Revolutionizing Fluorinated Triazole Production Scalable High-Purity Synthesis Pathways for Global Pharmaceutical Supply Chains
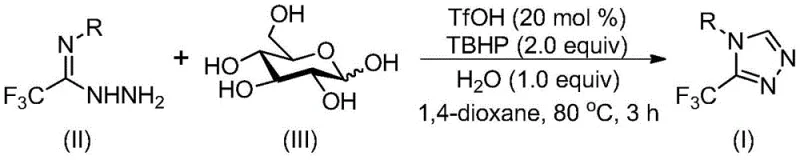
The recently granted Chinese patent CN113880781A represents a significant advancement in fluorinated heterocyclic chemistry through its innovative approach to synthesizing biologically critical 3-trifluoromethyl substituted 1,2,4-triazole compounds using glucose as a renewable carbon source. This methodology addresses longstanding challenges in traditional triazole synthesis by eliminating stringent anhydrous and anaerobic requirements while maintaining exceptional reaction efficiency across diverse substrate scopes. The strategic integration of biomass-derived glucose not only enhances process sustainability but also significantly reduces raw material costs through utilization of nature's most abundant carbohydrate feedstock. Furthermore, the cascade cyclization mechanism enables direct conversion without intermediate isolation steps thereby minimizing operational complexity and waste generation throughout the manufacturing process. This breakthrough holds particular relevance for pharmaceutical manufacturers seeking environmentally responsible routes to fluorinated building blocks essential for modern drug discovery pipelines while simultaneously improving supply chain resilience through simplified logistics and reduced dependency on specialized reagents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for trifluoromethylated triazoles typically require harsh reaction conditions including elevated temperatures exceeding 100°C under strictly anhydrous and anaerobic environments which necessitate specialized equipment and complex operational protocols that significantly increase capital expenditure and operational costs. These methods often employ expensive transition metal catalysts that introduce challenging purification requirements due to potential metal contamination risks which are particularly problematic for pharmaceutical applications requiring ultra-high purity standards. Furthermore, conventional approaches frequently suffer from narrow substrate scope limitations where functional group tolerance restricts structural diversity and complicates access to novel analogs needed for drug discovery programs. The multi-step nature of existing processes also generates substantial waste streams requiring extensive treatment before disposal thereby increasing environmental compliance burdens and overall production costs. Additionally, scalability challenges emerge during process transfer from laboratory to manufacturing scale due to exothermic reaction profiles that demand sophisticated temperature control systems not readily available in standard production facilities.
The Novel Approach
The patented methodology overcomes these limitations through a mild cascade cyclization process operating at moderate temperatures between 70°C and 90°C under ambient atmospheric conditions without requiring specialized moisture exclusion protocols which dramatically simplifies reactor design and operational procedures while reducing capital investment requirements. By utilizing trifluoromethanesulfonic acid as an efficient catalyst that tolerates aqueous environments alongside tert-butyl hydroperoxide as an oxidant system the process eliminates transition metal contamination concerns while maintaining high functional group compatibility across diverse aromatic substrates. The strategic incorporation of glucose as a sustainable carbon source provides inherent cost advantages through its natural abundance and low market price while enabling direct conversion pathways that avoid intermediate isolation steps thereby minimizing waste generation and environmental impact. This approach demonstrates exceptional scalability from gram-scale laboratory reactions to potential industrial production volumes due to its robust thermal profile and straightforward workup procedures involving standard filtration and chromatography techniques accessible across global manufacturing facilities.
Mechanistic Insights into TfOH-Catalyzed Triazole Formation
The reaction mechanism begins with acid-mediated cleavage of glucose under trifluoromethanesulfonic acid catalysis generating reactive aldehyde intermediates that undergo condensation with trifluoroethylimine hydrazide substrates forming hydrazone intermediates through nucleophilic addition processes. This critical step occurs efficiently at moderate temperatures due to the strong protonating ability of TfOH which activates both carbonyl groups while maintaining compatibility with aqueous reaction media that would typically disrupt conventional organometallic catalysis systems. Subsequent intramolecular cyclization proceeds through nucleophilic attack by the hydrazine nitrogen on the imine carbon center forming the triazole ring structure with simultaneous elimination of water molecules which drives the reaction equilibrium toward product formation without requiring additional dehydrating agents. The final aromatization step is facilitated by tert-butyl hydroperoxide oxidation which completes the electronic reorganization necessary to establish the fully conjugated heterocyclic system while maintaining excellent regioselectivity due to the inherent electronic properties of the trifluoromethyl group directing substitution patterns.
