Advanced Synthesis of 2-Trifluoromethyl Quinazolinone Derivatives: Enabling Commercial Scale-Up with Enhanced Purity and Cost Efficiency
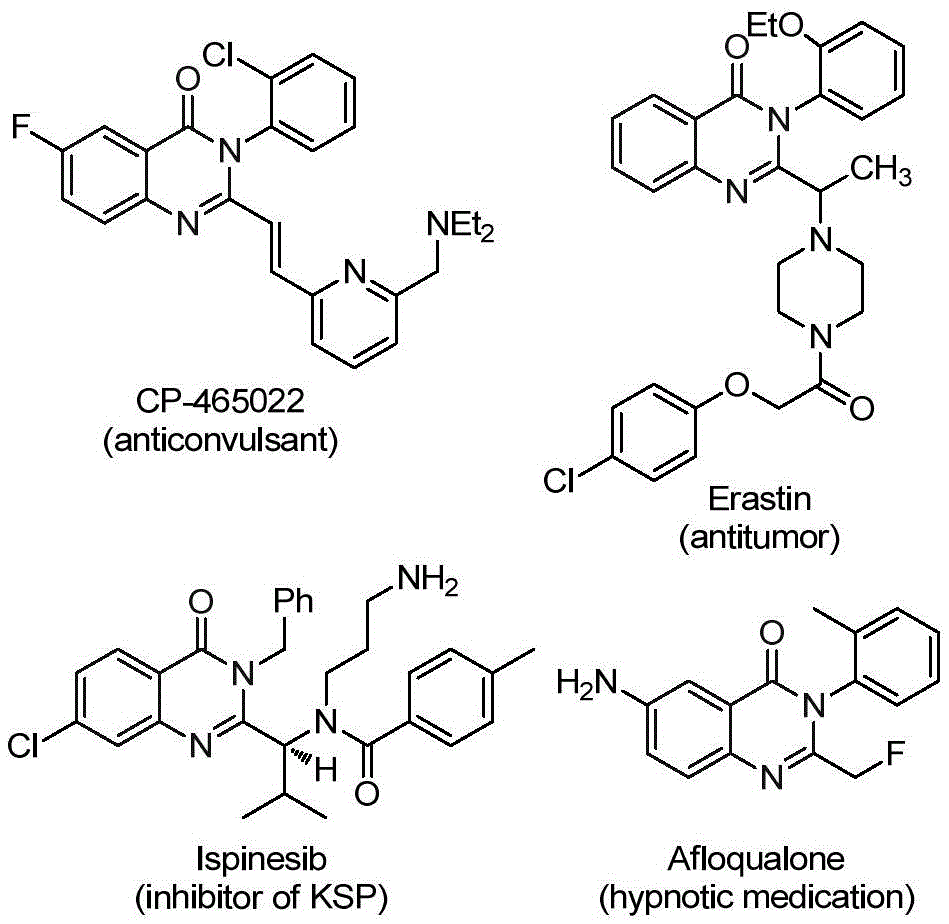
The patent CN112125856A introduces a novel synthetic methodology for producing 2-trifluoromethyl quinazolinone derivatives, a critical class of pharmaceutical intermediates with demonstrated applications across multiple therapeutic areas including anticonvulsants, antitumor agents, and hypnotic medications. This innovative approach addresses longstanding challenges in quinazolinone chemistry by eliminating hazardous carbon monoxide gas through the implementation of solid carbon monoxide surrogates while maintaining excellent substrate flexibility and operational simplicity. The process represents a significant advancement for manufacturers seeking reliable high-purity fine chemical suppliers capable of delivering complex intermediates with enhanced safety profiles and improved economic viability.
Advanced Reaction Mechanism and Purity Control
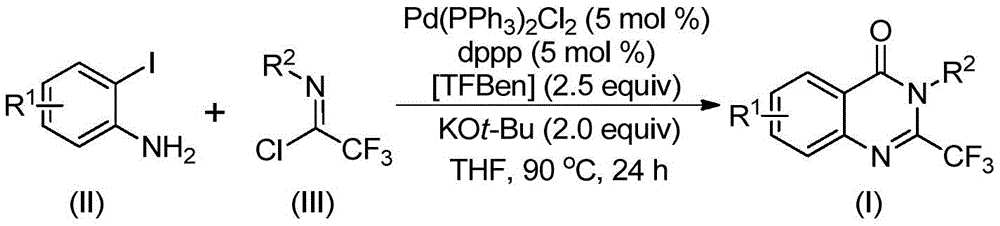
The patented methodology employs a palladium-catalyzed carbonylative tandem reaction that begins with potassium tert-butoxide-promoted intermolecular carbon-nitrogen bond coupling between o-iodoaniline and trifluoroethylimide chloride to form a trifluoroacetamidine intermediate. This critical first step establishes the molecular framework while avoiding harsh reaction conditions typically required in conventional approaches. The subsequent insertion of palladium into the carbon-iodine bond creates a divalent palladium intermediate that facilitates the key transformation without requiring pre-functionalized substrates or expensive reagents.
