Advanced Organocatalytic Route for High-Purity Tetrahydroquinoline Derivatives in Drug Discovery
Advanced Organocatalytic Route for High-Purity Tetrahydroquinoline Derivatives in Drug Discovery
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for greener, more efficient, and highly stereoselective synthetic routes. A significant breakthrough in this domain is documented in Chinese Patent CN102766092A, which discloses a novel method for synthesizing optically active tetrahydroquinoline derivatives using chiral spiro phosphoric acid as a catalyst. Tetrahydroquinoline scaffolds are ubiquitous in medicinal chemistry, serving as critical cores for numerous bioactive compounds and natural products. Traditional methods often struggle with achieving high enantiomeric purity or require harsh reaction conditions that limit substrate scope. This patent introduces a robust organocatalytic strategy that operates under remarkably mild conditions, typically between -30°C and 50°C, utilizing a three-component coupling of aldehydes, aromatic amines, and N-benzyloxycarbonyl ethylene amine. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this technology represents a paradigm shift towards more sustainable and cost-effective production of complex nitrogen-containing heterocycles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of optically active tetrahydroquinolines has relied heavily on the asymmetric Povarov reaction or transition-metal catalyzed cyclizations. While effective in certain contexts, these conventional methodologies present significant drawbacks for large-scale manufacturing. Many traditional protocols necessitate the use of expensive and toxic transition metals, such as copper or palladium complexes, which introduce severe regulatory hurdles regarding residual metal limits in final drug substances. Furthermore, these metal-catalyzed routes often require stringent anhydrous conditions, cryogenic temperatures well below -78°C, or prolonged reaction times that hinder throughput. The substrate scope is frequently narrow, particularly when aliphatic aldehydes are employed, leading to poor yields and disappointing enantioselectivity. Additionally, the separation of diastereomers and enantiomers from crude reaction mixtures often demands resource-intensive chromatographic processes, driving up the cost of goods and complicating the supply chain for high-purity pharmaceutical intermediates.
The Novel Approach
The methodology outlined in CN102766092A offers a compelling alternative by leveraging chiral spiro phosphoric acids as powerful Brønsted acid organocatalysts. This approach fundamentally simplifies the synthetic workflow by enabling a direct three-component condensation that proceeds with exceptional stereocontrol. As illustrated in the general reaction scheme below, the process combines an aldehyde, an arylamine, and an N-protected vinylamine in a single pot. 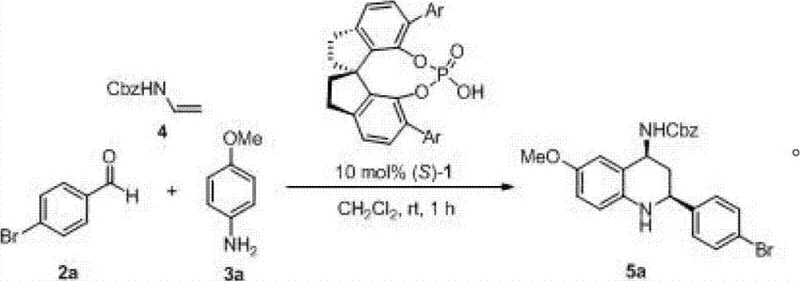 The beauty of this system lies in its operational simplicity and broad tolerance. Unlike metal-based systems, this organocatalytic route functions efficiently in common organic solvents like dichloromethane, chloroform, or toluene, without the need for rigorous exclusion of moisture beyond standard nitrogen protection. The reaction times are notably short, typically completing within 1 to 5 hours, which significantly enhances reactor turnover rates. Most critically, the method demonstrates remarkable versatility, successfully accommodating both aromatic and aliphatic aldehydes while maintaining high diastereoselectivity (cis:trans > 20:1) and enantiomeric excess. This universality makes it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates where diverse substitution patterns are required.
The beauty of this system lies in its operational simplicity and broad tolerance. Unlike metal-based systems, this organocatalytic route functions efficiently in common organic solvents like dichloromethane, chloroform, or toluene, without the need for rigorous exclusion of moisture beyond standard nitrogen protection. The reaction times are notably short, typically completing within 1 to 5 hours, which significantly enhances reactor turnover rates. Most critically, the method demonstrates remarkable versatility, successfully accommodating both aromatic and aliphatic aldehydes while maintaining high diastereoselectivity (cis:trans > 20:1) and enantiomeric excess. This universality makes it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates where diverse substitution patterns are required.
