Revolutionizing Biaryl Production: A Deep Dive into Iron-Catalyzed Cross-Coupling for Commercial Scale-Up
The landscape of organic synthesis for complex aromatic structures is undergoing a significant transformation, driven by the urgent need for sustainable and cost-effective manufacturing processes. Patent CN110372544B introduces a groundbreaking methodology for the synthesis of biaryl compounds and their derivatives, utilizing a novel iron-catalyzed system that challenges the dominance of traditional precious metal catalysts. Biaryl motifs are ubiquitous in high-value sectors, serving as the structural backbone for critical active pharmaceutical ingredients (APIs) such as Losartan, natural products like Nantenine, and advanced functional materials including liquid crystals like NCB 807. The ability to construct these carbon-carbon bonds efficiently is paramount for the global supply chain of fine chemicals. This patent delineates a sophisticated catalytic protocol that leverages the synergistic effects of iron salts and titanium alkoxides, offering a pathway that is not only economically superior but also environmentally benign compared to legacy technologies.
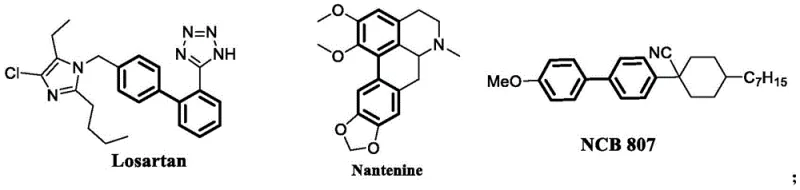
The strategic importance of this technology cannot be overstated for stakeholders managing the production of pharmaceutical intermediates and agrochemical precursors. By shifting the catalytic burden from scarce resources to earth-abundant metals, this innovation addresses both the volatility of raw material pricing and the stringent regulatory pressures regarding heavy metal residues in final drug products. The detailed experimental data within the patent demonstrates high yields and exceptional functional group tolerance, validating its potential for immediate adoption in commercial settings. As we delve deeper into the technical specifics, it becomes clear that this represents a paradigm shift in how we approach cross-coupling reactions, moving away from the atom-inefficient and toxic pathways of the past toward a greener, more robust future for industrial organic chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
For decades, the construction of biaryl scaffolds has relied heavily on transition metal-catalyzed cross-coupling reactions, predominantly utilizing palladium or nickel as the catalytic center. While these methods, such as the Suzuki-Miyaura or Stille couplings, have been indispensable tools in medicinal chemistry, they suffer from profound limitations when translated to large-scale industrial manufacturing. Palladium, being a precious metal, is subject to extreme price volatility and supply chain constraints, creating significant financial risk for long-term production contracts. Furthermore, nickel, although cheaper, poses severe toxicity concerns, necessitating rigorous and costly purification steps to ensure that residual metal levels in pharmaceutical products meet strict regulatory limits. Beyond the catalyst itself, traditional methods often require the pre-functionalization of coupling partners into organoboron, organozinc, or organotin species.
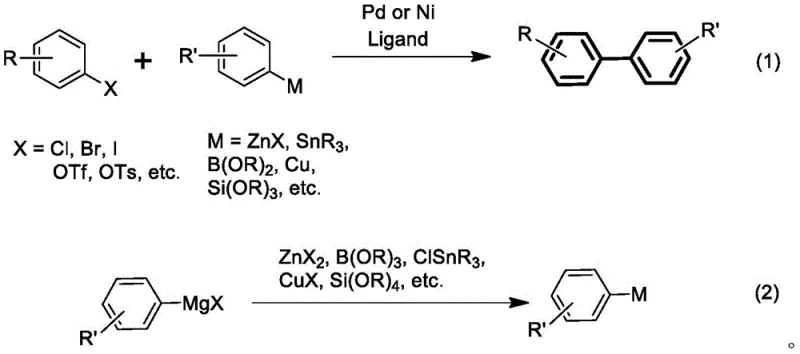
This pre-functionalization adds multiple synthetic steps, reducing overall atom economy and generating substantial quantities of hazardous waste, particularly when tin or zinc reagents are employed. The use of Grignard reagents in traditional palladium or nickel catalysis has historically been problematic due to their high reactivity, which often leads to the destruction of sensitive functional groups such as esters, nitriles, and ketones prior to the desired coupling event. Consequently, manufacturers are forced into a compromise between reactivity and selectivity, often resorting to protecting group strategies that further inflate costs and extend lead times. These cumulative inefficiencies create a bottleneck in the supply of high-purity biaryl intermediates, driving the industry to seek alternative catalytic systems that can bypass these inherent drawbacks while maintaining high performance standards.
