Innovative Palladium-Catalyzed Route to High-Purity Trifluoromethyl Quinazolinones for Commercial Scale-Up
The recently granted Chinese patent CN113045503B introduces a groundbreaking methodology for synthesizing structurally diverse trifluoromethyl-substituted quinazolinone compounds through an innovative palladium-catalyzed carbonylation cascade reaction. This technical advancement addresses critical limitations in current pharmaceutical intermediate production by enabling efficient construction of these biologically significant heterocyclic frameworks under remarkably mild conditions compared to conventional approaches. The methodology demonstrates exceptional versatility across a wide range of substrate combinations while maintaining high operational simplicity that facilitates seamless integration into existing manufacturing workflows. Crucially, this patent provides a robust foundation for producing high-value intermediates essential for next-generation drug development programs targeting various therapeutic areas including antiviral and anticancer applications. The documented process represents a significant leap forward in synthetic methodology by eliminating multiple problematic steps inherent in traditional routes while simultaneously enhancing overall process efficiency and material economy through strategic catalyst design.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Existing synthetic methodologies for producing trifluoromethyl quinazolinones face substantial challenges that severely limit their industrial applicability and economic viability within pharmaceutical manufacturing environments. Traditional approaches typically require harsh reaction conditions involving elevated temperatures or pressures that necessitate specialized equipment and increase operational risks while simultaneously demanding expensive pre-functionalized substrates that significantly elevate raw material costs. These conventional routes frequently suffer from narrow substrate scope with poor functional group tolerance that restricts structural diversity and complicates the synthesis of complex derivatives required for advanced drug discovery programs. Furthermore, the multi-step sequences inherent in standard protocols generate substantial waste streams and require extensive purification procedures that dramatically reduce overall process efficiency and increase environmental impact. The consistently low yields observed across multiple literature methods create significant barriers to commercial implementation by making large-scale production economically unfeasible while also introducing unacceptable variability in product quality that fails to meet stringent pharmaceutical standards.
The Novel Approach
The patented methodology overcomes these critical limitations through an elegant palladium-catalyzed carbonylation cascade reaction that operates under remarkably mild conditions while utilizing inexpensive and readily available starting materials including trifluoroethylimidoyl chloride and diverse amine substrates. This innovative approach eliminates the need for pre-functionalized reagents by directly incorporating the trifluoromethyl group through a streamlined single-step transformation that significantly reduces both process complexity and environmental footprint. The exceptional substrate compatibility demonstrated across various functional groups enables the synthesis of structurally diverse quinazolinone derivatives without requiring specialized handling procedures or exotic reagents that would complicate scale-up efforts. By operating at moderate temperatures with standard laboratory equipment, this method substantially improves process safety while simultaneously enhancing operational flexibility across different manufacturing environments. Most significantly, the documented high yields and simplified purification protocol directly translate to improved economic viability and consistent product quality that meets rigorous pharmaceutical requirements.
Mechanistic Insights into Palladium-Catalyzed Carbonylation
The reaction mechanism proceeds through a sophisticated cascade sequence initiated by base-promoted intermolecular carbon-nitrogen bond coupling between trifluoroethylimidoyl chloride and amine substrates to form key trifluoroacetamidine intermediates. This critical transformation occurs under mild conditions without requiring expensive transition metal catalysts or specialized activation protocols that would complicate industrial implementation while maintaining excellent functional group tolerance across diverse substrate combinations. The subsequent palladium insertion into the carbon-iodine bond forms a crucial divalent palladium intermediate that serves as the foundation for the carbonylation step essential to quinazolinone ring formation. TFBen functions as an efficient carbon monoxide surrogate that releases CO under controlled thermal conditions to facilitate insertion into the carbon-palladium bond, creating the acyl palladium species necessary for cyclization. The base-mediated formation of a seven-membered ring palladium intermediate represents a key innovation that enables smooth reductive elimination to yield the final trifluoromethyl quinazolinone product with exceptional regioselectivity and minimal side reactions.
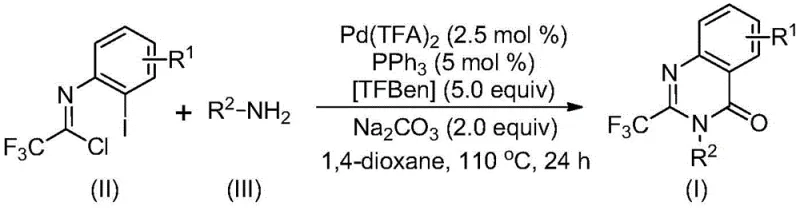
The documented mechanism provides critical insights into impurity control through precise regulation of reaction parameters including temperature profile management and stoichiometric control of catalytic components that prevent common side reactions such as over-reduction or undesired cyclization pathways. The use of sodium carbonate as base creates optimal pH conditions that minimize hydrolysis side products while promoting efficient cyclization through controlled deprotonation steps essential for ring closure. Substrate design considerations revealed through extensive experimentation demonstrate how strategic selection of amine components directly influences both reaction kinetics and final product purity by modulating electronic effects on the cyclization transition state. The well-defined catalytic cycle ensures consistent product quality across different substrate combinations by maintaining stable intermediate concentrations throughout the reaction sequence without accumulation of problematic byproducts.
