Scalable Metal-Free Synthesis of 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Derivatives for Advanced Pharma Applications
Scalable Metal-Free Synthesis of 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Derivatives for Advanced Pharma Applications
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and cost-effective methodologies for constructing complex heterocyclic scaffolds, particularly those containing nitrogen-rich motifs like 1,2,4-triazoles. A groundbreaking development in this sector is detailed in Chinese Patent CN113307790B, which discloses a highly efficient preparation method for 3-quinolyl-5-trifluoromethyl substituted 1,2,4-triazole compounds. These structures are not merely academic curiosities; they serve as critical building blocks for bioactive molecules and functional materials, including potential ligands for organic light-emitting diodes (OLEDs). The patent introduces a transformative oxidative cyclization strategy that bypasses the limitations of classical synthesis, utilizing a metal-free catalytic system composed of tetrabutylammonium iodide (TBAI) and tert-butyl hydroperoxide (TBHP). This innovation represents a significant leap forward for any organization aiming to secure a reliable pharmaceutical intermediate supplier capable of delivering high-purity compounds with improved process economics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinolyl-substituted 1,2,4-triazoles has been plagued by inefficiencies that hinder commercial viability. Traditional protocols often rely on quinoline-2-carboxylic acid as the primary starting material, necessitating a cumbersome five-step reaction sequence to achieve the final target molecule. This multi-step approach is inherently problematic for large-scale manufacturing due to the accumulation of impurities, the requirement for rigorous purification between steps, and the overall low atom economy. Furthermore, the patent data highlights that these legacy methods typically result in a dismal total yield of approximately 17%, rendering them economically unfeasible for high-volume production. The severe reaction conditions often required, such as extreme temperatures or the use of hazardous reagents, further exacerbate safety concerns and operational costs, making these conventional routes unsuitable for modern, green chemistry standards demanded by top-tier procurement managers.
The Novel Approach
In stark contrast to these archaic methods, the technology described in CN113307790B offers a streamlined, one-pot solution that dramatically enhances efficiency and yield. By employing cheap and readily available 2-methylquinoline and trifluoroacetimidohydrazide as starting materials, the process leverages an oxidative cyclization mechanism promoted by the TBAI/TBHP system. This novel approach eliminates the need for anhydrous or oxygen-free environments, simplifying the operational requirements significantly. The reaction proceeds smoothly in common organic solvents like DMSO at moderate temperatures of 80-100°C. Most impressively, the method demonstrates exceptional substrate tolerance, allowing for the introduction of diverse substituents on both the quinoline and phenyl rings without compromising yield. As illustrated in the general reaction scheme below, this direct coupling strategy converts simple precursors into complex triazole derivatives with yields reaching as high as 97% in optimized examples, marking a substantial improvement over the 17% ceiling of previous technologies.
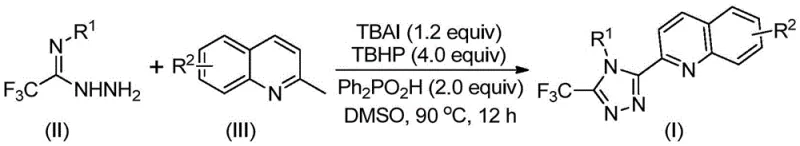
Mechanistic Insights into TBAI/TBHP Promoted Oxidative Cyclization
Understanding the mechanistic underpinnings of this reaction is crucial for R&D directors evaluating its robustness and reproducibility. The process is believed to initiate with the oxidation of the methyl group on the 2-methylquinoline substrate. Under the influence of the TBAI and TBHP catalytic system, the methyl group is oxidatively converted into a reactive 2-quinolinecarbaldehyde intermediate in situ. This aldehyde then undergoes a condensation reaction with the trifluoroacetimidohydrazide to form a dehydrated hydrazone intermediate. Following this condensation, the system facilitates an oxidative iodination event, which activates the hydrazone for subsequent intramolecular electrophilic substitution. This cyclization step closes the triazole ring, followed by aromatization to yield the final stable 3-quinolyl-5-trifluoromethyl-1,2,4-triazole product. Alternatively, the patent suggests the possibility of a radical-mediated pathway, highlighting the versatility of the oxidative conditions. Crucially, the absence of transition metals means there is no risk of metal contamination in the final API, a critical quality attribute that simplifies downstream processing and regulatory approval.
From an impurity control perspective, this mechanism offers distinct advantages. The use of diphenylphosphoric acid as an additive helps to modulate the acidity of the reaction medium, promoting the desired cyclization while suppressing side reactions such as over-oxidation or polymerization of the sensitive aldehyde intermediate. The high selectivity observed across various substrates (Examples 1-15) indicates that the reaction conditions are finely tuned to favor the formation of the 1,2,4-triazole core over other potential isomers. For instance, electron-donating groups like methoxy or methyl on the phenyl ring (R1) and various substituents on the quinoline ring (R2) are well-tolerated, consistently delivering high purity products. This broad scope ensures that the process is not limited to a single molecule but serves as a platform technology for generating a library of diversified triazole intermediates, providing significant value for drug discovery teams exploring structure-activity relationships.
