Advanced Palladium-Catalyzed Synthesis of Naphthalenone Derivatives for Scalable Pharmaceutical Manufacturing
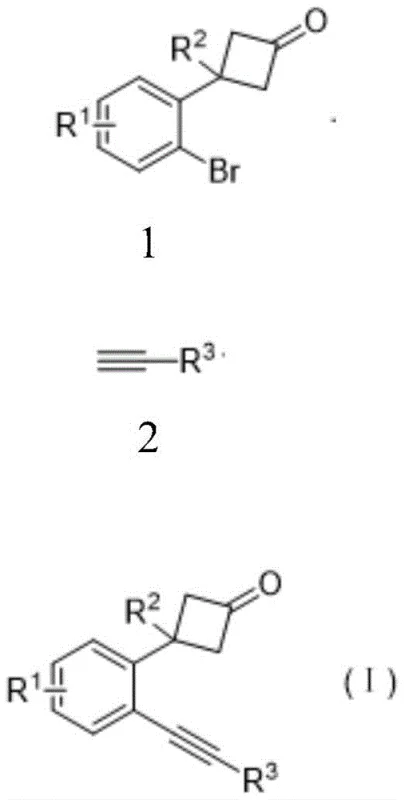
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing complex polycyclic scaffolds, particularly those containing fused ring systems like naphthalenones which serve as critical cores in numerous bioactive molecules. Patent CN112920033A discloses a groundbreaking two-step synthetic strategy that addresses longstanding challenges in the preparation of o-alkynyl phenylcyclobutanones and their subsequent conversion into naphthalenone derivatives. This innovation represents a significant leap forward in process chemistry, offering a streamlined pathway that bypasses the limitations of traditional transition-metal co-catalyzed systems. By leveraging a specialized palladium-catalyzed cross-coupling followed by a Lewis acid-mediated cyclization, the inventors have established a protocol that delivers high purity products with exceptional efficiency. For R&D directors and procurement specialists alike, this technology promises not only improved access to valuable chemical intermediates but also a more sustainable and cost-effective manufacturing route.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of beta-substituted cyclobutanones bearing alkynyl groups has been fraught with difficulties, primarily due to the instability of the four-membered ring under harsh reaction conditions and the propensity for side reactions when using standard catalytic systems. Traditional Sonogashira coupling reactions, which typically rely on a palladium-copper co-catalytic system, often suffer from low yields and poor selectivity when applied to sterically hindered substrates like o-bromophenyl cyclobutanones. The presence of copper salts frequently leads to undesirable homocoupling of the terminal alkyne, known as Glaser coupling, which generates difficult-to-remove impurities that complicate downstream purification. Furthermore, conventional methods often require elevated temperatures and extended reaction times that can degrade the sensitive cyclobutanone moiety, resulting in complex mixtures that are economically unviable for large-scale production. These inefficiencies create significant bottlenecks in the supply chain, driving up costs and extending lead times for critical pharmaceutical intermediates.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes a copper-free palladium catalytic system for the initial coupling step, effectively eliminating the primary source of homocoupling impurities. By employing Pd(TFA)2 in conjunction with the bidentate ligand DPEphos, the reaction achieves remarkable conversion rates at moderate temperatures of 90°C within just 12 hours. This specific catalyst-ligand combination stabilizes the active palladium species, facilitating the oxidative addition and reductive elimination steps necessary for coupling without compromising the integrity of the cyclobutanone ring. The subsequent transformation into naphthalenone is achieved through a distinct Lewis acid-catalyzed cascade using Cu(OTf)2, which triggers a controlled ring-opening and recyclization sequence. This dual-catalyst strategy ensures high atom economy and simplifies the isolation process, as the products can be purified using standard silica gel chromatography with common solvent systems like petroleum ether and ethyl acetate.
Mechanistic Insights into Pd-Catalyzed Coupling and Lewis Acid Cyclization
The mechanistic pathway for the first step involves a classic palladium catalytic cycle initiated by the oxidative addition of the aryl bromide bond to the Pd(0) species generated in situ from the Pd(TFA)2 precursor. The bulky DPEphos ligand plays a crucial role in preventing the aggregation of palladium nanoparticles, thereby maintaining a high concentration of active monomeric catalyst species throughout the reaction duration. Following oxidative addition, the terminal alkyne coordinates to the metal center and undergoes deprotonation by the mild base potassium carbonate, forming a palladium-acetylide intermediate. This intermediate then undergoes transmetallation and subsequent reductive elimination to release the desired o-alkynyl phenylcyclobutanone product while regenerating the active Pd(0) catalyst. The absence of copper co-catalysts in this cycle is a key design feature that prevents the formation of diyne byproducts, ensuring a cleaner reaction profile and higher isolated yields ranging from 50% to over 90% depending on the substrate electronics.
The second stage of the synthesis involves a fascinating cascade reaction mediated by the Lewis acid copper(II) triflate. Upon coordination of the copper catalyst to the carbonyl oxygen of the cyclobutanone, the strained four-membered ring undergoes a selective ring-opening to generate a highly reactive conjugated ketene intermediate. This transient species immediately engages in an intramolecular [2+2] cycloaddition with the proximal alkynyl group, forming a transient cyclobutene-fused intermediate. Subsequently, a retro-[2+2] cycloaddition occurs, driven by the restoration of aromaticity in the newly formed naphthalene ring system, ultimately yielding the stable naphthalenone product. This elegant rearrangement not only constructs the fused ring system efficiently but also installs the ketone functionality at the precise position required for further functionalization, demonstrating the power of strain-release driving forces in organic synthesis.
