Revolutionizing Aromatic Reduction: Metal-Free Photo-Induced Synthesis for Commercial Scale-Up
Revolutionizing Aromatic Reduction: Metal-Free Photo-Induced Synthesis for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more efficient, sustainable, and cost-effective methodologies for constructing core molecular scaffolds. A significant breakthrough in this domain is documented in patent CN113354498A, which discloses a novel method for reducing aromatic C-N, C-O, C-Cl, C-Br, and C-I bonds directly into aromatic C-H or C-D bonds. This technology represents a paradigm shift away from traditional transition-metal catalysis and hazardous diazonium chemistry, offering a robust platform for the synthesis of high-purity pharmaceutical intermediates and deuterated compounds. By leveraging simple light induction—using either ultraviolet or visible light—in the presence of inexpensive bases and common solvents, this process achieves high yields and exceptional deuteration rates without the need for precious metal catalysts or explosive intermediates. For R&D directors and procurement managers alike, this innovation promises to streamline supply chains and reduce the environmental footprint of complex aromatic synthesis.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the conversion of aromatic heteroatoms into hydrogen or deuterium has been fraught with significant technical and safety challenges that hinder efficient commercial production. Traditional methods for aromatic C-N bond reduction often necessitate the formation of diazonium salt intermediates, which are notoriously unstable, sensitive to temperature fluctuations, and pose severe explosion risks during handling and storage, thereby complicating safety protocols in manufacturing facilities. Furthermore, conventional reduction strategies for C-O and C-halogen bonds frequently rely on expensive transition metal catalysts such as palladium or platinum, which not only inflate raw material costs but also introduce the risk of toxic metal residues in the final active pharmaceutical ingredients (APIs), requiring costly and time-consuming purification steps to meet stringent regulatory limits. Additionally, existing deuteration methods often suffer from poor site-selectivity, leading to multi-site labeling that fails to meet the precise metabolic stability requirements of modern deuterated drug development, while also typically requiring harsh acidic or basic conditions that limit functional group tolerance.
The Novel Approach
In stark contrast to these legacy technologies, the method disclosed in patent CN113354498A utilizes a groundbreaking photo-induced strategy that completely eliminates the need for transition metal catalysts, photosensitizers, or hazardous reducing agents. This novel approach employs stable and easily accessible starting materials, such as aromatic quaternary ammonium salts, aromatic triflates, and various aromatic halides, which are subjected to light irradiation in the presence of mild inorganic or organic bases like sodium carbonate or DMAP. The reaction proceeds efficiently under mild thermal conditions ranging from 0°C to 50°C in common solvents including THF, DMF, or methanol, offering a remarkably green and environmentally friendly profile. By avoiding the generation of explosive intermediates and removing the dependency on scarce precious metals, this technology provides a safer, more economical, and operationally simpler route for the large-scale production of aromatic cores and their deuterated analogues, addressing the critical pain points of both safety and cost in chemical manufacturing.
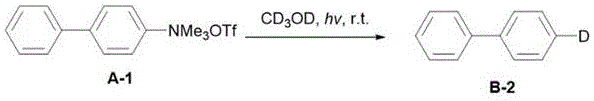
Mechanistic Insights into Photo-Induced Radical Cleavage and Deuteration
The underlying chemical mechanism of this transformative process involves the direct homolytic cleavage of the aromatic carbon-heteroatom bond induced by photon energy, generating highly reactive aromatic radical intermediates without the mediation of a metal center. Upon irradiation with ultraviolet or visible light, the stable aromatic precursor—whether it be a quaternary ammonium salt, a triflate, or a halide—undergoes excitation that facilitates the fragmentation of the C-N, C-O, or C-X bond, releasing the heteroatom leaving group and forming a transient aryl radical species. This radical intermediate is then rapidly quenched through a hydrogen atom transfer (HAT) or deuterium atom transfer (DAT) process from the surrounding solvent molecules, such as methanol or deuterated methanol, effectively capping the radical site to form the desired C-H or C-D bond. The presence of a base in the reaction mixture plays a crucial role in facilitating this transformation, potentially by stabilizing intermediates or assisting in the proton/deuteron transfer steps, ensuring high reaction efficiency and selectivity even in complex molecular environments.
From an impurity control perspective, this radical-based mechanism offers distinct advantages over ionic or metal-catalyzed pathways, primarily due to the absence of metal-ligand complexes that can lead to difficult-to-remove organometallic byproducts. The simplicity of the reagent system—comprising only the substrate, a benign base, and a solvent—minimizes the formation of side products associated with catalyst decomposition or ligand exchange reactions, resulting in a cleaner crude reaction profile that simplifies downstream purification. Furthermore, the high specificity of the light-induced bond cleavage allows for precise site-selective modification, enabling the targeted deuteration of specific positions on the aromatic ring without affecting other sensitive functional groups, which is critical for maintaining the biological activity and metabolic profile of the final drug candidate. This mechanistic clarity ensures that the process is not only chemically robust but also highly predictable and scalable for industrial applications.
