Advanced Iodine-Catalyzed Synthesis of Quinoline Derivatives for Commercial Pharmaceutical Manufacturing
Advanced Iodine-Catalyzed Synthesis of Quinoline Derivatives for Commercial Pharmaceutical Manufacturing
The pharmaceutical and fine chemical industries continuously seek robust, scalable, and cost-effective methodologies for constructing privileged scaffolds such as the quinoline nucleus. Patent CN102060761B introduces a transformative approach to synthesizing 2,4-disubstituted quinoline derivatives through an iodine-catalyzed multicomponent reaction. This technology leverages readily available aromatic amines, aldehydes, and terminal alkynes to generate high-value heterocyclic intermediates under mild reflux conditions. Unlike traditional methods that often rely on harsh acidic environments or expensive transition metal catalysts, this protocol utilizes molecular iodine in nitromethane, offering a distinct advantage in terms of operational simplicity and environmental compatibility. For R&D directors and procurement specialists, this represents a significant opportunity to optimize supply chains for antimalarial agents, anti-tuberculosis drugs, and other bioactive compounds where the quinoline core is essential.
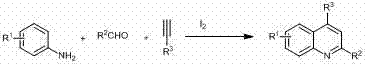
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinoline ring system has relied heavily on classical methodologies such as the Skraup synthesis, which typically necessitates the use of strong mineral acids like sulfuric acid and potent oxidants under vigorous heating conditions. These harsh parameters often lead to poor functional group tolerance, resulting in significant byproduct formation and challenging purification workflows that drive up manufacturing costs. Furthermore, more modern approaches utilizing transition metal catalysis, specifically involving gold, copper, or iron complexes, while effective, introduce substantial economic and regulatory burdens. The reliance on precious metals not only inflates the raw material expenditure but also mandates rigorous downstream processing to ensure residual metal levels comply with stringent ICH Q3D guidelines for pharmaceutical ingredients. Consequently, manufacturers face extended lead times and increased waste generation due to the necessity of metal scavenging resins and additional chromatographic steps.
The Novel Approach
The methodology disclosed in CN102060761B circumvents these traditional bottlenecks by employing molecular iodine as a benign, inexpensive, and highly efficient catalyst. This metal-free strategy operates under neutral to mildly acidic conditions generated in situ, thereby preserving sensitive functional groups that would otherwise degrade under classical Skraup conditions. The reaction proceeds smoothly in nitromethane, a solvent that facilitates the necessary solubility and thermal stability for the multicomponent coupling. By eliminating the dependency on transition metals, this novel approach inherently simplifies the post-reaction workup; the catalyst can be easily quenched and removed using standard aqueous sodium thiosulfate washes. This shift from complex metal catalysis to simple halogen catalysis translates directly into reduced operational complexity, lower capital expenditure on specialized removal equipment, and a drastically simplified impurity profile, making it an ideal candidate for reliable pharmaceutical intermediate supplier networks aiming for lean manufacturing.
Mechanistic Insights into Iodine-Catalyzed Multicomponent Cyclization
The mechanistic pathway of this transformation is a sophisticated yet elegant sequence of condensation and cycloaddition events initiated by the activation of the carbonyl component. Initially, the aromatic amine condenses with the aldehyde to form an imine intermediate, a process that is facilitated by the Lewis acidic nature of the iodine catalyst. This imine species then acts as a dipole or reactive partner in a subsequent [2+4] cycloaddition with the terminal alkyne. The iodine catalyst plays a dual role here: it activates the imine towards nucleophilic attack and potentially activates the alkyne through halogen bonding interactions, lowering the energy barrier for the cyclization step. The resulting dihydroquinoline intermediate is not the final product; rather, it undergoes a spontaneous or air-mediated oxidation step to restore aromaticity, yielding the fully conjugated quinoline system. This oxidative aromatization is crucial and occurs efficiently without the need for stoichiometric external oxidants, further enhancing the atom economy of the process.
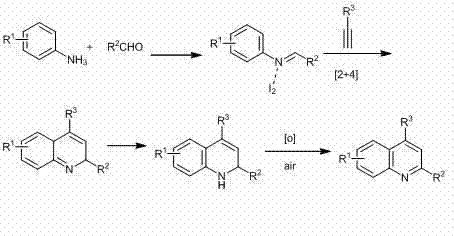
From an impurity control perspective, the mechanism offers inherent selectivity that benefits process chemistry teams. Because the reaction relies on the specific electronic interplay between the imine and the alkyne activated by iodine, side reactions such as polymerization of the alkyne or over-oxidation of the amine are minimized. The use of nitromethane as a solvent provides a polar environment that stabilizes the charged intermediates involved in the cyclization, preventing the formation of oligomeric byproducts common in less polar media. Furthermore, the mildness of the iodine catalyst ensures that sensitive substituents on the aromatic rings, such as methoxy or halo groups, remain intact throughout the reaction duration of 8 to 15 hours. This high level of chemoselectivity ensures that the crude reaction mixture contains predominantly the desired quinoline derivative, significantly reducing the burden on downstream purification units and allowing for high-purity quinoline derivatives to be isolated via simple recrystallization techniques.
