Scalable Metal-Free Synthesis of 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazoles for Pharma
Scalable Metal-Free Synthesis of 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazoles for Pharma
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more efficient, cost-effective, and environmentally benign synthetic routes. A significant breakthrough in this domain is detailed in Chinese patent CN113307790B, which discloses a robust preparation method for 3-quinolyl-5-trifluoromethyl substituted 1,2,4-triazole compounds. These heterocyclic scaffolds are critical building blocks in medicinal chemistry, frequently appearing in the molecular frameworks of bioactive agents ranging from antifungal drugs to kinase inhibitors. The structural versatility of the 1,2,4-triazole ring allows it to serve not only as a pharmacophore but also as a potent bidentate ligand in coordination chemistry, useful in fields extending to organic light-emitting diodes (OLEDs). As a reliable pharmaceutical intermediate supplier, understanding and adopting such innovative methodologies is crucial for maintaining competitive advantage in the global supply chain.
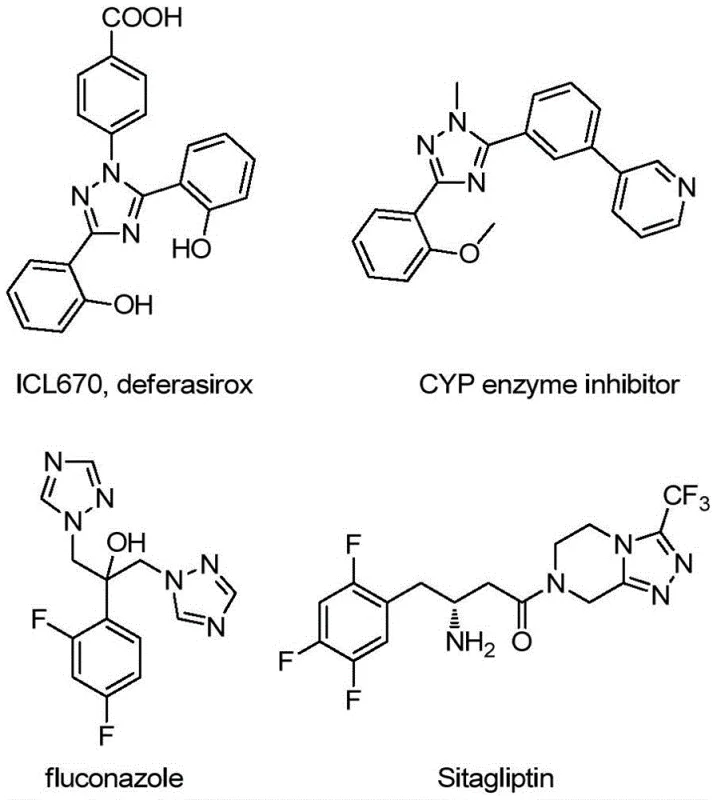
The significance of this technology lies in its ability to construct complex nitrogen-rich heterocycles with high atom economy and operational simplicity. Unlike legacy processes that rely on multi-step sequences involving expensive precursors, this invention utilizes readily available starting materials to access diverse substituted triazoles. For R&D directors and process chemists, the ability to introduce both quinolinyl and trifluoromethyl motifs simultaneously opens new avenues for drug discovery, particularly for targets requiring enhanced metabolic stability and lipophilicity. The method described offers a streamlined pathway that bypasses the bottlenecks of traditional synthesis, positioning it as a highly attractive option for the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinolyl-substituted 1,2,4-triazoles has been fraught with inefficiencies that hinder large-scale production. Conventional literature methods typically employ quinoline-2-carboxylic acid as the primary raw material. This approach is inherently flawed due to its reliance on a five-step reaction sequence, each step introducing potential yield losses and purification challenges. Under the severe reaction conditions often required for these transformations, the total yield rarely exceeds 17%, making the process economically unviable for industrial applications. Furthermore, the use of carboxylic acid derivatives often necessitates activation steps that generate stoichiometric waste, complicating downstream processing and increasing the environmental footprint. For procurement managers, these factors translate into higher costs of goods sold (COGS) and longer lead times, as the low overall yield requires significantly larger quantities of starting materials to produce the same amount of final product.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN113307790B represents a paradigm shift towards efficiency and sustainability. This novel approach utilizes cheap and easily obtained 2-methylquinoline and trifluoroacetimidohydrazide as the core starting materials. By employing a catalytic system comprising tetrabutylammonium iodide (TBAI) and tert-butyl peroxide (TBHP), the reaction promotes an oxidative cyclization that directly constructs the triazole ring. This strategy eliminates the need for pre-functionalized carboxylic acids and reduces the synthetic sequence to a single pot operation. The result is a dramatic improvement in efficiency, with reported yields reaching as high as 97% for certain substrates. This leap in performance not only enhances the economic viability of the process but also aligns with green chemistry principles by reducing waste generation and energy consumption, offering substantial cost reduction in API manufacturing.
