Advanced One-Pot Synthesis of Deuterated Aromatics for Commercial Scale-Up and Drug Development
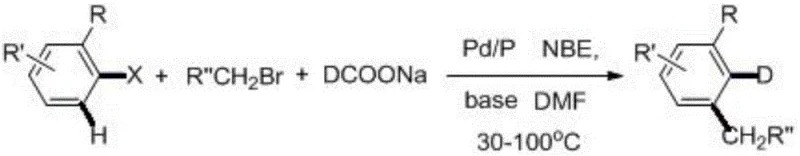
The pharmaceutical and advanced materials industries are increasingly recognizing the critical value of deuterium substitution in molecular design, a trend solidified by the FDA approval of deuterated drugs like Austedo. Patent CN110054541B, published in May 2022, introduces a groundbreaking preparation method for deuterated aromatic compounds that addresses long-standing synthetic challenges. This technology utilizes a sophisticated one-pot strategy that seamlessly combines palladium-catalyzed carbon-halogen bond reduction with participation of deuterium anions and a palladium-catalyzed coupling reaction involving bromides. By leveraging simple halogenated aromatic hydrocarbons and brominated alkanes as starting raw materials, alongside sodium deuterated formate as the deuterium source, this method achieves exceptional efficiency. The reaction proceeds in N,N-dimethylformamide (DMF) solvent using a metal palladium complex catalyst and organic phosphine ligands, fully reacting at moderate temperatures of 30-100°C under inert gas protection. This innovation represents a significant leap forward for any organization seeking a reliable deuterated aromatic compound supplier, as it bypasses the cumbersome multi-step syntheses traditionally required to install deuterium atoms at specific metabolic sites or within optoelectronic frameworks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of deuterium atoms into aromatic systems has been plagued by significant technical and economic hurdles that hinder efficient cost reduction in pharmaceutical intermediates manufacturing. The first conventional approach, hydrogen-deuterium exchange, while atom-economical in theory, suffers from poor regioselectivity and low deuteration rates, often requiring specific positioning groups on the substrate that limit its applicability to complex molecules. The second common method, dehalogenation deuteration, offers better site selectivity but necessitates the pre-introduction of a halogen leaving group, adding extra synthetic steps and generating additional waste streams before the actual deuteration can even occur. Furthermore, traditional methods frequently rely on expensive deuterated solvents acting as both the reaction medium and the deuterium source, which drastically inflates the production cost and makes large-scale commercialization economically unviable. These limitations create a bottleneck for R&D teams who require high-purity deuterated standards for metabolic profiling or for the development of next-generation OLED materials with enhanced stability.
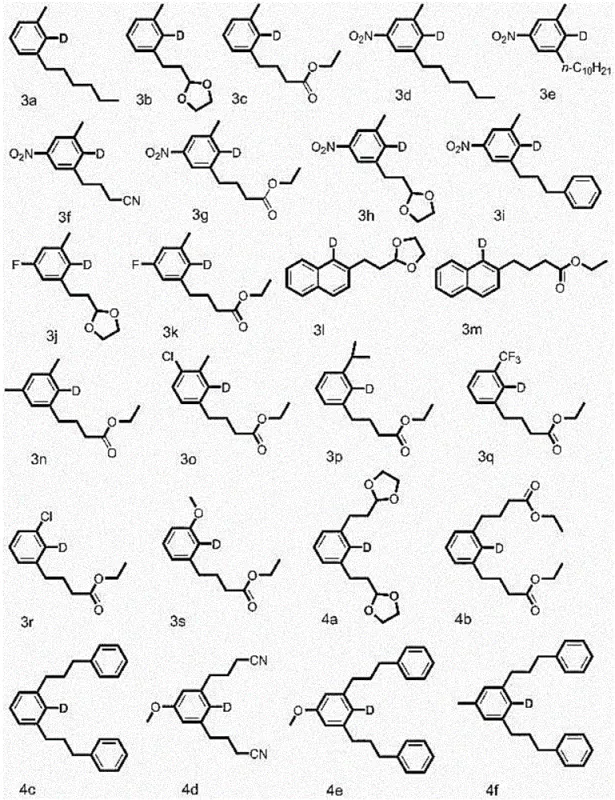
The Novel Approach
The methodology disclosed in CN110054541B fundamentally disrupts this status quo by enabling the direct synthesis of complex polysubstituted deuterated aromatic compounds from simple precursors in a single operational step. This novel approach creatively merges the reduction of carbon-halogen bonds with the coupling of alkyl bromides, facilitated by a palladium catalyst system that includes norbornene (NBE) as a crucial mediator. The versatility of this system is demonstrated by its tolerance to a wide range of functional groups, including nitro, cyano, ester, and ether moieties, as well as various halogen substituents on the aromatic ring. By avoiding the use of expensive deuterated reagents as solvents and instead utilizing sodium deuteroformate, the process achieves a deuteration rate of the target product exceeding 98 percent while maintaining a lean cost structure. The broad substrate scope allows for the creation of diverse molecular architectures, ranging from simple alkyl-substituted benzenes to complex naphthalene derivatives, providing a robust platform for the commercial scale-up of complex deuterated intermediates.