The Novel Approach
The methodology disclosed in patent CN110372544B presents a transformative solution by replacing the conventional palladium or nickel catalysts with a synergistic system composed of iron salts and titanium alkoxides. This novel approach fundamentally alters the reaction landscape by enabling the direct use of aryl Grignard reagents with aryl halides or pseudo-halides, thereby eliminating the need for pre-formed organoboron or organozinc intermediates. The integration of titanium species, such as Ti(OEt)4, alongside iron catalysts like FeCl3 creates a unique chemical environment that moderates the reactivity of the Grignard reagent. This modulation is critical, as it prevents the nucleophilic attack on sensitive functional groups while still facilitating the desired cross-coupling reaction with high efficiency. The result is a streamlined synthetic route that reduces the number of unit operations, minimizes solvent consumption, and drastically cuts down on the generation of toxic heavy metal waste.
Moreover, the utilization of iron, the fourth most abundant element in the Earth's crust, ensures a stable and low-cost supply of the catalytic metal, decoupling production economics from the fluctuations of the precious metals market. The process operates under relatively mild thermal conditions, typically ranging from 35°C to 110°C, which enhances energy efficiency and simplifies reactor requirements. The compatibility of this system with a wide array of functional groups, including esters, amides, and nitriles, allows for the late-stage functionalization of complex molecules without the need for extensive protecting group manipulation. This level of operational simplicity and chemical robustness makes the iron-titanium catalytic system an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates, offering a clear pathway to cost reduction and sustainability improvements in fine chemical manufacturing.
Mechanistic Insights into FeCl3-Catalyzed Cross-Coupling
The success of this iron-catalyzed synthesis lies in the intricate interplay between the iron center, the ligand environment, and the titanium co-catalyst. The proposed mechanism involves the formation of a highly active iron-titanium heterobimetallic species that facilitates the oxidative addition of the aryl halide and the subsequent transmetallation with the Grignard reagent. Unlike traditional iron-catalyzed couplings which often struggle with homocoupling side reactions or poor functional group tolerance, the addition of titanium alkoxides and phenols acts as a crucial modifier. These additives likely coordinate to the magnesium center of the Grignard reagent, forming a less aggressive 'ate' complex that is sufficiently nucleophilic for coupling but sufficiently tempered to avoid attacking electrophilic sites on the substrate. This delicate balance is further tuned by the choice of ligands, such as TMEDA or N-heterocyclic carbenes, which stabilize the low-valent iron species necessary for the catalytic cycle.
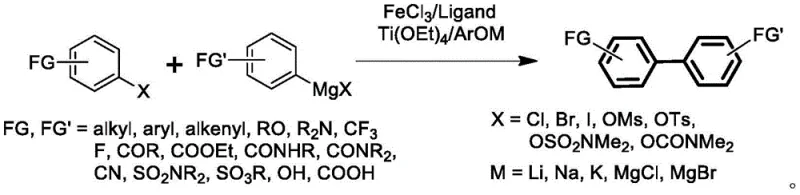
From an impurity control perspective, this mechanistic pathway offers distinct advantages. The high chemoselectivity ensures that side products arising from the reaction of the Grignard reagent with ester or nitrile groups are minimized, leading to cleaner crude reaction profiles. This reduction in byproduct formation simplifies downstream purification, often allowing for crystallization rather than chromatography, which is a significant benefit for industrial processing. Furthermore, the iron catalyst, being non-precious, does not require the same level of scavenging as palladium, although standard aqueous workups effectively remove iron residues as insoluble hydroxides or oxides. The titanium byproducts, primarily converting to titanium dioxide upon hydrolysis, are non-toxic and can potentially be recovered or disposed of with minimal environmental impact. This mechanistic understanding underscores the robustness of the process, providing R&D teams with the confidence to apply this chemistry to a broad scope of substrates with predictable outcomes.
How to Synthesize Biaryl Derivatives Efficiently
The practical implementation of this synthesis involves a straightforward protocol that can be easily adapted to existing manufacturing infrastructure. The process begins with the in situ preparation of a titanium-modified Grignard reagent, followed by its addition to a mixture containing the aryl halide, iron catalyst, and ligand. This one-pot strategy minimizes handling of sensitive intermediates and reduces exposure to air and moisture, enhancing operational safety. The detailed standardized synthesis steps below outline the precise stoichiometry and conditions required to achieve optimal yields, ensuring reproducibility across different batches and scales. By adhering to these guidelines, manufacturers can leverage the full potential of this iron-titanium catalytic system to produce high-quality biaryl intermediates.
