Advanced Manufacturing of Naphthalenone Derivatives via Optimized Palladium-Copper Cascade Catalysis
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing complex polycyclic architectures, particularly those containing naphthalenone scaffolds which serve as pivotal intermediates in drug discovery. Patent CN112920033A discloses a groundbreaking dual-catalytic strategy that addresses the longstanding challenges in synthesizing o-alkynyl phenylcyclobutanones and their subsequent conversion into naphthalenones. This technical disclosure outlines a highly efficient pathway utilizing a palladium-catalyzed Sonogashira coupling followed by a Lewis acid-mediated cascade cyclization. For R&D directors and process chemists, this represents a significant advancement over traditional methods, offering a streamlined route to access valuable chemical space with improved atom economy and operational simplicity. The ability to construct these fused ring systems under relatively mild conditions opens new avenues for the scalable production of bioactive molecules.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of alkynyl groups onto strained ring systems like cyclobutanones has been fraught with difficulties, particularly when attempting to functionalize the beta-position relative to the carbonyl group. Traditional palladium-copper co-catalyzed Sonogashira reactions often suffer from low conversion rates, extensive formation of homocoupling byproducts, and the inability to tolerate the steric strain inherent in the cyclobutanone ring. Furthermore, previous literature reports indicate that synthesizing beta-(2-alkynylphenyl) cyclobutanones via standard protocols frequently results in poor yields and complex impurity profiles that are difficult to separate. The harsh conditions often required to drive these sluggish reactions can lead to the decomposition of the sensitive four-membered ring, thereby compromising the overall mass balance and making the process economically unviable for large-scale manufacturing.
The Novel Approach
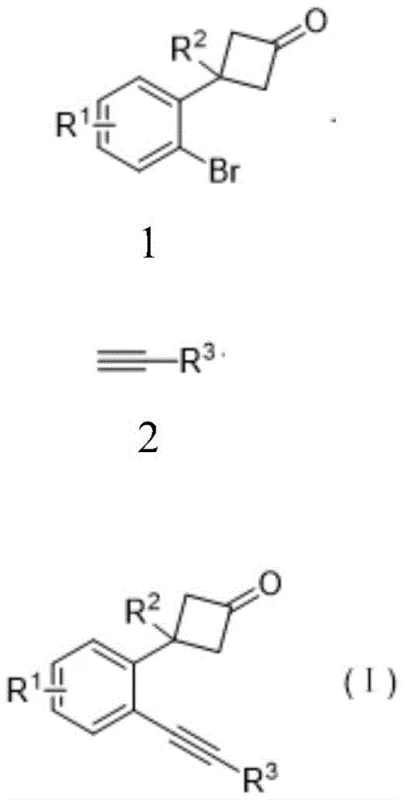
The methodology presented in the patent data revolutionizes this synthetic challenge by employing a specialized palladium catalyst system comprising Pd(TFA)2 and the bidentate ligand DPEphos. This specific catalytic combination enables the smooth coupling of o-bromophenyl cyclobutanone with various terminal alkynes at moderate temperatures ranging from 80°C to 100°C. Unlike conventional approaches, this optimized system achieves exceptional yields, with specific examples demonstrating conversion efficiencies reaching up to 90% under optimized conditions. The subsequent transformation into naphthalenones utilizes a copper(II) triflate catalyst to trigger a unique ring-opening cyclization sequence. This two-step tandem approach not only bypasses the limitations of direct functionalization but also provides a versatile platform for generating diverse naphthalenone derivatives with high structural fidelity.
Mechanistic Insights into Pd-Catalyzed Coupling and Cu-Mediated Cyclization
The success of this synthetic route relies heavily on the precise interplay between the palladium catalyst and the phosphine ligand during the initial cross-coupling phase. The mechanism likely proceeds through the oxidative addition of the aryl bromide to the active Pd(0) species, followed by transmetallation with the copper-acetylide generated in situ. The bulky and electron-rich nature of the DPEphos ligand stabilizes the palladium center, preventing aggregation and facilitating the reductive elimination step to release the desired o-alkynyl product. This mechanistic pathway is crucial for maintaining the integrity of the cyclobutanone ring, which is susceptible to nucleophilic attack or thermal degradation under less controlled conditions. The use of potassium carbonate as a mild base further ensures that the reaction environment remains conducive to coupling without promoting unwanted side reactions such as aldol condensations.
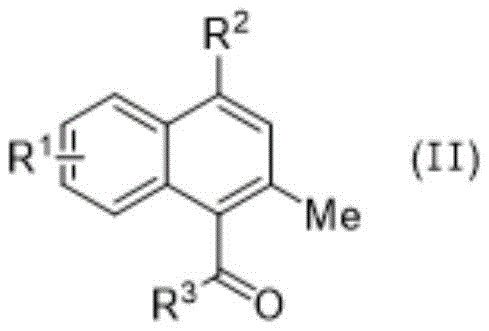
Following the isolation of the o-alkynyl phenylcyclobutanone intermediate, the second stage involves a fascinating Lewis acid-catalyzed rearrangement. Upon exposure to Cu(OTf)2, the cyclobutanone ring undergoes a selective ring-opening to generate a conjugated ketene intermediate. This highly reactive species then participates in an intramolecular [2+2] cycloaddition with the proximal alkyne moiety. The process concludes with a retro-[2+2] cycloaddition that restores the carbonyl functionality while establishing the fused naphthalene framework. This cascade sequence is elegantly efficient, transforming a simple acyclic precursor into a complex polycyclic ketone in a single operational step. The regeneration of the copper catalyst at the end of the cycle underscores the catalytic nature of this transformation, minimizing metal waste and simplifying downstream purification processes.