Mechanistic Insights into Palladium-Catalyzed Dual Functionalization
The core of this technological breakthrough lies in the intricate catalytic cycle that orchestrates both the installation of the alkyl chain and the deuterium atom simultaneously. The reaction initiates with the oxidative addition of the palladium(0) species into the carbon-halogen bond of the aromatic substrate, forming an aryl-palladium intermediate. In the presence of norbornene (NBE), this intermediate undergoes migratory insertion, a key step that activates the ortho-position of the aromatic ring, allowing for subsequent functionalization. The sodium deuteroformate serves a dual purpose: it acts as a base to facilitate the reaction and, more critically, as the source of deuterium anions that participate in the reductive elimination or transmetallation steps to install the deuterium label. The organic phosphine ligands stabilize the palladium center throughout this dynamic cycle, preventing catalyst deactivation and ensuring high turnover numbers. This mechanistic pathway ensures that the deuterium is incorporated with high fidelity at the desired position, minimizing isotopic scrambling which is a common issue in acid-catalyzed exchange methods.
From an impurity control perspective, this mechanism offers distinct advantages over traditional routes by minimizing side reactions associated with harsh acidic or basic conditions. The use of mild bases such as potassium phosphate or potassium acetate, combined with the moderate temperature range of 30-100°C, suppresses the formation of homocoupling byproducts and debromination impurities that often plague radical-based deuteration strategies. The high selectivity of the palladium-norbornene system ensures that the alkyl bromide couples exclusively at the intended site, while the deuterium source is efficiently utilized without excessive consumption. This precision is vital for regulatory compliance in pharmaceutical manufacturing, where the impurity profile of a drug substance must be strictly controlled. The ability to achieve greater than 98 percent deuteration purity directly from the reactor reduces the burden on downstream purification processes, thereby enhancing the overall yield and quality of the final high-purity deuterated aromatic compound.
How to Synthesize Deuterated Aromatic Compounds Efficiently
Implementing this synthesis route requires careful attention to the stoichiometry of the deuterium source and the selection of the appropriate palladium ligand system to maximize yield. The patent data indicates that a molar ratio of halogenated aromatic hydrocarbon to deuterated sodium formate between 1:1 and 1:5 is optimal, with a catalyst loading of 2% to 5% relative to the substrate. The reaction is typically conducted in polar aprotic solvents like DMF or DMSO, which facilitate the solubility of the inorganic salts and the organometallic intermediates. For R&D teams looking to replicate this success, the detailed standardized synthesis steps provided below outline the precise order of addition and workup procedures necessary to achieve the reported yields of up to 81 percent in model systems.
- Combine halogenated aromatic hydrocarbon, brominated alkane, sodium deuteroformate, palladium catalyst, organic phosphine ligand, and norbornene in DMF solvent.
- Maintain reaction temperature between 30-100°C under inert gas protection (nitrogen) for 10-36 hours to ensure full conversion.