Mechanistic Insights into Chiral Spiro Phosphoric Acid Catalysis
To fully appreciate the value of this technology for process chemistry, one must understand the mechanistic underpinnings of the chiral spiro phosphoric acid catalyst. These catalysts, characterized by their rigid binaphthyl-derived spiro backbone, act as chiral Brønsted acids. The core of the mechanism involves the dual activation of the reactants through a network of hydrogen bonds. The phosphoryl oxygen of the catalyst acts as a hydrogen bond acceptor to activate the imine intermediate formed in situ from the aldehyde and amine, while the acidic proton of the P-OH group activates the nucleophilic enecarbamate. This simultaneous activation organizes the transition state within a well-defined chiral pocket created by the bulky substituents on the spiro framework. 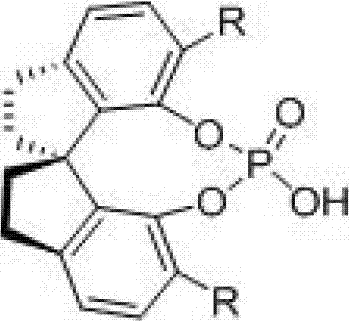 The rigidity of the spiro structure is paramount; it restricts conformational freedom, ensuring that the nucleophilic attack occurs from a specific face of the imine, thereby dictating the absolute configuration of the newly formed stereocenters. This precise spatial control is what allows the reaction to achieve enantiomeric excess values often exceeding 90%, and in optimized cases reaching >99% ee. For the R&D team, understanding this mechanism is crucial for troubleshooting and optimizing the process, as slight modifications to the R groups on the catalyst (such as introducing electron-withdrawing trifluoromethyl groups or bulky naphthyl groups) can fine-tune the acidity and steric environment to match specific challenging substrates.
The rigidity of the spiro structure is paramount; it restricts conformational freedom, ensuring that the nucleophilic attack occurs from a specific face of the imine, thereby dictating the absolute configuration of the newly formed stereocenters. This precise spatial control is what allows the reaction to achieve enantiomeric excess values often exceeding 90%, and in optimized cases reaching >99% ee. For the R&D team, understanding this mechanism is crucial for troubleshooting and optimizing the process, as slight modifications to the R groups on the catalyst (such as introducing electron-withdrawing trifluoromethyl groups or bulky naphthyl groups) can fine-tune the acidity and steric environment to match specific challenging substrates.
Furthermore, the mechanism inherently suppresses the formation of unwanted byproducts and trans-isomers. The tight ion-pairing between the chiral phosphate anion and the protonated iminium cation prevents the free rotation that often leads to racemization or isomerization in solution. This results in a crude product profile that is exceptionally clean, dominated by the desired cis-isomer. From a purification standpoint, this is a massive advantage. High diastereoselectivity means that the difficult separation of cis/trans isomers is largely obviated, allowing for simpler crystallization or flash chromatography protocols. This purity profile is essential for meeting the stringent specifications required for API intermediates, reducing the burden on downstream processing and quality control laboratories.
How to Synthesize Optically Active Tetrahydroquinoline Derivatives Efficiently
Implementing this synthesis in a laboratory or pilot plant setting is straightforward, adhering to standard organic synthesis safety protocols. The process begins with the preparation of the reaction vessel under an inert atmosphere to prevent oxidation of sensitive amine components. The stoichiometry is flexible, typically employing a molar ratio of aldehyde to arylamine to N-Cbz-vinylamine of 1:1:1 to 1:1.2, ensuring complete consumption of the limiting reagent. The catalyst loading is economically viable, ranging from 1 mol% to 15 mol%, allowing process chemists to balance cost against reaction rate. Detailed standardized operating procedures for mixing, temperature control, and workup are essential for reproducibility. For a comprehensive guide on the specific execution of this protocol, please refer to the technical steps outlined below.
- Mix aldehyde, aromatic amine, and N-benzyloxycarbonyl ethylene amine in an organic solvent such as dichloromethane under nitrogen protection.
- Add the chiral spiro phosphoric acid catalyst (1-15 mol%) to the reaction mixture at temperatures ranging from -30 to 50°C.