Mechanistic Insights into TBAI/TBHP Promoted Oxidative Cyclization
The success of this transformation relies on a sophisticated yet elegant mechanistic pathway driven by the TBAI/TBHP catalytic system. The reaction initiates with the oxidation of the methyl group on the 2-methylquinoline substrate. In the presence of the iodide catalyst and peroxide oxidant, the methyl group is converted into a reactive 2-quinoline carbaldehyde equivalent in situ. This transient aldehyde species then undergoes a condensation reaction with the trifluoroacetimidohydrazide to form a dehydrated hydrazone intermediate. This step is critical as it sets the stage for the subsequent ring closure. The use of diphenylphosphoric acid as an additive further facilitates this condensation and stabilizes the intermediate species, ensuring high conversion rates even with diverse electronic substituents on the aromatic rings.
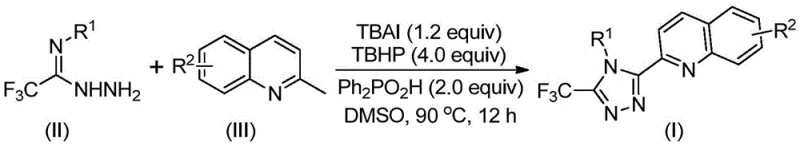
Following the formation of the hydrazone, the mechanism proceeds through an oxidative iodination and intramolecular electrophilic substitution. The iodine species, generated from the TBAI precursor, acts as an electrophile to activate the hydrazone carbon, promoting nucleophilic attack by the adjacent nitrogen atom to close the five-membered triazole ring. Finally, an aromatization step restores the aromaticity of the newly formed heterocycle, yielding the stable 3-quinolyl-5-trifluoromethyl-1,2,4-triazole product. Alternatively, the patent suggests that a free radical process may also contribute to the reaction pathway, highlighting the robustness of the oxidative conditions. This mechanistic understanding is vital for process optimization, as it explains the broad substrate tolerance observed, allowing for the introduction of electron-withdrawing or electron-donating groups without compromising the reaction outcome.
From an impurity control perspective, this mechanism offers distinct advantages. The one-pot nature of the reaction minimizes the isolation of unstable intermediates, which are often sources of difficult-to-remove impurities in multi-step syntheses. The byproducts of the oxidation, primarily tert-butanol and iodide salts, are relatively easy to separate from the organic product during the workup phase. The use of column chromatography as a standard purification technique, as described in the patent examples, ensures that the final high-purity pharmaceutical intermediates meet stringent quality specifications required for downstream drug development. The absence of transition metal catalysts like palladium or copper further simplifies the impurity profile, eliminating the need for costly and time-consuming heavy metal scavenging steps that are mandatory in many cross-coupling reactions.
How to Synthesize 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Efficiently
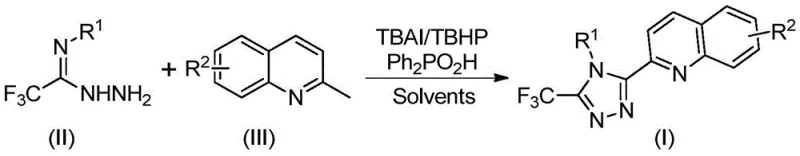
Implementing this synthesis in a laboratory or pilot plant setting is straightforward, leveraging standard equipment and commercially available reagents. The protocol involves charging a reaction vessel with the requisite amounts of tetrabutylammonium iodide, aqueous tert-butyl peroxide, diphenylphosphoric acid, trifluoroacetimidohydrazide, and the specific 2-methylquinoline derivative. The choice of solvent is critical, with dimethyl sulfoxide (DMSO) identified as the optimal medium for maximizing solubility and reaction rate. The mixture is then heated to a temperature range of 80-100°C and maintained for 8 to 14 hours. Detailed standardized synthesis steps are provided in the guide below.
