Revolutionizing Thioester Production: A Scalable Carbonylation Strategy for Global Supply Chains
Revolutionizing Thioester Production: A Scalable Carbonylation Strategy for Global Supply Chains
The landscape of organic synthesis is continuously evolving to meet the rigorous demands of modern pharmaceutical and fine chemical manufacturing, particularly regarding safety, efficiency, and scalability. A significant breakthrough in this domain is documented in patent CN113004181B, which discloses a highly efficient method for preparing thioester compounds through a transition metal-catalyzed carbonylation reaction. This innovative approach fundamentally shifts the paradigm by utilizing inexpensive and readily available benzyl chloride compounds and sulfonyl chlorides as starting materials, effectively bypassing the traditional reliance on malodorous and catalyst-poisoning thiol reagents. For global procurement and R&D teams, this technology represents a critical advancement in the reliable supply of high-purity thioester intermediates, offering a robust pathway that combines operational simplicity with exceptional functional group tolerance. The methodology not only streamlines the synthetic route but also enhances the overall sustainability of the process by employing tungsten hexacarbonyl as a dual-purpose reagent, thereby reducing the complexity of the reaction system.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of thioester compounds has predominantly relied on the direct acylation of thiols with carboxylic acids or their derivatives, a process fraught with significant logistical and technical challenges for industrial applications. Thiols, while effective nucleophiles, are notorious for their extremely unpleasant and pervasive odors, which pose severe health and safety risks in large-scale manufacturing environments and necessitate specialized containment infrastructure. Furthermore, sulfur-containing compounds like thiols have a strong tendency to poison transition metal catalysts, leading to reduced catalytic turnover, lower reaction yields, and increased costs associated with catalyst loading and recovery. Alternative methods involving the oxidative coupling of aldehydes or substitution reactions of halogenated alkanes often suffer from limited substrate scope, harsh reaction conditions, or the generation of difficult-to-remove byproducts. These conventional limitations create substantial bottlenecks in the supply chain for pharmaceutical intermediates, where consistency, purity, and operator safety are paramount concerns for any reliable thioester compound supplier aiming to serve the global market.
The Novel Approach
In stark contrast to these legacy methods, the novel carbonylation strategy outlined in the patent data introduces a transformative solution by leveraging sulfonyl chlorides as a superior sulfur source, effectively eliminating the need for handling volatile and toxic thiols. This approach utilizes a palladium-catalyzed system where benzyl chlorides react with sulfonyl chlorides in the presence of tungsten hexacarbonyl, triethylamine, and water to generate the desired thioester backbone with high efficiency. The use of sulfonyl chlorides not only mitigates the odor issues but also prevents catalyst deactivation, ensuring consistent reaction performance across a wide variety of substrates. Additionally, the integration of tungsten hexacarbonyl serves a dual purpose as both the carbonyl source and the reducing agent, which drastically simplifies the reagent profile and removes the necessity for external reducing agents or high-pressure carbon monoxide gas. This streamlined protocol offers a safer, more cost-effective, and environmentally friendlier alternative for cost reduction in pharmaceutical intermediate manufacturing, making it an ideal candidate for commercial scale-up.
Mechanistic Insights into Palladium-Catalyzed Carbonylation
The core of this synthetic innovation lies in the sophisticated interplay between the palladium catalyst, the phosphine ligand, and the unique reactivity of tungsten hexacarbonyl under thermal conditions. The reaction initiates with the oxidative addition of the benzyl chloride substrate to the active palladium(0) species, generated in situ from palladium acetate and the Xantphos ligand, forming a benzyl-palladium(II) intermediate. Subsequently, tungsten hexacarbonyl acts as a solid-state carbon monoxide reservoir, releasing CO under the reaction temperature of 100°C to facilitate the migratory insertion of the carbonyl group into the palladium-carbon bond. This step is critical for constructing the acyl-palladium species, which is then poised for nucleophilic attack. The presence of water and triethylamine facilitates the activation of the sulfonyl chloride, likely generating a reactive sulfur species in situ that can intercept the acyl-palladium complex. This mechanistic pathway avoids the formation of free thiol intermediates, thereby preserving the integrity of the catalytic cycle and ensuring high turnover numbers even with sensitive functional groups present on the aromatic rings.