- Prepare the titanium reagent by mixing Ti(OEt)4 and phenol in THF, then adding the aryl Grignard reagent at room temperature.
- In a separate vessel, dissolve the aryl halide or pseudo-halide in THF and add the iron catalyst (FeCl3), ligand (TMEDA), and optional phenate.
- Combine the titanium reagent with the iron mixture, add toluene co-solvent, and heat to reflux (35-110°C) until the reaction is complete.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this iron-catalyzed technology translates into tangible strategic benefits that extend far beyond simple reaction yields. The primary advantage lies in the drastic simplification of the supply chain for raw materials. By eliminating the dependency on palladium and the need for specialized organoboron or organotin reagents, companies can source starting materials from a much broader and more competitive supplier base. This diversification reduces the risk of supply disruptions caused by geopolitical instability or mining constraints associated with precious metals. Additionally, the removal of toxic heavy metals from the process flow significantly lowers the costs associated with waste disposal and environmental compliance, which are increasingly becoming major line items in the operational budgets of chemical manufacturers.
- Cost Reduction in Manufacturing: The substitution of expensive palladium catalysts with inexpensive iron salts results in a direct and substantial decrease in raw material costs. Since iron is orders of magnitude cheaper than palladium, the catalyst cost per kilogram of product is negligible. Furthermore, the ability to use Grignard reagents directly avoids the multi-step synthesis and purification of boronic acids or zinc reagents, effectively cutting out entire stages of production. This reduction in step count not only saves on reagents but also reduces labor, energy, and equipment usage, leading to a significantly lower cost of goods sold (COGS) for the final biaryl intermediate.
- Enhanced Supply Chain Reliability: Relying on earth-abundant metals like iron and titanium insulates the production process from the volatility of the precious metals market. Supply chains for iron and titanium are mature, global, and highly stable, ensuring consistent availability of catalytic materials regardless of market fluctuations. Moreover, the robustness of the reaction conditions means that the process is less susceptible to variations in raw material quality, further stabilizing production schedules. This reliability is crucial for meeting the just-in-time delivery requirements of downstream pharmaceutical clients and maintaining uninterrupted supply of critical API intermediates.
- Scalability and Environmental Compliance: The mild reaction conditions and the absence of highly toxic reagents make this process inherently safer and easier to scale from laboratory to pilot and commercial plant scales. The waste stream, primarily consisting of iron and titanium salts, is far less hazardous than those containing tin or palladium, simplifying wastewater treatment and solid waste management. This alignment with green chemistry principles not only reduces environmental liability but also enhances the corporate sustainability profile, which is increasingly important for securing contracts with major multinational pharmaceutical companies that prioritize eco-friendly supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this iron-catalyzed synthesis method. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on the practical aspects of adopting this technology. Understanding these details is essential for technical teams evaluating the feasibility of integrating this process into their existing manufacturing workflows.
Q: Why is the iron-titanium catalytic system superior to traditional palladium methods?
A: The iron-titanium system eliminates the need for expensive palladium and toxic nickel, significantly reducing raw material costs. Furthermore, it allows for the direct use of Grignard reagents without pre-functionalization into boron or zinc species, streamlining the synthetic route and minimizing toxic waste generation.
Q: Can this method tolerate sensitive functional groups like esters and nitriles?
A: Yes, a key breakthrough of this patent is the high chemical selectivity achieved through the synergistic catalysis. The specific combination of iron salts, titanium alkoxides, and ligands modulates the reactivity of the Grignard reagent, allowing it to couple successfully even in the presence of sensitive groups such as esters, amides, nitriles, and ketones.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Absolutely. The process operates under mild conditions (35-110°C) and utilizes abundant, low-toxicity metals. The absence of heavy metal residues simplifies purification, and the robustness of the catalytic system ensures consistent yields, making it highly scalable for commercial production of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Biaryl Intermediate Supplier
The technological advancements detailed in patent CN110372544B represent a significant leap forward in the efficient production of biaryl compounds, yet realizing their full commercial potential requires a partner with deep expertise in process development and scale-up. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of iron-catalyzed reactions, ensuring that the theoretical benefits of cost and efficiency are fully actualized in every batch we produce. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every gram of material meets the exacting standards required for pharmaceutical applications.
We invite you to collaborate with us to explore how this innovative synthesis route can optimize your supply chain and reduce your manufacturing costs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific product portfolio, demonstrating the economic impact of switching to this iron-catalyzed method. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a more sustainable and cost-effective supply of high-purity biaryl intermediates for your future projects.
