Revolutionizing Carbonyl-Bridged Biheterocyclic Synthesis via Safe Palladium-Catalyzed Multicomponent Strategies for Commercial Scale
Revolutionizing Carbonyl-Bridged Biheterocyclic Synthesis via Safe Palladium-Catalyzed Multicomponent Strategies for Commercial Scale
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex heterocyclic scaffolds that serve as critical building blocks for bioactive molecules. A significant breakthrough in this domain is documented in Chinese Patent CN115353511A, which discloses a novel multi-component method for synthesizing carbonyl-bridged biheterocyclic compounds. This technology addresses long-standing challenges in organic synthesis by enabling the efficient assembly of indolinone and imidazole motifs through a palladium-catalyzed cascade reaction. Unlike traditional approaches that often rely on hazardous reagents or multi-step sequences with poor atom economy, this invention utilizes a safe, in situ carbon monoxide source derived from formic acid and acetic anhydride. The process operates under remarkably mild thermal conditions, typically around 30°C, and demonstrates exceptional compatibility with diverse functional groups. For research and development teams focused on drug discovery, this methodology offers a streamlined pathway to access diversified libraries of trifluoromethyl-containing biheterocycles, which are increasingly valued for their metabolic stability and binding affinity in medicinal chemistry applications.
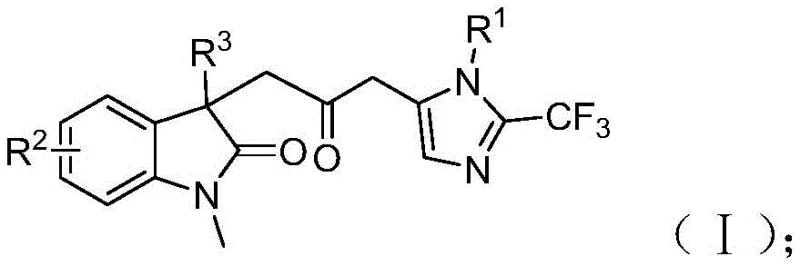
The core structural motif generated by this process, as illustrated in the patent documentation, features a carbonyl bridge connecting an indolinone ring system with a trifluoromethyl-substituted imidazole ring. This specific architecture is highly relevant for the development of next-generation therapeutic agents, particularly in oncology and anti-inflammatory fields where dual-heterocyclic systems exhibit potent biological activity. The ability to introduce a trifluoromethyl group directly during the cyclization process adds significant value, as fluorination is a key strategy for modulating the lipophilicity and pKa of drug candidates. By integrating the formation of multiple chemical bonds—including carbon-carbon, carbon-nitrogen, and carbon-oxygen bonds—into a single operational sequence, this technology drastically reduces the number of isolation steps required. Consequently, this leads to higher overall throughput and reduced waste generation, aligning perfectly with the principles of green chemistry that modern chemical manufacturers strive to implement in their production facilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of carbonyl-bridged biheterocyclic compounds has been fraught with significant technical and safety hurdles that impede efficient manufacturing. Conventional strategies often necessitate the use of pressurized carbon monoxide gas, which poses severe safety risks due to its high toxicity and flammability, requiring specialized high-pressure reactors and extensive safety infrastructure that drive up capital expenditure. Furthermore, traditional methods frequently involve the direct coupling of two pre-formed heterocyclic substrates, which can suffer from low reactivity, poor regioselectivity, and the need for harsh reaction conditions such as elevated temperatures or strong bases that limit functional group tolerance. Another common approach involves transition metal-catalyzed tandem cyclizations that may require expensive ligands or stoichiometric amounts of oxidants, leading to increased production costs and complex waste streams that are difficult to treat. These limitations not only extend the lead time for process development but also create bottlenecks in the supply chain when scaling from milligram discovery quantities to kilogram commercial batches, making it difficult for procurement managers to secure reliable sources of high-purity intermediates.
