Scalable Synthesis of Carbonyl-Bridged Biheterocycles via Safe Palladium Catalysis for Pharma Intermediates
Scalable Synthesis of Carbonyl-Bridged Biheterocycles via Safe Palladium Catalysis for Pharma Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex heterocyclic scaffolds, which serve as critical cores for bioactive molecules. A significant breakthrough in this domain is detailed in Chinese Patent CN115353511A, which discloses a novel multi-component method for synthesizing carbonyl-bridged biheterocyclic compounds. This technology represents a paradigm shift from traditional, hazardous carbonylation techniques to a safer, more efficient cascade reaction protocol. By leveraging a palladium-catalyzed system that generates carbon monoxide in-situ from formic acid and acetic anhydride, the process circumvents the logistical and safety nightmares associated with handling toxic CO gas. For R&D directors and procurement specialists alike, this innovation offers a pathway to high-purity intermediates with reduced operational risk and enhanced process reliability.
The core of this invention lies in its ability to construct indolinone-imidazole fused systems through a one-pot tandem reaction. These biheterocyclic structures are prevalent in medicinal chemistry due to their broad-spectrum biological activities, making them highly valuable targets for drug discovery programs. The patent outlines a procedure where trifluoroethylimidoyl chloride, propargylamine, and substituted acrylamides converge in the presence of a PdCl2/TFP catalyst system. The reaction proceeds smoothly at a mild temperature of 30°C, demonstrating exceptional functional group tolerance. This mildness is crucial for preserving sensitive moieties often found in advanced pharmaceutical intermediates, thereby reducing the need for protective group strategies and streamlining the overall synthetic route.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of carbonyl-bridged heterocycles has been plagued by significant safety and efficiency hurdles. Traditional carbonylation reactions typically rely on the direct use of carbon monoxide gas, which is highly toxic and requires specialized high-pressure equipment and rigorous safety protocols. This dependency creates a substantial barrier to entry for many manufacturing facilities, inflating capital expenditure and operational costs. Furthermore, conventional methods often involve multi-step sequences with harsh reaction conditions, such as high temperatures or strong acids, which can lead to substrate decomposition and the formation of complex impurity profiles. These factors collectively result in lower overall yields and increased waste generation, posing challenges for both economic viability and environmental compliance in large-scale production settings.
The Novel Approach
In stark contrast, the methodology described in CN115353511A introduces a transformative approach by utilizing a formic acid and acetic anhydride mixture as a safe, liquid carbon monoxide surrogate. This innovation allows the carbonylation step to occur under atmospheric pressure and mild thermal conditions, effectively democratizing access to these complex transformations. The multi-component nature of the reaction enables the simultaneous formation of multiple chemical bonds—specifically C-C and C-N bonds—in a single operational step. This telescoping of reactions not only accelerates the synthesis timeline but also minimizes solvent usage and purification steps. As illustrated in the reaction scheme below, the convergence of three distinct building blocks into a sophisticated biheterocyclic framework exemplifies the power of modern catalytic design.
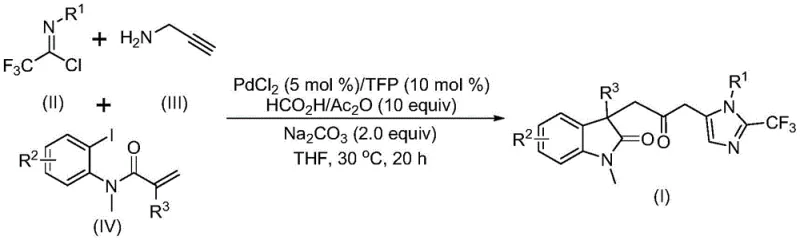
The versatility of this new approach is further evidenced by its broad substrate scope. The patent data indicates that various substituents, including halogens, alkyl groups, and electron-withdrawing groups like trifluoromethyl and nitro, are well-tolerated on the aromatic rings. This flexibility allows chemists to rapidly generate diverse libraries of analogs for structure-activity relationship (SAR) studies without needing to re-optimize reaction conditions for each new substrate. Such adaptability is a key asset for reliable pharmaceutical intermediate suppliers aiming to support fast-paced drug discovery pipelines.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
Understanding the mechanistic underpinnings of this transformation is vital for R&D teams looking to implement or modify the process. The reaction is believed to initiate with the oxidative addition of a zero-valent palladium species into the carbon-iodine bond of the acrylamide substrate. This is followed by an intramolecular Heck-type cyclization, generating a divalent alkyl-palladium intermediate. Crucially, the carbon monoxide required for the subsequent carbonylation step is released in-situ from the decomposition of the formic acid/acetic anhydride adduct. This CO then inserts into the palladium-carbon bond to form an acyl-palladium intermediate, setting the stage for the final ring closure.
Parallel to the palladium cycle, the trifluoroethylimidoyl chloride and propargylamine undergo a base-promoted intermolecular reaction to form a trifluoroacetamidine species, which subsequently isomerizes. The acyl-palladium intermediate then activates this amidine compound, catalyzing an intramolecular cyclization that furnishes the final carbonyl-bridged biheterocyclic product. This intricate dance of catalytic cycles ensures high atom economy and selectivity. The use of trifurylphosphine (TFP) as a ligand is particularly noteworthy, as it likely stabilizes the active palladium species and facilitates the difficult reductive elimination step, thereby driving the reaction to completion with high yields, often exceeding 80% for optimized substrates.