How to Synthesize Naphthalenone Derivatives Efficiently
The practical implementation of this chemistry requires careful attention to reaction parameters to maximize yield and purity. The protocol begins with the preparation of the coupling mixture under an inert atmosphere to prevent oxidation of the catalyst. Precise stoichiometry is maintained with a slight excess of the terminal alkyne to drive the equilibrium forward. Following the coupling reaction, the crude mixture is subjected to standard aqueous workup and column chromatography to isolate the intermediate. The subsequent cyclization step is performed in chloroform with a catalytic amount of water, which plays a critical role in facilitating the ring-opening event. Detailed standardized operating procedures for scaling this synthesis from gram to kilogram quantities are essential for ensuring reproducibility and safety in a production environment.
- React o-bromophenyl cyclobutanone with terminal alkyne using Pd(TFA)2 and DPEphos ligand in 1,4-dioxane at 90°C for 12 hours.
- Purify the resulting o-alkynyl phenylcyclobutanone intermediate via column chromatography.
- Treat the intermediate with Cu(OTf)2 catalyst in chloroform at 65°C for 18 hours to effect ring-opening cyclization into naphthalenone.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented methodology offers substantial strategic benefits that directly impact the bottom line and operational resilience. The reliance on commercially available starting materials, such as o-bromophenyl cyclobutanone and common terminal alkynes, ensures a stable and secure supply chain free from the bottlenecks associated with exotic reagents. The use of standard solvents like 1,4-dioxane and chloroform further simplifies logistics, as these are readily sourced from multiple global suppliers, reducing the risk of single-source dependency. Moreover, the high yields reported in the patent data translate directly into reduced raw material consumption per unit of product, effectively lowering the cost of goods sold (COGS) without compromising on quality standards.
- Cost Reduction in Manufacturing: The elimination of expensive and toxic co-catalysts traditionally used in Sonogashira reactions significantly reduces the cost burden associated with catalyst procurement and waste disposal. By utilizing a highly active Pd(TFA)2 system at low loading levels (2.5 mol%), the process minimizes the amount of precious metal required, which is a major cost driver in fine chemical synthesis. Additionally, the simplified purification steps reduce the consumption of silica gel and solvents during chromatography, leading to further operational savings. The overall efficiency of the process means that less energy is consumed per kilogram of product, contributing to a more sustainable and cost-effective manufacturing profile.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route against variations in substrate electronics ensures consistent production output even when sourcing raw materials from different vendors. The tolerance for diverse functional groups means that the same core process can be adapted to produce a wide library of analogues without requiring extensive re-optimization, thereby accelerating time-to-market for new drug candidates. This flexibility allows supply chain managers to respond rapidly to changing demand signals, ensuring continuity of supply for critical pharmaceutical intermediates. The use of stable catalysts and mild reaction conditions also reduces the risk of batch failures due to equipment malfunction or operator error.
- Scalability and Environmental Compliance: The reaction conditions described are inherently scalable, moving seamlessly from laboratory flasks to industrial reactors without the need for specialized high-pressure equipment. The absence of hazardous reagents and the use of recyclable solvents align with modern green chemistry principles, facilitating easier regulatory approval and environmental compliance. Waste generation is minimized through high atom economy and efficient catalyst turnover, reducing the environmental footprint of the manufacturing process. This alignment with sustainability goals not only mitigates regulatory risk but also enhances the brand reputation of the manufacturing partner in the eyes of environmentally conscious clients.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthetic pathway. Understanding these nuances is critical for process engineers and quality assurance teams tasked with validating the method for commercial production. The answers provided are derived directly from the experimental data and mechanistic insights contained within the patent documentation, ensuring accuracy and reliability for decision-making purposes.
Q: What is the optimal catalyst system for the initial coupling reaction?
A: The patent data indicates that Pd(TFA)2 combined with the DPEphos ligand provides superior yields (up to 90%) compared to other palladium sources like Pd(OAc)2 or PdCl2.
Q: Can this method tolerate diverse substituents on the aromatic ring?
A: Yes, the methodology demonstrates broad substrate scope, successfully accommodating electron-donating groups like methoxy and methyl, as well as electron-withdrawing groups, maintaining high efficiency.
Q: Why is Cu(OTf)2 preferred for the cyclization step?
A: Cu(OTf)2 acts as a potent Lewis acid that facilitates the critical ring-opening of the cyclobutanone moiety, enabling the subsequent intramolecular cyclization to form the naphthalenone core efficiently.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Naphthalenone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing high-quality intermediates for the development of next-generation therapeutics. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and reliability. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of o-alkynyl phenylcyclobutanone or naphthalenone derivative meets the highest industry standards. Our commitment to technical excellence allows us to navigate the complexities of multi-step syntheses, delivering products that facilitate your R&D breakthroughs.
We invite you to collaborate with us to leverage this advanced synthetic technology for your specific applications. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and timeline. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our manufacturing capabilities can accelerate your supply chain and reduce your overall development costs.