How to Synthesize 2,4-Disubstituted Quinolines Efficiently
The practical execution of this synthesis is designed for maximum operational ease, requiring only standard laboratory glassware and heating mantles capable of maintaining reflux temperatures. The process begins with the precise metering of aromatic amines, aldehydes, and terminal alkynes into nitromethane, followed by the addition of catalytic amounts of molecular iodine. The reaction mixture is then heated to reflux, typically maintained for a period ranging from 8 to 15 hours depending on the specific steric and electronic nature of the substrates employed. Upon completion, the reaction is quenched not with complex reagents but with a simple aqueous sodium thiosulfate solution to reduce any residual iodine, followed by standard aqueous washing and drying procedures. The detailed standardized synthesis steps see the guide below.
- Mix aromatic amine, aldehyde, and nitromethane solvent, then add terminal alkyne and molecular iodine catalyst.
- Heat the reaction mixture to reflux conditions for 8 to 15 hours to facilitate the multicomponent cyclization.
- Perform workup by washing with sodium thiosulfate and water, drying the organic phase, and purifying via recrystallization or distillation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iodine-catalyzed protocol offers profound strategic advantages rooted in cost efficiency and logistical simplicity. The elimination of precious metal catalysts removes a major variable cost driver and mitigates the supply risk associated with fluctuating prices of commodities like gold or palladium. Additionally, the simplicity of the workup procedure, which avoids column chromatography in favor of crystallization or distillation, allows for faster batch turnover times and higher throughput in existing manufacturing facilities. This process intensification capability means that production capacity can be effectively increased without significant capital investment in new infrastructure, directly addressing the need for cost reduction in fine chemical manufacturing.
- Cost Reduction in Manufacturing: The substitution of expensive transition metal catalysts with commodity-grade molecular iodine results in a drastic reduction in direct material costs. Since iodine is used in catalytic quantities (0.05 to 0.2 equivalents) and is inexpensive compared to organometallic complexes, the overall cost of goods sold (COGS) is significantly lowered. Furthermore, the removal of heavy metal scavenging steps eliminates the need for costly resin beds and the associated waste disposal fees, leading to substantial cost savings in the overall production budget. The high yields reported, reaching up to 82% in optimized examples, ensure that raw material utilization is maximized, further enhancing the economic viability of the process for large-scale operations.
- Enhanced Supply Chain Reliability: The reliance on universally available starting materials such as anilines, benzaldehydes, and phenylacetylenes ensures a robust and resilient supply chain that is not subject to the geopolitical constraints often affecting rare earth or precious metal supplies. The use of nitromethane, a common industrial solvent, further simplifies logistics and inventory management. By adopting a synthesis route that utilizes bulk chemicals with established global supply networks, manufacturers can significantly reduce lead time for high-purity intermediates and ensure continuous production schedules even during periods of market volatility. This reliability is critical for maintaining the continuity of supply for downstream API manufacturing.
- Scalability and Environmental Compliance: The operational simplicity of running a reflux reaction in a single pot makes this technology exceptionally amenable to commercial scale-up of complex heterocycles. The absence of toxic heavy metals simplifies the environmental compliance landscape, reducing the burden of wastewater treatment and hazardous waste disposal. The process generates minimal hazardous waste compared to traditional methods, aligning with green chemistry principles and corporate sustainability goals. This ease of scale-up from gram to kilogram and ton scales ensures that the technology can seamlessly transition from R&D pilot plants to full commercial production without the need for extensive re-engineering of the process parameters.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this iodine-catalyzed quinoline synthesis technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent literature to provide clarity for potential partners and technical stakeholders.
Q: What are the primary advantages of using iodine over transition metals for quinoline synthesis?
A: Iodine catalysis eliminates the need for expensive and toxic transition metals like gold or copper, significantly reducing raw material costs and simplifying the purification process by removing the requirement for heavy metal scavenging steps.
Q: What represents the typical yield range for this iodine-catalyzed protocol?
A: According to the experimental data in patent CN102060761B, the reaction yields vary depending on the substrate but generally range from 60% to 82%, demonstrating robust efficiency across different electronic environments.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the method utilizes simple reflux conditions in nitromethane and straightforward aqueous workups, making it highly scalable and operationally convenient for commercial manufacturing without requiring specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinoline Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in the competitive landscape of pharmaceutical intermediate production. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising laboratory results of patent CN102060761B can be successfully translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of quinoline derivatives meets the highest quality standards required by global regulatory bodies. Our commitment to process excellence allows us to deliver consistent quality while optimizing the cost structure for our clients.
We invite you to collaborate with us to leverage this advanced iodine-catalyzed technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and target specifications. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in heterocyclic chemistry can accelerate your development timelines and enhance your supply chain efficiency.