- Mix tetrabutylammonium iodide (TBAI), tert-butyl peroxide (TBHP), diphenylphosphoric acid, trifluoroacetimidohydrazide, and 2-methylquinoline in DMSO solvent.
- Heat the reaction mixture to 80-100°C and stir for 8-14 hours to allow oxidative cyclization to proceed.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the target triazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement specialists, the adoption of this synthetic route offers compelling strategic benefits that extend beyond simple yield improvements. The elimination of precious metal catalysts is a primary driver for cost optimization. Traditional methods often rely on palladium or copper catalysts, which are subject to volatile market pricing and require rigorous removal protocols to meet regulatory limits for residual metals in pharmaceutical products. By utilizing an organocatalytic system based on iodine and peroxide, this process removes the burden of metal scavenging resins and the associated validation testing, leading to significantly reduced processing costs and faster batch release times.
- Cost Reduction in Manufacturing: The economic impact of this method is profound due to the use of commodity chemicals as starting materials. 2-methylquinoline and trifluoroacetic acid derivatives are produced on a massive industrial scale, ensuring a stable and low-cost supply base. The high atom efficiency of the oxidative cyclization means that less raw material is wasted, directly lowering the variable cost per kilogram of the intermediate. Furthermore, the simplified workup procedure, which involves basic filtration and chromatography, reduces the consumption of solvents and silica gel compared to multi-step extractions and recrystallizations. These factors combine to create a manufacturing process that is inherently leaner and more cost-effective, providing a buffer against raw material price fluctuations.
- Enhanced Supply Chain Reliability: Supply continuity is a critical metric for any pharmaceutical manufacturer. The robustness of this reaction conditions—specifically its tolerance to moisture and oxygen—means that it can be performed in standard reactors without the need for specialized inert atmosphere equipment or rigorously dried solvents. This operational flexibility reduces the risk of batch failures due to environmental excursions and allows for faster turnaround times between batches. Additionally, the broad substrate scope demonstrated in the patent examples indicates that the same platform technology can be used to synthesize a wide variety of analogues. This versatility allows manufacturers to respond quickly to changing demand for different drug candidates without retooling or developing entirely new processes, thereby enhancing overall supply chain agility.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the plant floor often introduces unforeseen challenges, but this method is designed with scalability in mind. The exothermic nature of the peroxide oxidation is manageable within the specified temperature range, and the use of aqueous peroxide solutions mitigates safety risks associated with handling neat oxidants. From an environmental standpoint, the avoidance of heavy metals and the use of water-compatible oxidants align with increasingly strict global environmental regulations. The waste stream is simpler to treat, and the reduced solvent usage contributes to a lower E-factor (environmental factor). This compliance readiness minimizes the regulatory hurdles for new facility approvals and supports corporate sustainability goals, making it an attractive option for long-term production contracts.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis technology. These answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing clarity for potential partners evaluating this route for their supply chain.
Q: What are the advantages of this metal-free synthesis over traditional methods?
A: Traditional methods often require quinoline-2-carboxylic acid and involve five steps with harsh conditions and low total yields (around 17%). This novel method uses cheap 2-methylquinoline and achieves yields up to 97% in a single pot without heavy metal catalysts.
Q: Does this process require strict anhydrous or oxygen-free conditions?
A: No, one of the key operational benefits of this patented process (CN113307790B) is that it does not require anhydrous or anaerobic conditions, significantly simplifying the operational requirements for large-scale manufacturing.
Q: What is the substrate scope for the R1 and R2 groups?
A: The method demonstrates excellent tolerance. R1 can be various substituted aryl groups (methyl, methoxy, halogen, trifluoromethyl), and R2 on the quinoline ring can be H, alkyl, alkoxy, halogen, or nitro groups, allowing for diverse library synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic methodology described in CN113307790B for the production of high-value heterocyclic intermediates. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering products with stringent purity specifications, utilizing our rigorous QC labs to verify that every batch meets the exacting standards required by the global pharmaceutical industry. Our infrastructure is designed to handle complex oxidative chemistries safely and efficiently, guaranteeing a consistent supply of critical building blocks for your drug development programs.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data for our catalog items or to discuss route feasibility assessments for your proprietary molecules. Let us help you optimize your supply chain and accelerate your time to market with our superior manufacturing capabilities.