The Novel Approach
In stark contrast to these legacy techniques, the method described in patent CN115353511A introduces a transformative one-pot multicomponent reaction that elegantly bypasses the need for external carbon monoxide gas. By employing a mixture of formic acid and acetic anhydride, the system generates the necessary carbonyl species in situ under ambient pressure, thereby eliminating the need for specialized gas handling equipment and significantly enhancing operational safety. This novel approach leverages a palladium catalyst system, specifically palladium chloride paired with trifurylphosphine, to orchestrate a complex cascade involving oxidative addition, intramolecular Heck reaction, and carbonylation in a seamless sequence. The reaction proceeds efficiently at a mild temperature of 30°C in common organic solvents like tetrahydrofuran, demonstrating remarkable substrate versatility that allows for the incorporation of various substituents on the aromatic rings without compromising yield. This strategic shift from hazardous gas-phase reagents to safe liquid-phase surrogates represents a paradigm shift in process chemistry, offering a reliable pharmaceutical intermediate supplier a distinct competitive advantage through simplified operations and reduced regulatory compliance burdens.
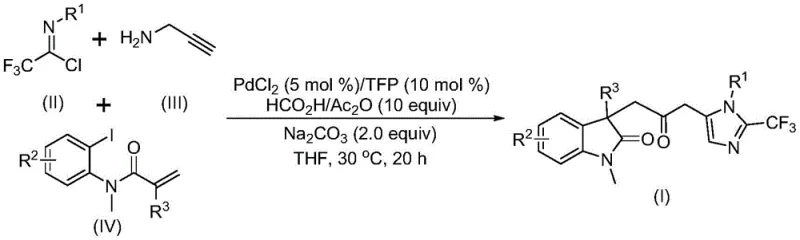
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
The mechanistic pathway of this transformation is a sophisticated example of transition metal catalysis that ensures high selectivity and efficiency. The cycle likely initiates with the reduction of the palladium(II) precatalyst to an active palladium(0) species, which then undergoes oxidative addition into the carbon-iodine bond of the acrylamide derivative. This key step generates a divalent alkyl-palladium intermediate that is poised for subsequent intramolecular insertion. Following this, the intermediate undergoes an intramolecular Heck-type reaction to form the indolinone core, establishing the first heterocyclic ring with high stereochemical control. Crucially, the presence of the formic acid and acetic anhydride mixture facilitates the release of carbon monoxide, which inserts into the palladium-carbon bond to form an acyl-palladium species. This carbonylation step is the linchpin of the process, bridging the two heterocyclic systems. Finally, the acyl-palladium intermediate activates the trifluoroacetamidine compound, formed from the reaction of trifluoroethylimidoyl chloride and propargylamine, triggering a final intramolecular cyclization to close the imidazole ring and release the final carbonyl-bridged product while regenerating the active catalyst.
From an impurity control perspective, this mechanism offers inherent advantages that are critical for meeting stringent purity specifications required in pharmaceutical manufacturing. The concerted nature of the cascade reaction minimizes the accumulation of reactive intermediates that could otherwise lead to side reactions or polymerization. The use of a mild base like sodium carbonate helps to neutralize acidic byproducts without promoting degradation of sensitive functional groups such as esters or nitriles that might be present on the substrate. Furthermore, the specific choice of the trifurylphosphine ligand enhances the stability of the palladium center, preventing the formation of palladium black which can act as a heterogeneous catalyst for non-selective pathways. This high level of chemoselectivity ensures that the crude reaction mixture contains fewer byproducts, simplifying the downstream purification process. For quality control laboratories, this translates to cleaner chromatograms and higher recovery rates during column chromatography or crystallization, ultimately ensuring that the final high-purity pharmaceutical intermediates meet the rigorous standards demanded by global regulatory agencies.
How to Synthesize Carbonyl-Bridged Biheterocyclic Compounds Efficiently
To implement this advanced synthesis route effectively, operators must adhere to precise stoichiometric ratios and mixing protocols to maximize the turnover number of the palladium catalyst. The standard procedure involves charging a reaction vessel with the palladium source, ligand, and base, followed by the sequential addition of the three key coupling partners in an aprotic solvent. Maintaining the reaction temperature strictly at 30°C is vital to balance the rate of CO generation with the rate of the catalytic cycle, preventing the buildup of pressure or the decomposition of sensitive reagents. Detailed standardized operating procedures regarding the order of addition and stirring rates are essential to ensure reproducibility across different batch sizes. For a comprehensive guide on the exact molar equivalents and workup procedures validated in the patent examples, please refer to the structured synthesis guide below.
- Combine palladium chloride, trifurylphosphine ligand, sodium carbonate, and the formic acid/acetic anhydride mixture in an organic solvent such as THF.