Impurity control is achieved through multiple intrinsic mechanisms within this cascade process where the mild reaction conditions prevent thermal decomposition pathways that typically generate polymeric byproducts in conventional high-temperature syntheses. The selective oxidation step mediated by tert-butyl hydroperoxide specifically targets only the required intermediate species without over-oxidation side reactions that could produce unwanted fluorinated impurities commonly observed in alternative methodologies. Furthermore the aqueous-compatible nature of this system eliminates moisture-induced degradation products that plague traditional anhydrous routes while maintaining excellent chemoselectivity due to the precise electronic tuning provided by the trifluoromethyl group which directs cyclization regiochemistry toward single isomer formation. This inherent selectivity reduces downstream purification burden significantly improving overall process efficiency while ensuring consistent high-purity output meeting pharmaceutical industry specifications without requiring additional costly separation steps.
How to Synthesize CF3-Triazole Efficiently
This innovative synthesis pathway represents a significant advancement over conventional methodologies by leveraging sustainable biomass feedstocks within a streamlined manufacturing process that eliminates multiple operational complexities while maintaining excellent yield profiles across diverse substrate classes. The methodology demonstrates exceptional practicality through its compatibility with standard laboratory equipment and straightforward procedure that requires no specialized training or infrastructure investments making it immediately adoptable by existing manufacturing facilities worldwide. Detailed standardized synthesis protocols have been developed based on extensive optimization studies documented in the patent literature which provide precise guidance for achieving optimal results across various production scales from research quantities to commercial volumes. The following section outlines critical implementation parameters that ensure consistent success when adopting this technology.
- Combine trifluoromethanesulfonic acid catalyst with trifluoroethylimine hydrazide substrate and glucose carbon source in anhydrous organic solvent under ambient atmospheric conditions.
- Initiate reaction at precisely controlled temperature between 70°C and 90°C using tert-butyl hydroperoxide oxidant with water additive to facilitate cascade cyclization.
- Conduct post-processing through filtration followed by silica gel-assisted column chromatography purification to isolate high-purity triazole products without specialized equipment.
Commercial Advantages for Procurement and Supply Chain Teams
This patented methodology delivers substantial value across procurement and supply chain operations by addressing fundamental pain points associated with traditional triazole synthesis routes through its inherently simplified process design and sustainable raw material sourcing strategy. The elimination of transition metal catalysts removes significant cost drivers related to catalyst procurement handling and post-reaction removal processes while simultaneously reducing quality control burdens associated with metal residue testing requirements that delay product release timelines. Furthermore the use of readily available biomass-derived glucose as primary carbon source creates supply chain resilience through multiple global sourcing options that mitigate single-point failure risks common in specialized chemical supply networks while maintaining consistent pricing stability unaffected by petrochemical market fluctuations.
- Cost Reduction in Manufacturing: The removal of expensive transition metal catalysts combined with simplified workup procedures significantly lowers raw material expenses while eliminating specialized equipment requirements reduces capital investment needs substantially improving overall manufacturing economics without requiring additional operational expenditures.
- Enhanced Supply Chain Reliability: Utilization of globally available glucose feedstock ensures consistent raw material availability across multiple geographic regions while eliminating moisture-sensitive handling requirements simplifies logistics operations reducing lead time variability through more predictable production scheduling capabilities.
- Scalability and Environmental Compliance: The aqueous-compatible process design enables seamless scale-up from laboratory to commercial production volumes without requiring major equipment modifications while generating minimal waste streams that align with green chemistry principles reducing environmental compliance costs significantly.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial concerns regarding implementation of this patented methodology based on detailed analysis of its operational parameters and performance characteristics documented within CN113880781A. These responses provide evidence-based insights derived directly from experimental data presented in the patent specification without speculative assumptions or unsubstantiated claims regarding performance metrics.
Q: How does this glucose-based method overcome traditional triazole synthesis limitations requiring strict anhydrous conditions?
A: The patented process eliminates anhydrous requirements through water-tolerant TfOH catalysis where glucose cracking occurs under aqueous conditions enabling simplified reactor setup and reduced operational complexity compared to conventional moisture-sensitive routes.
Q: What sustainability advantages does biomass-derived glucose provide in fluorinated heterocycle manufacturing?
A: Using naturally abundant glucose as carbon source reduces petrochemical dependency while maintaining high atom economy through cascade reactions that minimize waste streams and lower environmental impact without compromising product quality.
Q: How does this methodology ensure consistent high-purity output suitable for pharmaceutical applications?
A: The mild reaction conditions prevent thermal degradation pathways while selective oxidation controls byproduct formation resulting in inherently cleaner reaction profiles that simplify purification to meet stringent pharmaceutical quality standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable CF3-Triazole Supplier
Our company possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs equipped with advanced analytical capabilities specifically designed for fluorinated heterocyclic compounds. This patented glucose-based methodology aligns perfectly with our commitment to sustainable manufacturing practices while leveraging our deep expertise in complex heterocycle synthesis to deliver consistent high-quality intermediates meeting global regulatory requirements across all major markets including FDA EMA and PMDA jurisdictions.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team which will provide specific COA data and route feasibility assessments tailored to your unique production requirements enabling informed decision-making regarding implementation timelines and resource allocation strategies.