How to Synthesize Deuterated Aromatic Compounds Efficiently
The synthesis of deuterated aromatic compounds using this photo-induced protocol is designed to be straightforward and adaptable to standard laboratory and pilot plant equipment, requiring minimal specialized infrastructure beyond a suitable light source. The general procedure involves charging a reaction vessel, preferably made of quartz to ensure optimal light transmission, with the aromatic substrate and a stoichiometric amount of a mild base such as sodium carbonate or sodium phosphate. Following the addition of the deuterated solvent, typically CD3OD or deuterated acetone depending on solubility and reactivity requirements, the mixture is stirred under an inert atmosphere or in air while being irradiated with a UV or visible light source at controlled temperatures. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by adding the aromatic substrate (quaternary ammonium salt, triflate, or halide) and a suitable base such as sodium carbonate or DMAP into a quartz reaction vessel.
- Add the appropriate solvent, utilizing deuterated methanol (CD3OD) or deuterated acetone for deuteration purposes, or standard organic solvents like THF for hydrogenation.
- Irradiate the mixture with ultraviolet or visible light at temperatures between 0-50°C until the reaction is complete, followed by standard purification via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this metal-free photo-chemical technology offers profound strategic advantages that directly impact the bottom line and operational resilience of pharmaceutical manufacturing. By eliminating the requirement for expensive transition metal catalysts like palladium or rhodium, companies can achieve substantial cost savings in raw material procurement while simultaneously removing the logistical complexities associated with sourcing and managing precious metal inventories. The use of stable, non-explosive starting materials such as aromatic halides and quaternary ammonium salts significantly enhances supply chain reliability, as these commodities are widely available from multiple global suppliers and do not require the specialized hazardous material handling protocols mandated for diazonium salts, thereby reducing insurance and compliance costs.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the complete removal of precious metal catalysts and the avoidance of complex ligand systems, which traditionally account for a significant portion of the cost of goods sold in aromatic functionalization reactions. Additionally, the simplified work-up procedure, which does not require extensive metal scavenging steps or rigorous filtration to remove catalyst residues, leads to reduced processing time and lower consumption of purification media, further driving down the overall manufacturing cost per kilogram of the final intermediate. The ability to use inexpensive and readily available bases like sodium carbonate instead of strong, corrosive alkali metals also contributes to a safer and more cost-effective operational environment, minimizing waste disposal costs associated with hazardous chemical neutralization.
- Enhanced Supply Chain Reliability: The reliance on stable and commodity-grade starting materials ensures a robust and uninterrupted supply chain, mitigating the risks associated with the volatility of the precious metals market or the scarcity of specialized reagents. Since the reaction can be performed in common organic solvents and does not require strictly anhydrous or oxygen-free conditions in many cases, the logistical burden on the supply chain is significantly reduced, allowing for more flexible sourcing strategies and inventory management. This stability is particularly valuable for long-term production contracts where consistency of supply and quality is paramount, as the process is less susceptible to batch-to-batch variations caused by catalyst degradation or sensitivity to trace impurities in reagents.
- Scalability and Environmental Compliance: From a sustainability and scalability standpoint, this photo-induced method aligns perfectly with modern green chemistry principles, generating minimal hazardous waste and avoiding the discharge of heavy metals into the environment, which simplifies regulatory compliance and wastewater treatment processes. The reaction conditions are mild and easily controllable, facilitating seamless scale-up from laboratory gram-scale experiments to multi-ton commercial production without the need for complex pressure vessels or extreme temperature controls. The high atom economy and the potential for using continuous flow photo-reactors further enhance the scalability of the process, offering a future-proof manufacturing solution that can adapt to increasing market demands for deuterated drugs and complex aromatic intermediates while maintaining a low environmental footprint.
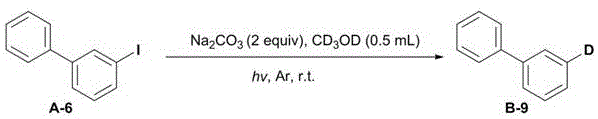
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photo-induced reduction technology, based on the specific data and embodiments provided in the patent literature. These insights are intended to clarify the operational parameters and potential benefits for stakeholders evaluating this method for their own production pipelines.
Q: Does this photo-induced reduction method require expensive transition metal catalysts?
A: No, the method described in patent CN113354498A operates without any transition metal catalysts or photosensitizers, relying solely on light induction and inexpensive bases, which significantly reduces raw material costs and eliminates heavy metal residue concerns.
Q: What is the deuteration efficiency achievable with this protocol?
A: The patent data demonstrates exceptionally high deuteration rates, often exceeding 99%, when using deuterated solvents like CD3OD, making it highly suitable for the synthesis of deuterated drug candidates where isotopic purity is critical.
Q: Is this process safe for large-scale manufacturing compared to traditional diazonium methods?
A: Yes, unlike traditional methods that require unstable and potentially explosive diazonium salt intermediates, this process uses stable aromatic quaternary ammonium salts or halides under mild conditions (0-50°C), drastically improving operational safety for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Deuterated Aromatic Compounds Supplier
As the demand for deuterated drugs and high-purity aromatic intermediates continues to surge, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM provides a strategic advantage in navigating the complexities of modern chemical synthesis. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the innovative photo-induced reduction methods described in patent CN113354498A can be seamlessly translated from the bench to the plant. Our state-of-the-art facilities are equipped with advanced photo-reactors and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of deuterated compound or aromatic intermediate we deliver meets the highest standards of quality and isotopic enrichment required by global regulatory agencies.
We invite you to collaborate with our technical team to explore how this metal-free technology can optimize your specific project requirements and drive down your overall development costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your target molecule, along with specific COA data and route feasibility assessments that demonstrate the viability of this cutting-edge synthetic approach for your supply chain.