- Add the trifluoroethylimidoyl chloride, propargylamine, and acrylamide derivatives to the reaction vessel under stirring.
- Maintain the reaction mixture at 30°C for 12 to 20 hours, followed by filtration and column chromatography purification to isolate the target biheterocycle.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented technology offers substantial strategic benefits that extend far beyond simple yield improvements. The elimination of toxic carbon monoxide gas from the process flow fundamentally alters the risk profile of the manufacturing site, removing the need for expensive gas detection systems, specialized ventilation, and high-pressure containment vessels. This reduction in infrastructure requirements directly correlates to lower capital expenditure for new production lines and reduced maintenance costs for existing facilities. Moreover, the reliance on commercially available and inexpensive starting materials such as propargylamine and acrylamide derivatives ensures a stable and resilient supply chain that is less susceptible to market volatility compared to processes requiring exotic or custom-synthesized reagents. The ability to source these raw materials from multiple vendors enhances supply security, mitigating the risk of production stoppages due to single-source dependencies.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the simplification of the operational workflow and the reduction of waste disposal costs. By consolidating multiple bond-forming events into a single pot, the method eliminates the need for intermediate isolation, drying, and purification steps, which are traditionally labor-intensive and solvent-heavy. This telescoping of steps significantly reduces the consumption of organic solvents and silica gel, leading to a smaller environmental footprint and lower costs associated with solvent recovery and hazardous waste treatment. Additionally, the use of palladium chloride, a relatively inexpensive palladium source compared to other complexes, combined with the high turnover efficiency of the catalyst system, minimizes the cost contribution of precious metals to the final product. These factors collectively contribute to a more cost-competitive manufacturing process without sacrificing quality.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route under mild conditions enhances the reliability of supply for critical pharmaceutical intermediates. Because the reaction does not require extreme temperatures or pressures, it is less prone to thermal runaways or equipment failures that can cause unplanned downtime. The broad substrate scope means that a single manufacturing platform can be adapted to produce a wide variety of analogues by simply swapping out the starting aryl halides or imidoyl chlorides, providing flexibility to respond quickly to changing customer demands. This adaptability ensures that supply chain heads can maintain continuity of supply even when specific drug candidates undergo structural optimization during clinical development, as the core process remains valid across a wide range of derivatives.
- Scalability and Environmental Compliance: Scaling this process from gram-scale laboratory experiments to multi-kilogram commercial production is facilitated by the homogeneous nature of the reaction and the absence of gaseous reagents. The patent data confirms successful execution at the gram level with high yields, indicating a smooth path to larger scales without encountering mass transfer limitations often associated with gas-liquid reactions. From an environmental compliance standpoint, the use of formic acid as a CO surrogate generates water and carbon dioxide as benign byproducts, aligning with increasingly strict environmental regulations regarding volatile organic compound emissions. This green chemistry profile simplifies the permitting process for new manufacturing sites and supports corporate sustainability goals, making the technology attractive for long-term investment in eco-friendly production capabilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed multicomponent synthesis. These answers are derived directly from the experimental data and beneficial effects reported in the patent literature, providing clarity on the practical aspects of adopting this technology. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: How does this method improve safety compared to traditional carbonylation?
A: Unlike conventional methods requiring high-pressure toxic carbon monoxide gas cylinders, this protocol generates CO in situ from formic acid and acetic anhydride, significantly reducing facility safety risks and regulatory burdens.
Q: What is the substrate scope for the R1 and R3 groups?
A: The reaction demonstrates excellent functional group tolerance, accommodating alkyl, substituted aryl groups with electron-donating or withdrawing substituents like methyl, methoxy, halogens, and nitro groups without compromising yield.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly validates the method at the gram scale with simple post-treatment procedures, indicating strong potential for commercial scale-up due to mild conditions and readily available starting materials.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocyclic Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic methodology described in patent CN115353511A for the production of high-value pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped to handle palladium-catalyzed reactions with the highest safety standards, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the exacting requirements of global pharmaceutical clients. We are committed to leveraging this advanced chemistry to deliver cost-effective and reliable supplies of complex biheterocyclic building blocks.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this safer and more efficient manufacturing process. We encourage you to contact us today to obtain specific COA data for our catalog compounds or to initiate route feasibility assessments for your proprietary molecules, ensuring that your supply chain is optimized for speed, quality, and sustainability.