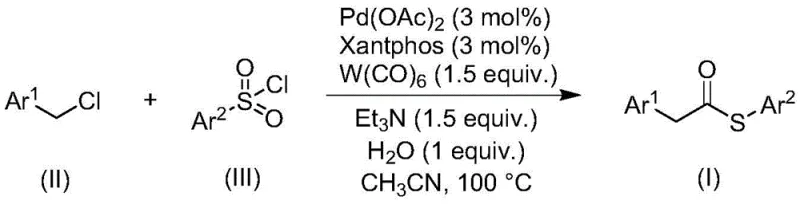
Furthermore, the impurity control mechanism inherent in this design is robust, primarily due to the high chemoselectivity of the palladium catalyst system towards the specific activation of the benzyl chloride bond over other potential reactive sites. The use of Xantphos, a bidentate ligand with a large bite angle, stabilizes the palladium center and promotes the reductive elimination step, which releases the final thioester product and regenerates the active catalyst. This stabilization minimizes side reactions such as homocoupling of the benzyl chloride or hydrolysis of the sulfonyl chloride, which are common pitfalls in less optimized systems. The reaction conditions, specifically the use of acetonitrile as a polar aprotic solvent, further enhance the solubility of the ionic intermediates and facilitate the smooth progression of the catalytic cycle. By avoiding the use of gaseous CO and toxic thiols, the process inherently reduces the risk of forming sulfur-based impurities or carbonyl-metal clusters that are difficult to separate, resulting in a cleaner crude reaction profile that simplifies downstream purification efforts for high-purity thioester intermediates.
How to Synthesize Thioester Compounds Efficiently
The practical implementation of this carbonylation method is designed for ease of execution in standard laboratory and pilot plant settings, requiring no specialized high-pressure equipment typically associated with carbonylation reactions. The procedure involves charging a reaction vessel with the benzyl chloride derivative, the sulfonyl chloride derivative, palladium acetate, Xantphos ligand, tungsten hexacarbonyl, triethylamine, and a stoichiometric amount of water in acetonitrile. The mixture is then heated to 100°C and stirred for approximately 24 hours, allowing the slow release of CO from the tungsten complex to drive the reaction to completion without the hazards of pressurized gas cylinders. Following the reaction, the workup is straightforward, involving simple filtration to remove metal residues followed by silica gel treatment and column chromatography to isolate the pure product. Detailed standardized synthesis steps for specific derivatives are provided in the guide below to assist technical teams in replicating these results.
- Combine palladium acetate, Xantphos ligand, tungsten hexacarbonyl, triethylamine, water, benzyl chloride, and sulfonyl chloride in acetonitrile solvent.
