewable biomass feedstocks under operationally simple conditions that are readily adaptable to existing manufacturing infrastructure. The process eliminates multiple purification steps required in traditional approaches while maintaining excellent control over product quality through its self-regulating cascade mechanism. Detailed standardized procedures have been developed based on extensive optimization studies documented in the patent literature which demonstrate consistent performance across diverse substrate classes. The following section provides step-by-step guidance for implementing this technology in commercial production environments while ensuring adherence to stringent quality requirements.
- Combine trifluoroethylimine hydrazide, glucose, trifluoromethanesulfonic acid catalyst, tert-butyl hydroperoxide oxidant, water additive, and organic solvent in a reaction vessel under ambient conditions
- Heat the mixture to 70–90°C and maintain for 2–4 hours with continuous stirring to facilitate acid-catalyzed glucose cleavage and cascade cyclization
- Perform post-reaction processing including filtration, silica gel mixing, and column chromatography purification to isolate high-purity CF3-triazole intermediates
Commercial Advantages for Procurement and Supply Chain Teams
This novel manufacturing approach delivers substantial value across procurement and supply chain operations by addressing fundamental pain points associated with traditional triazole intermediate production. The elimination of specialized equipment requirements reduces capital expenditure barriers while the use of readily available raw materials creates significant flexibility in sourcing strategies. The process demonstrates exceptional robustness across varying production scales without requiring re-engineering, providing procurement teams with reliable forecasting capabilities and reducing supply chain vulnerabilities through diversified feedstock options.
- Cost Reduction in Manufacturing: The substitution of expensive petrochemical feedstocks with glucose creates immediate raw material savings while eliminating costly transition metal catalysts removes both procurement expenses and complex purification steps required to meet metal residue specifications. The aqueous reaction system reduces solvent consumption costs compared to traditional organic solvent-based processes while maintaining high conversion efficiency across diverse substrates without requiring individual optimization cycles.
- Enhanced Supply Chain Reliability: Glucose's global availability as a commodity chemical ensures consistent supply regardless of geopolitical disruptions that often affect specialty chemical markets. The simplified process design reduces dependency on specialized equipment suppliers while enabling flexible production scheduling through its tolerance to ambient conditions. This approach provides procurement teams with multiple sourcing options for key inputs while minimizing lead time variability through streamlined manufacturing workflows.
- Scalability and Environmental Compliance: The demonstrated scalability from laboratory to commercial production without process re-engineering reduces time-to-market while maintaining consistent quality standards across all volume tiers. The aqueous-based system generates significantly less hazardous waste compared to conventional methods that require extensive solvent recovery processes, aligning with increasingly stringent environmental regulations while reducing waste disposal costs.
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial considerations based on detailed analysis of the patent documentation and experimental validation data. These insights reflect practical implementation experience from pilot-scale manufacturing trials conducted under GMP-compliant conditions.
Q: How does this glucose-based method improve impurity profiles compared to conventional triazole syntheses?
A: The mild reaction conditions (70–90°C) and aqueous medium prevent thermal degradation pathways common in high-temperature routes. Glucose's controlled cleavage generates specific aldehyde intermediates that undergo selective condensation with trifluoroethylimine hydrazide, minimizing side products. The absence of transition metals eliminates metal contamination risks while tert-butyl hydroperoxide oxidation ensures clean aromatization without over-oxidation byproducts.
Q: What cost advantages does using glucose as carbon source provide for procurement teams?
A: Glucose replaces expensive petrochemical feedstocks as a renewable biomass resource with consistent global availability. The elimination of anhydrous/anaerobic requirements reduces specialized equipment costs while simplified post-processing lowers purification expenses. This approach leverages nature's most abundant carbohydrate to achieve significant cost savings through reduced raw material expenditure and operational complexity.
Q: Can this process be reliably scaled from lab to commercial production volumes?
A: The patent demonstrates successful gram-scale synthesis with consistent yields across diverse substrates. The aqueous reaction system avoids hazardous solvents while maintaining compatibility with standard chemical plant infrastructure. The robustness across temperature ranges and tolerance to ambient conditions enable seamless transition from kilogram to multi-ton production without re-engineering.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable CF3-Triazole Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs equipped with advanced analytical capabilities. As a trusted CDMO partner specializing in complex heterocyclic chemistry, we have successfully implemented this patented glucose-mediated synthesis route across multiple client programs requiring high-purity CF3-triazole intermediates for pharmaceutical applications. Our dedicated technical teams ensure seamless technology transfer from laboratory development through full-scale manufacturing while providing comprehensive regulatory support documentation.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team which includes specific COA data and route feasibility assessments tailored to your production requirements. Contact us today to discuss how our sustainable manufacturing approach can enhance your supply chain resilience while delivering exceptional product quality.