How to Synthesize o-Alkynyl Phenylcyclobutanone Efficiently
To implement this robust synthetic route in a laboratory or pilot plant setting, strict adherence to the optimized reaction parameters is essential to maximize yield and minimize impurity formation. The process begins with the careful selection of high-purity starting materials, specifically the o-bromophenyl cyclobutanone derivative and the chosen terminal alkyne, which are dissolved in anhydrous 1,4-dioxane to ensure optimal solubility and reaction kinetics. The addition of the palladium catalyst and ligand must be performed under an inert atmosphere to prevent oxidation of the active species, and the reaction mixture is heated to 90°C for a period of 12 hours to ensure complete consumption of the starting material. Detailed operational procedures, including specific stoichiometric ratios and workup protocols, are critical for reproducibility and are outlined in the standardized guide below.
- React o-bromophenyl cyclobutanone with terminal alkyne using Pd(TFA)2 catalyst, DPEphos ligand, and K2CO3 base in 1,4-dioxane at 90°C for 12 hours to form o-alkynyl phenylcyclobutanone.
- Purify the intermediate product using column chromatography with a petroleum ether and ethyl acetate mixture.
- React the purified o-alkynyl phenylcyclobutanone with Cu(OTf)2 catalyst in chloroform at 65°C for 18 hours to effect ring-opening cyclization into the final naphthalenone product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers substantial strategic advantages by addressing key pain points associated with the sourcing of complex heterocyclic intermediates. The elimination of copper co-catalysts in the primary coupling step drastically reduces the burden on downstream purification processes, as there is no need for extensive metal scavenging treatments to remove residual copper traces that often plague pharmaceutical intermediates. This simplification translates directly into reduced processing time and lower consumption of specialized purification resins, leading to significant cost reductions in API manufacturing without compromising on quality standards. Furthermore, the use of commodity chemicals such as potassium carbonate and 1,4-dioxane ensures that the supply chain remains resilient against fluctuations in the availability of exotic reagents, thereby enhancing overall supply security.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived primarily from the high catalytic efficiency and the simplified purification workflow. By achieving yields of up to 90% with only 2.5 mol% of palladium catalyst, the method minimizes the consumption of expensive precious metals, which is a major cost driver in fine chemical synthesis. Additionally, the avoidance of copper salts eliminates the generation of heavy metal waste, reducing the environmental compliance costs associated with waste disposal and treatment. The ability to use standard column chromatography with inexpensive solvent mixtures for purification further lowers the operational expenditure compared to methods requiring preparative HPLC or complex crystallization sequences.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable starting materials ensures a consistent and reliable supply of the final naphthalenone products. Unlike processes that depend on unstable organometallic reagents or cryogenic conditions, this method operates at moderate temperatures using robust catalysts that can be sourced from multiple global suppliers. This flexibility mitigates the risk of production delays caused by single-source dependencies or logistical disruptions. Moreover, the scalability of the reaction, demonstrated by its tolerance to various substituents on the aromatic rings, allows manufacturers to adapt the process quickly to produce different analogues without needing to re-optimize the entire synthetic route from scratch.
- Scalability and Environmental Compliance: From an environmental perspective, the process aligns well with green chemistry principles by maximizing atom economy and minimizing hazardous waste generation. The reaction solvents, such as 1,4-dioxane and chloroform, are widely used in the industry and have established recycling protocols, facilitating their recovery and reuse in a closed-loop system. The high selectivity of the catalysts reduces the formation of side products, meaning less solvent and energy are required for separation and purification steps. This efficiency makes the process highly suitable for commercial scale-up, allowing producers to meet increasing market demand for high-purity pharmaceutical intermediates while maintaining a reduced carbon footprint and adhering to stringent regulatory standards.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthetic technology, providing clarity for potential partners and licensees. These answers are derived directly from the experimental data and comparative studies presented in the patent documentation, ensuring accuracy and relevance for industrial application. Understanding these nuances is vital for assessing the feasibility of integrating this method into existing production lines.
Q: Why is Pd(TFA)2 preferred over other palladium catalysts for this synthesis?
A: According to the patent data, Pd(TFA)2 demonstrates superior catalytic activity compared to alternatives like Pd(OAc)2 or PdCl2, achieving yields up to 90% versus 74% or lower with other catalysts under identical conditions.
Q: What are the advantages of this method over traditional Sonogashira coupling?
A: This method avoids the use of copper co-catalysts in the initial coupling step, which significantly reduces the formation of homocoupling byproducts (Glaser coupling) and simplifies the downstream purification process.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process utilizes readily available raw materials, operates at moderate temperatures (80-100°C), and employs standard workup procedures like column chromatography, making it highly amenable to commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable o-Alkynyl Phenylcyclobutanone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this advanced synthetic route for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of o-alkynyl phenylcyclobutanone or naphthalenone derivative meets the highest industry standards for potency and impurity profiles. We are committed to leveraging our technical expertise to optimize this patented process for your specific needs, delivering cost-effective solutions that accelerate your drug development timelines.
We invite you to collaborate with us to explore how this innovative chemistry can enhance your product portfolio and reduce your overall manufacturing costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities align with your strategic goals for reliable pharmaceutical intermediate supply.