- Heat the reaction mixture to 100°C and maintain stirring for 24 hours under inert atmosphere to ensure complete conversion.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the high-purity thioester product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this patented methodology offers profound advantages that directly address the pain points of cost, safety, and supply continuity in the fine chemical sector. By replacing expensive and hazardous thiol reagents with stable, commodity-grade sulfonyl chlorides and benzyl chlorides, the process significantly lowers the raw material input costs and reduces the regulatory burden associated with handling volatile sulfur compounds. The elimination of high-pressure carbon monoxide gas further simplifies the facility requirements, allowing for production in standard glass-lined reactors rather than specialized autoclaves, which translates to faster turnaround times and greater flexibility in manufacturing scheduling. For supply chain leaders, this means a more resilient production capability that is less susceptible to disruptions caused by specialized reagent shortages or safety shutdowns, ensuring a steady flow of critical intermediates for downstream drug synthesis.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of costly and difficult-to-handle thiols with inexpensive sulfonyl chlorides and the dual functionality of tungsten hexacarbonyl. By serving as both the carbonyl source and the reducing agent, tungsten hexacarbonyl eliminates the need for purchasing separate reducing agents and complex gas delivery systems, thereby reducing the overall bill of materials. Furthermore, the high catalyst efficiency and the use of a robust ligand system minimize the amount of precious palladium required per kilogram of product, lowering the metal cost burden. The simplified workup procedure, which avoids complex extraction or distillation steps often needed to remove foul-smelling thiols, also reduces labor and utility costs, contributing to substantial cost savings in pharmaceutical intermediate manufacturing without compromising on yield or quality.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials such as benzyl chlorides and sulfonyl chlorides ensures a stable and diversified supply base, as these commodities are produced by numerous chemical manufacturers globally. Unlike specialized thiols which may have limited suppliers and long lead times, these precursors can be sourced competitively, reducing the risk of single-source dependency. The mild reaction conditions and the absence of hazardous gases mean that the process can be scaled up rapidly in existing facilities without major capital expenditure on new infrastructure. This agility allows for responsive production planning, enabling suppliers to meet fluctuating demand from pharmaceutical clients more effectively and reducing lead time for high-purity thioester intermediates during critical project phases.
- Scalability and Environmental Compliance: The environmental footprint of this synthesis is markedly lower than traditional methods, aligning with the increasing regulatory pressures for green chemistry in the fine chemical industry. By avoiding the emission of volatile organic sulfur compounds and the use of toxic carbon monoxide gas, the process improves workplace safety and reduces the complexity of waste gas treatment systems. The reaction generates minimal hazardous waste, and the use of acetonitrile, a solvent that can be efficiently recovered and recycled, supports sustainable manufacturing practices. This compliance with stringent environmental standards facilitates smoother regulatory approvals for new drug applications and enhances the corporate social responsibility profile of the manufacturing partner, making it a preferred choice for commercial scale-up of complex organic sulfides in regulated markets.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the feasibility of this technology for their specific projects, we have compiled answers to common inquiries based on the detailed experimental data provided in the patent literature. These questions address the versatility of the substrate scope, the specific roles of key reagents, and the practical aspects of scaling the reaction for industrial production. Understanding these technical nuances is essential for R&D directors and process chemists who are looking to integrate this method into their existing synthetic routes for API intermediates.
Q: Why is sulfonyl chloride preferred over thiols in this synthesis?
A: Sulfonyl chlorides are used to avoid the unpleasant odor and catalyst poisoning associated with traditional thiol reagents, significantly improving operational safety and catalyst longevity.
Q: What is the role of Tungsten Hexacarbonyl in this reaction?
A: Tungsten hexacarbonyl serves a dual function as both the carbonyl source (CO donor) and the reducing agent, eliminating the need for additional reducing agents and simplifying the reagent profile.
Q: Can this method tolerate diverse functional groups on the aromatic rings?
A: Yes, the method demonstrates wide substrate tolerance, successfully accommodating substituents such as methyl, methoxy, halogens, and tert-butyl groups on both the benzyl and sulfonyl components.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thioester Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team of expert process chemists has thoroughly analyzed the carbonylation technology described in patent CN113004181B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this innovation to your supply chain. We are committed to delivering high-purity thioester intermediates that meet stringent purity specifications, utilizing our rigorous QC labs to ensure every batch conforms to the highest international standards. Our state-of-the-art facilities are equipped to handle the specific requirements of palladium-catalyzed reactions, ensuring safe and efficient manufacturing that aligns with your project timelines and quality expectations.
We invite you to collaborate with us to leverage this cutting-edge synthesis route for your next development program. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating how this sulfonyl chloride-based approach can optimize your budget. We encourage you to contact us today to discuss your requirements,索取 specific COA data for our catalog compounds, or request route feasibility assessments for custom targets, ensuring your project moves forward with the most efficient and reliable chemistry available.