- Stir the reaction for 1 to 5 hours, monitor by TLC, and purify the resulting optically active tetrahydroquinoline derivative via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this organocatalytic technology translates directly into tangible operational efficiencies and risk mitigation. The shift from metal-based catalysis to organocatalysis removes a significant bottleneck in the supply chain: the sourcing and handling of precious metals. By eliminating the need for expensive transition metal catalysts, manufacturers can drastically simplify their raw material inventory and reduce exposure to volatile metal markets. Moreover, the absence of heavy metals in the reaction mixture means that the costly and time-consuming metal scavenging steps—often required to meet ppm-level specifications for pharmaceutical ingredients—are completely unnecessary. This streamlining of the downstream process leads to substantial cost savings in terms of both materials and processing time, enhancing the overall margin profile of the final intermediate.
- Cost Reduction in Manufacturing: The economic benefits of this process are multifaceted, stemming primarily from the simplicity of the reaction conditions and the efficiency of the catalyst. Since the reaction proceeds at mild temperatures ranging from -30°C to 50°C, there is no need for energy-intensive cryogenic cooling systems that are often required for traditional asymmetric syntheses. This reduction in energy consumption directly lowers utility costs. Additionally, the high selectivity of the reaction minimizes the formation of waste byproducts, improving the atom economy and reducing the volume of solvent and silica gel required for purification. The ability to use common, inexpensive solvents like dichloromethane further contributes to cost reduction in API manufacturing, making the process financially attractive for large-scale production runs.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness and generality of this synthetic method. The catalyst system tolerates a wide variety of functional groups, including halogens, nitro groups, and ethers, meaning that a single catalytic platform can be used to produce a diverse library of tetrahydroquinoline derivatives. This versatility reduces the need for developing unique, bespoke synthetic routes for each new analog, accelerating the timeline from discovery to commercial supply. Furthermore, the starting materials—aldehydes, anilines, and protected vinylamines—are commodity chemicals with stable global supply chains. This ensures consistent availability of raw materials and reduces the lead time for high-purity pharmaceutical intermediates, allowing manufacturers to respond quickly to market demands.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the plant floor often reveals hidden complexities, but this organocatalytic route is inherently scalable. The reaction does not generate hazardous gaseous byproducts, and the exotherm is manageable under the specified conditions, facilitating safe scale-up to multi-kilogram or tonne scales. From an environmental perspective, the elimination of toxic heavy metals aligns perfectly with green chemistry principles and increasingly strict environmental regulations. The reduced waste generation and lower energy footprint contribute to a more sustainable manufacturing profile. This compliance with environmental standards not only mitigates regulatory risk but also enhances the corporate social responsibility profile of the supply chain, a factor of growing importance to global pharmaceutical partners.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of any new synthetic methodology is critical for successful implementation. The following questions address common inquiries regarding the practical application, scope, and performance of this chiral spiro phosphoric acid catalyzed synthesis. These answers are derived directly from the experimental data and technical specifications provided in the patent literature, ensuring accuracy and relevance for technical decision-makers evaluating this technology for their own pipelines.
Q: What are the key advantages of using chiral spiro phosphoric acid over traditional metal catalysts?
A: Chiral spiro phosphoric acids operate under mild conditions (-30 to 50°C) without requiring toxic heavy metals, eliminating the need for expensive metal scavenging steps and ensuring higher purity profiles suitable for pharmaceutical applications.
Q: What is the typical enantiomeric excess (ee) achieved with this method?
A: The patented method consistently achieves high enantioselectivity, often exceeding 90% ee and reaching up to >99% ee depending on the substrate, with a strong preference for the cis-isomer (cis:trans > 20:1).
Q: Is this process scalable for commercial production of API intermediates?
A: Yes, the process utilizes common organic solvents like dichloromethane and operates at near-ambient temperatures, making it highly adaptable for scale-up from laboratory grams to multi-kilogram commercial batches without complex cryogenic requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrahydroquinoline Derivatives Supplier
The technological potential of chiral spiro phosphoric acid catalysis is immense, offering a pathway to high-value chiral building blocks that are essential for modern drug discovery. At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating such innovative academic and patent technologies into robust, commercial-grade manufacturing processes. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with state-of-the-art reactors capable of maintaining the precise temperature controls (-30°C to 50°C) required for this chemistry, and our rigorous QC labs ensure that every batch meets stringent purity specifications, including enantiomeric excess and residual solvent limits.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this advanced synthetic route for your next project. Whether you require custom synthesis of specific tetrahydroquinoline analogs or technology transfer for in-house production, our team is ready to assist. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific molecule. We are prepared to provide specific COA data from pilot runs and detailed route feasibility assessments to demonstrate how this method can optimize your supply chain and reduce overall manufacturing costs.