How to Synthesize Trifluoromethyl Quinazolinones Efficiently
This patented methodology represents a significant advancement in synthetic efficiency for producing high-value trifluoromethyl quinazolinone intermediates through its innovative palladium-catalyzed carbonylation approach that eliminates multiple problematic steps inherent in traditional routes while maintaining exceptional operational simplicity suitable for industrial implementation. The process demonstrates remarkable versatility across diverse substrate combinations without requiring specialized equipment or exotic reagents that would complicate scale-up efforts within standard pharmaceutical manufacturing facilities. By operating under mild conditions with readily available starting materials, this method substantially improves both economic viability and environmental sustainability compared to conventional approaches while delivering consistent high-purity products meeting stringent pharmaceutical requirements. Detailed standardized synthesis steps are provided below to facilitate seamless technology transfer and implementation within your manufacturing operations.
- Prepare the reaction mixture by combining trifluoroethylimidoyl chloride with amine substrates under inert atmosphere using dioxane as solvent.
- Introduce palladium trifluoroacetate catalyst with triphenylphosphine ligand and TFBen carbon monoxide source at precise stoichiometric ratios.
- Conduct thermal reaction at controlled temperature followed by standardized purification protocol to obtain high-purity intermediates.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthetic route delivers substantial commercial benefits specifically designed to address critical pain points faced by procurement and supply chain professionals within pharmaceutical manufacturing organizations seeking reliable sources of high-quality intermediates. The methodology directly tackles persistent challenges related to supply chain vulnerability by utilizing widely available starting materials that eliminate dependence on specialized suppliers while simultaneously reducing exposure to market fluctuations affecting exotic reagents required by conventional methods. The streamlined process design significantly enhances operational flexibility across different manufacturing environments by removing complex purification requirements that typically create bottlenecks during scale-up activities while improving overall resource utilization efficiency throughout production cycles. These advantages collectively translate into meaningful improvements in supply chain resilience and cost structure optimization without requiring substantial capital investment or process revalidation efforts.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts required by alternative routes combined with simplified purification protocols substantially reduces overall production costs through decreased raw material expenditure and lower energy consumption during manufacturing operations while maintaining consistent high yields across diverse substrate combinations.
- Enhanced Supply Chain Reliability: Utilization of readily available commercial starting materials significantly improves supply chain robustness by eliminating dependencies on specialized suppliers while enabling flexible sourcing strategies that mitigate raw material availability risks through multiple viable procurement channels.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability from laboratory to industrial production volumes with minimal environmental impact due to reduced waste generation and simplified solvent recovery protocols that align with modern green chemistry principles while meeting stringent regulatory requirements.
Frequently Asked Questions (FAQ)
The following questions address critical technical and commercial considerations based on detailed analysis of patent CN113045503B's technical specifications and implementation requirements for pharmaceutical intermediate production environments. These insights derive directly from experimental data documented within the patent claims regarding process parameters, substrate scope limitations, and quality control metrics essential for successful technology transfer.
Q: What limitations do conventional methods face in synthesizing trifluoromethyl quinazolinones?
A: Traditional approaches suffer from harsh reaction conditions requiring expensive pre-activated substrates with narrow functional group tolerance and consistently low yields due to complex multi-step sequences that complicate industrial implementation.
Q: How does this palladium-catalyzed method improve substrate compatibility and process efficiency?
A: The novel route utilizes readily available starting materials with exceptional functional group tolerance across diverse amine substrates while operating under milder conditions that significantly enhance reaction efficiency and scalability without requiring specialized equipment.
Q: Can this synthesis achieve commercial-scale production while maintaining high purity?
A: Yes, the streamlined process demonstrates excellent scalability from laboratory to industrial production volumes with simplified purification protocols that consistently deliver high-purity intermediates meeting stringent pharmaceutical quality specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Quinazolinone Supplier
Our company possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs equipped with advanced analytical capabilities specifically designed for complex heterocyclic intermediates like trifluoromethyl quinazolinones. This patented methodology represents just one example of our commitment to developing innovative synthetic solutions that address critical challenges in pharmaceutical intermediate manufacturing while delivering consistent high-quality products meeting global regulatory standards across multiple therapeutic areas including antiviral and anticancer drug development programs.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team who will provide specific COA data and route feasibility assessments tailored to your unique production requirements while demonstrating how our expertise can optimize your supply chain performance through reliable access to high-purity intermediates.