How to Synthesize Carbonyl-Bridged Biheterocycles Efficiently
Implementing this synthesis requires careful attention to reagent quality and reaction parameters to maximize yield and purity. The process is designed to be operationally simple, involving the mixing of all components in a suitable organic solvent such as tetrahydrofuran (THF). The mild temperature requirement of 30°C means that standard laboratory glassware or stainless steel reactors can be used without the need for specialized heating or cooling jackets. Detailed standardized synthesis steps follow the logic of the patent examples, ensuring reproducibility across different scales.
- Mix palladium chloride catalyst, trifurylphosphine ligand, sodium carbonate base, and the formic acid/acetic anhydride CO source in an organic solvent like THF.
- Add trifluoroethylimidoyl chloride, propargylamine, and the acrylamide substrate to the reaction mixture and stir at 30°C for 12 to 20 hours.
- Upon completion, filter the mixture, adsorb onto silica gel, and purify via column chromatography to isolate the target carbonyl-bridged biheterocyclic compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology translates into tangible strategic benefits beyond mere chemical elegance. The elimination of toxic carbon monoxide gas from the supply chain removes a major regulatory burden and safety liability. Facilities no longer need to invest in expensive gas detection systems or maintain high-pressure storage infrastructure, leading to significant overhead cost reductions. Furthermore, the use of commercially available and inexpensive starting materials, such as propargylamine and acryloyl chloride derivatives, ensures a stable and resilient supply base. This reduces the risk of production delays caused by raw material shortages, a critical factor for maintaining continuity in pharmaceutical manufacturing.
- Cost Reduction in Manufacturing: The streamlined one-pot nature of this reaction drastically reduces the number of unit operations required. By combining multiple bond-forming events into a single vessel, manufacturers save on solvent consumption, energy for heating and cooling, and labor hours associated with intermediate isolations. The mild reaction temperature of 30°C further contributes to energy efficiency compared to processes requiring reflux conditions. Additionally, the avoidance of expensive transition metal removal steps, often necessitated by other catalytic systems, simplifies the downstream purification process, directly lowering the cost of goods sold (COGS) for the final intermediate.
- Enhanced Supply Chain Reliability: The robustness of this catalytic system against various functional groups means that supply chains are less vulnerable to disruptions caused by the need for highly specialized, custom-synthesized precursors. The starting materials are commodity chemicals or easily derived from them, ensuring long-term availability. This stability allows for better forecasting and inventory management. Moreover, the scalability demonstrated in the patent, extending from milligram to gram scales without loss of efficiency, provides confidence that the process can be seamlessly transferred to pilot and commercial plants to meet fluctuating market demands.
- Scalability and Environmental Compliance: From an environmental perspective, the in-situ generation of CO minimizes the release of greenhouse gases and toxic emissions associated with gas handling. The reaction produces fewer byproducts, simplifying waste treatment and disposal protocols. The ability to run the reaction in common solvents like THF, which can be readily recovered and recycled, aligns with green chemistry principles. This environmental friendliness not only reduces disposal costs but also enhances the corporate sustainability profile, a growing requirement for suppliers partnering with major multinational pharmaceutical corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis technology. They are derived from the specific experimental data and beneficial effects outlined in the patent documentation, providing clarity for potential partners evaluating this route for their own production needs.
Q: What is the primary safety advantage of this synthesis method compared to traditional carbonylation?
A: Unlike conventional methods that require handling toxic carbon monoxide gas cylinders under high pressure, this patent utilizes a formic acid and acetic anhydride mixture to generate carbon monoxide in-situ. This eliminates the need for specialized high-pressure gas infrastructure and significantly reduces workplace safety hazards associated with CO leakage.
Q: What are the typical reaction conditions and catalysts used in this process?
A: The process operates under mild conditions, specifically at 30°C in tetrahydrofuran (THF) solvent. It employs a palladium chloride (PdCl2) catalyst system with trifurylphosphine (TFP) as the ligand, along with sodium carbonate as the base, ensuring high efficiency without extreme thermal stress.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the patent explicitly demonstrates the scalability of the method to gram-level reactions. The use of commercially available starting materials, mild reaction temperatures, and simplified post-treatment procedures (filtration and chromatography) indicates strong potential for commercial scale-up in pharmaceutical intermediate manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocycle Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the multi-component carbonylation technology described in CN115353511A for producing high-value pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle palladium-catalyzed reactions with stringent purity specifications, ensuring that every batch meets the rigorous quality standards required by global regulatory agencies. Our rigorous QC labs employ advanced analytical techniques to verify the structural integrity and impurity profile of these complex biheterocyclic compounds, guaranteeing consistency and reliability for your drug development programs.
We invite you to collaborate with us to leverage this efficient synthesis route for your next project. By partnering with our technical procurement team, you can obtain a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise can accelerate your timeline to market while optimizing your manufacturing budget.