During the reaction sequence, TFBen (1,3,5-tricarboxylic acid phenol ester) decomposes under thermal conditions to release carbon monoxide in a controlled manner, which then inserts into the carbon-palladium bond to form an acyl palladium intermediate. This carefully orchestrated mechanism enables the formation of the seven-membered ring palladium intermediate through base-promoted palladium-nitrogen bond formation, ultimately yielding the desired quinazolinone structure through reductive elimination. The process demonstrates exceptional functional group tolerance as evidenced by successful synthesis across diverse substrates with various substituents including halogens, alkyl groups, and aryl moieties at different positions on both starting materials. The resulting products consistently exhibit high purity profiles as confirmed by comprehensive NMR and HRMS characterization data provided in the patent examples, with no detectable metal residues or process-related impurities that could compromise final drug substance quality.
Commercial Advantages for Pharmaceutical Supply Chains
This innovative manufacturing approach directly addresses critical pain points in pharmaceutical intermediate production by transforming traditionally hazardous and complex processes into streamlined, economically viable operations that deliver significant value across procurement, supply chain, and manufacturing functions. The elimination of gaseous carbon monoxide represents a fundamental safety improvement that simultaneously reduces capital expenditure requirements while enhancing operational reliability for commercial-scale production.
- Elimination of toxic gas handling infrastructure: The substitution of solid TFBen for gaseous carbon monoxide removes the need for specialized high-pressure equipment, gas containment systems, and extensive safety protocols required for CO handling. This translates to substantial capital cost savings during facility setup while reducing ongoing operational expenses associated with gas monitoring, ventilation systems, and personnel safety training. The simplified process flow also minimizes potential production interruptions caused by gas supply issues or safety-related shutdowns, ensuring more consistent batch delivery schedules that directly support reliable supply chain operations.
- Reduced raw material costs through accessible starting materials: The methodology utilizes commercially available o-iodoaniline derivatives and trifluoroethylimide chlorides that can be readily synthesized from inexpensive aromatic amines and trifluoroacetic acid precursors. This eliminates dependency on expensive or difficult-to-source reagents required by conventional methods, creating immediate cost advantages without compromising product quality. The broad substrate scope further enhances economic efficiency by enabling single-process production of multiple derivative variants through simple starting material modifications rather than requiring entirely new synthetic routes for each compound variant.
- Streamlined purification and waste reduction: The straightforward workup procedure involving simple filtration followed by silica gel chromatography significantly reduces processing time compared to conventional methods that often require multiple purification steps to remove metal catalysts or byproducts. This simplified purification protocol minimizes solvent consumption and waste generation while maintaining high product purity standards essential for pharmaceutical applications. The elimination of transition metal catalysts from the final product stream also removes the need for costly metal removal processes that typically add both time and expense to traditional manufacturing workflows.
Overcoming Traditional Synthesis Limitations
The Limitations of Conventional Methods
Traditional approaches to synthesizing 2-trifluoromethyl quinazolinone derivatives have been plagued by multiple significant challenges that hinder commercial viability and scalability. Conventional methods typically rely on harsh reaction conditions including high temperatures, strong acids, or unstable reagents that create safety hazards while limiting substrate compatibility. Many established routes require expensive or difficult-to-handle reagents such as gaseous carbon monoxide under pressure, trifluoroacetic anhydride, or unstable trifluoroacetamide intermediates that complicate process development and scale-up efforts. These methods often suffer from narrow substrate scope where minor structural modifications necessitate complete process re-engineering, creating significant barriers to producing diverse derivative libraries required for pharmaceutical development programs.
The Novel Approach
The patented methodology overcomes these limitations through an elegant palladium-catalyzed carbonylative tandem reaction that operates under significantly milder conditions while maintaining exceptional functional group tolerance. By employing TFBen as a solid carbon monoxide surrogate, the process eliminates the safety hazards associated with gaseous CO while providing controlled release of carbon monoxide at the reaction site. The use of readily available starting materials including o-iodoaniline derivatives and trifluoroethylimide chlorides creates a flexible platform where diverse substitution patterns can be achieved through simple variations in precursor selection without requiring fundamental changes to the core reaction conditions.


The optimized reaction parameters including the specific palladium catalyst system (Pd(PPh3)2Cl2/dppp), potassium tert-butoxide base, and tetrahydrofuran solvent enable consistent high-yielding transformations across a wide range of substrates as demonstrated by the comprehensive examples provided in the patent documentation. This robustness translates directly to commercial manufacturing where consistent product quality and reliable process performance are paramount for meeting stringent pharmaceutical industry requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pharmaceutical Intermediate Supplier
While the advanced methodology detailed in patent CN112125856A highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