How to Synthesize 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Efficiently
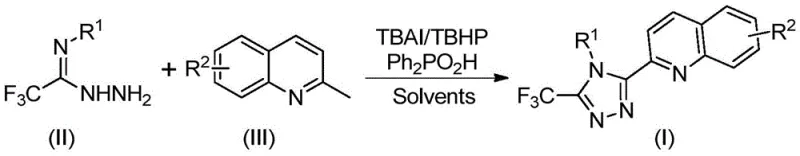
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific parameters to maximize yield and safety. The protocol involves charging a reaction vessel with the requisite molar ratios of tetrabutylammonium iodide, tert-butyl peroxide aqueous solution, diphenylphosphoric acid, trifluoroacetimidohydrazide, and 2-methylquinoline in a solvent like DMSO. The mixture is then heated to the optimal temperature range and monitored until conversion is complete. The simplicity of the work-up procedure, involving filtration and standard column chromatography, makes this method highly attractive for rapid iteration and scale-up. The specific reaction setup and stoichiometry used in the patent examples provide a clear blueprint for replication, ensuring that technical teams can achieve consistent results. Below is the visual representation of the specific experimental reaction conditions utilized in the patent examples, serving as a practical guide for execution.
- Combine tetrabutylammonium iodide, tert-butyl peroxide aqueous solution, diphenylphosphoric acid, trifluoroacetimidohydrazide, and 2-methylquinoline in an organic solvent such as DMSO.
- Heat the reaction mixture to a temperature range of 80-100°C and maintain stirring for 8 to 14 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target 3-quinolyl-5-trifluoromethyl-1,2,4-triazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the shift from traditional multi-step syntheses to this novel one-pot oxidative cyclization represents a strategic opportunity for cost reduction in pharmaceutical intermediate manufacturing. The primary economic driver is the drastic simplification of the synthetic route. By collapsing a five-step sequence into a single operational unit, the process inherently reduces labor costs, energy consumption, and solvent usage. Furthermore, the elimination of heavy metal catalysts is a major financial win; it removes the necessity for expensive scavenging resins or complex extraction protocols required to meet strict residual metal specifications in active pharmaceutical ingredients. This metal-free nature not only lowers direct material costs but also accelerates the production cycle time, allowing for faster turnaround on orders and improved responsiveness to market demand fluctuations.
- Cost Reduction in Manufacturing: The economic benefits of this process are multifaceted, stemming primarily from the use of commodity chemicals as starting materials. 2-methylquinoline and trifluoroacetic acid derivatives are widely available in the global chemical market at competitive prices, ensuring a stable and low-cost raw material base. The high reaction yields, frequently exceeding 80% and reaching up to 97% in optimized cases, mean that less raw material is wasted per kilogram of product produced, directly improving the cost of goods sold (COGS). Additionally, the avoidance of specialized equipment for handling air- or moisture-sensitive reagents reduces capital expenditure requirements for the manufacturing facility, making the technology accessible for a broader range of production partners.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness and flexibility of this synthetic method. Because the reaction does not require stringent anhydrous or anaerobic conditions, it is less susceptible to failure due to minor variations in environmental controls or operator error. The wide substrate scope allows for the sourcing of alternative starting materials if specific substituted quinolines become scarce, providing a buffer against supply disruptions. The use of common solvents like DMSO, which are produced at massive scales globally, further mitigates the risk of solvent shortages that can plague niche synthetic routes. This reliability ensures consistent delivery schedules, a critical metric for supply chain heads managing just-in-time inventory systems for downstream API production.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, this technology aligns perfectly with modern green chemistry principles. The reduction in step count inherently lowers the E-factor (mass of waste per mass of product), reducing the burden on waste treatment facilities and lowering disposal costs. The ability to run the reaction at moderate temperatures (80-100°C) minimizes energy intensity compared to high-temperature or cryogenic processes. The patent explicitly notes that the reaction can be easily expanded to gram-scale and implies potential for larger batches, suggesting that the heat transfer and mixing dynamics are manageable in larger reactors. This scalability ensures that the process can grow with demand, from clinical trial quantities to commercial tonnage, without requiring a complete process redevelopment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity for technical evaluators. Understanding these nuances is essential for assessing the fit of this methodology within your existing manufacturing portfolio and for anticipating potential challenges during technology transfer.
Q: What are the key advantages of this synthesis method over traditional routes?
A: Unlike traditional methods requiring quinoline-2-carboxylic acid which suffer from low yields (around 17%) and harsh multi-step conditions, this novel approach utilizes cheap 2-methylquinoline and achieves high yields (up to 97%) in a single pot without heavy metal catalysts.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly states the reaction can be easily expanded to gram-scale and potentially larger. The use of commercially available reagents like TBAI and TBHP, along with simple post-treatment involving filtration and chromatography, supports scalability.
Q: What represents the primary cost-saving factor in this manufacturing process?
A: The elimination of expensive transition metal catalysts removes the need for complex and costly heavy metal removal steps during purification. Additionally, the starting materials are inexpensive and widely available, significantly lowering the raw material cost base.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic route described in CN113307790B for producing high-value heterocyclic intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless. Our state-of-the-art facilities are equipped to handle the specific oxidative conditions and solvent systems required for this chemistry, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest industry standards. We understand that consistency and quality are non-negotiable in the pharmaceutical supply chain, and our dedicated technical team is prepared to validate this metal-free process to deliver the reliability your projects demand.
We invite you to collaborate with us to leverage this advanced manufacturing capability for your next project. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this efficient route can optimize your budget. We encourage you to contact our technical procurement team today to request specific COA data for similar triazole derivatives and to discuss route feasibility assessments for your target molecules. Let us help you secure a competitive advantage through superior chemistry and supply chain excellence.