- Extract the mixture with dichloromethane and water, concentrate the organic phase, and purify via column chromatography to obtain the target deuterated compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology translates into tangible strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the supply chain for raw materials; by utilizing commodity chemicals like bromohexane and iodo-toluene derivatives, manufacturers can mitigate the risks associated with sourcing exotic or custom-synthesized deuterated building blocks. This shift to readily available starting materials enhances supply continuity and reduces the lead time for high-purity deuterated intermediates, allowing for more responsive production scheduling in alignment with client demands. Furthermore, the elimination of expensive deuterated solvents from the process bill of materials results in substantial cost savings, as the bulk solvent can be standard grade DMF which is easily recovered and recycled. This economic efficiency makes the production of deuterated APIs and materials financially sustainable even at early clinical stages where budget constraints are often tight.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by replacing costly deuterated solvents with inexpensive sodium deuteroformate and standard organic solvents. This change in reagent strategy removes a major cost driver from the variable expense sheet, allowing for more competitive pricing of the final deuterated active ingredients. Additionally, the one-pot nature of the reaction reduces the number of unit operations, saving on labor, energy, and equipment usage time, which collectively contribute to a lower cost of goods sold (COGS) for complex deuterated molecules.
- Enhanced Supply Chain Reliability: By relying on robust, commodity-grade starting materials such as alkyl bromides and haloarenes, the manufacturing process becomes less vulnerable to supply disruptions common with specialized deuterated reagents. The mild reaction conditions also mean that the process can be executed in standard glass-lined or stainless steel reactors without requiring specialized cryogenic or high-pressure equipment, further broadening the pool of qualified contract manufacturing organizations (CMOs) capable of executing the synthesis. This flexibility ensures a stable and resilient supply chain capable of meeting fluctuating market demands for deuterated pharmaceutical intermediates.
- Scalability and Environmental Compliance: The reaction operates at moderate temperatures (30-100°C) and atmospheric pressure, presenting a low barrier to scale-up from gram to ton quantities. The use of defined catalytic amounts of palladium, which can potentially be recovered, aligns with green chemistry principles by minimizing heavy metal waste. Moreover, the high atom economy of the coupling-reduction sequence reduces the generation of saline waste streams compared to multi-step protection-deprotection strategies, facilitating easier waste treatment and compliance with increasingly stringent environmental regulations in chemical manufacturing hubs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this deuterium labeling technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on yield expectations, purity profiles, and operational parameters. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing process development pipelines for new drug entities or advanced electronic materials.
Q: What is the deuteration rate achievable with this novel one-pot method?
A: According to patent CN110054541B, the method consistently achieves a deuteration rate of more than 98 percent, ensuring high isotopic purity essential for metabolic stability studies and OLED applications.
Q: How does this process reduce production costs compared to traditional deuteration?
A: The process eliminates the need for expensive deuterated solvents by using non-deuterated DMF and inexpensive sodium deuteroformate as the deuterium source, significantly lowering raw material expenses.
Q: Is this synthesis method suitable for large-scale industrial manufacturing?
A: Yes, the reaction operates under mild conditions (30-100°C) using commercially available starting materials like halogenated aromatics and alkyl bromides, making it highly scalable for commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Deuterated Aromatic Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your deuterated projects move seamlessly from the laboratory bench to the marketplace. Our facilities are equipped with rigorous QC labs and advanced analytical instrumentation capable of verifying stringent purity specifications, including precise measurement of deuteration levels to ensure they meet the >98% benchmark required for regulatory filings. We understand the critical nature of isotopic purity in drug metabolism and pharmacokinetics (DMPK) studies and are committed to delivering materials that support your most demanding research and development goals.
We invite you to engage with our technical procurement team to discuss how this innovative palladium-catalyzed methodology can be tailored to your specific molecule. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this one-pot synthesis route for your pipeline candidates. We encourage potential partners to contact us immediately to索取 specific COA data and route feasibility assessments, allowing us to demonstrate how our commitment to quality and efficiency can accelerate your time to market for next-generation deuterated therapeutics and materials.
