Revolutionizing Nitrophenylacetonitrile Production: A Solvent-Free Tandem Strategy for Commercial Scale-Up
Introduction to Green Synthesis of Nitrophenylacetonitrile Derivatives
The pharmaceutical and fine chemical industries are constantly seeking more sustainable and efficient pathways for synthesizing complex intermediates. A groundbreaking development in this sector is detailed in patent CN109694335B, which discloses a novel solvent-free preparation method for disubstituted p-nitrophenylacetonitrile derivatives. These compounds serve as critical building blocks in medicinal chemistry, where the nitro and nitrile functional groups offer versatile handles for further transformation into carboxylic acids, amines, or ketones. The significance of this patent lies in its departure from traditional methodologies that rely heavily on organic solvents such as dimethyl sulfoxide or alcohols, which pose environmental hazards and increase production costs. By utilizing a tandem Knoevenagel condensation-reduction strategy followed by alkylation under solvent-free conditions, this technology addresses the urgent industry demand for greener manufacturing processes. For R&D directors and procurement managers alike, understanding the nuances of this patent is essential for evaluating potential supply chain optimizations and cost reduction strategies in the synthesis of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of mono-substituted and disubstituted p-nitrophenylacetonitriles has been plagued by several inefficiencies that hinder large-scale commercial viability. Traditional protocols typically require the use of substantial volumes of organic solvents to facilitate the reaction between p-nitroacetonitrile and aromatic aldehydes. Common solvents include ethanol, methanol, or dipolar aprotic solvents like DMSO, which not only increase the raw material costs but also generate significant volumes of hazardous waste that require expensive disposal procedures. Furthermore, existing methods often suffer from prolonged reaction times, with some prior art reporting durations of up to 24 hours to achieve acceptable conversion rates. The reliance on separate steps for condensation, reduction, and alkylation further complicates the workflow, necessitating intermediate isolation and purification which inevitably leads to material loss and reduced overall yield. These factors collectively contribute to a higher carbon footprint and elevated operational expenditures, making conventional routes less attractive for modern, sustainability-focused manufacturing facilities.
The Novel Approach
In stark contrast, the methodology described in patent CN109694335B introduces a streamlined, solvent-free paradigm that fundamentally reshapes the production landscape for these intermediates. The core innovation involves a one-pot tandem reaction where p-nitroacetonitrile, aromatic aldehydes, a base, and a dihydropyridine ester are mixed directly without any solvent medium. This mixture is heated to a moderate temperature range of 80-100°C, initiating a Knoevenagel condensation immediately followed by an in-situ reduction. The elimination of solvent not only drastically reduces the reaction volume but also enhances the effective concentration of reactants, thereby accelerating the reaction kinetics. Following this initial transformation, benzyl bromide and additional base are introduced directly into the same vessel to effect alkylation, completing the synthesis of the disubstituted target in a highly integrated manner. This approach simplifies the operational workflow, minimizes energy consumption associated with solvent removal, and significantly shortens the total processing time to between 0.5 and 12 hours, representing a substantial improvement over legacy technologies.
Mechanistic Insights into Solvent-Free Tandem Catalysis
The chemical elegance of this process lies in the synergistic interplay between the base catalyst and the dihydropyridine ester hydrogen source. In the first stage, the base deprotonates the active methylene group of the p-nitroacetonitrile, generating a nucleophilic carbanion that attacks the carbonyl carbon of the aromatic aldehyde. This initiates the Knoevenagel condensation, forming an alpha,beta-unsaturated nitrile intermediate. Crucially, instead of isolating this unstable intermediate, the dihydropyridine ester acts as a mild and selective hydride donor, reducing the double bond to yield the mono-substituted saturated nitrile. The choice of base is pivotal; experimental data within the patent demonstrates that organic amine bases, particularly N,N-diisopropylethylamine (DIEA), provide superior performance compared to inorganic bases like potassium carbonate. DIEA likely offers better solubility in the molten reaction mixture and optimal basicity to drive both the condensation and the subsequent alkylation steps efficiently.
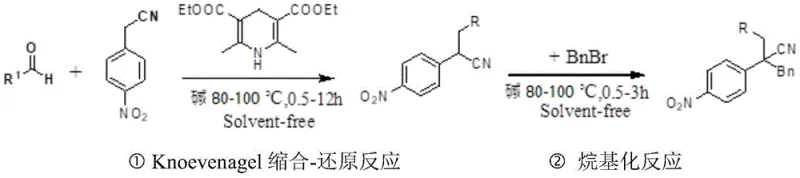
Following the reduction phase, the reaction mixture undergoes an alkylation step where the newly formed acidic proton at the benzylic position is abstracted by the base. The resulting carbanion then performs a nucleophilic substitution on the added benzyl bromide, installing the second substituent to form the final disubstituted product. The solvent-free environment plays a critical role here by preventing the dilution of reactive species, which helps drive the equilibrium towards the product side. Moreover, the use of dihydropyridine esters as hydrogen sources avoids the toxicity and safety concerns associated with traditional reducing agents like metal hydrides. This mechanistic pathway ensures high chemoselectivity, preserving sensitive functional groups on the aromatic rings while achieving high yields, often exceeding 85% for a wide variety of substrates including those with electron-withdrawing or electron-donating substituents.
How to Synthesize Disubstituted p-Nitrophenylacetonitrile Efficiently
Implementing this solvent-free protocol requires precise control over stoichiometry and thermal conditions to maximize yield and purity. The process begins with the careful weighing of p-nitroacetonitrile, the chosen aromatic aldehyde, the base catalyst, and the dihydropyridine ester in a molar ratio of approximately 1:1.2:1:1.2. These components are charged into a reaction vessel equipped with mechanical stirring and heating capabilities. The mixture is then heated to a temperature between 80°C and 100°C and maintained under agitation for a period ranging from 0.5 to 12 hours, depending on the specific reactivity of the aldehyde substrate. Once the initial condensation-reduction is complete, a second charge of base and benzyl bromide is added directly to the hot reaction mass. The temperature is maintained or slightly adjusted to 80-100°C for an additional 0.5 to 3 hours to ensure complete alkylation. Detailed standard operating procedures for scaling this reaction are provided below.
- Mix p-nitroacetonitrile, aromatic aldehyde, base, and dihydropyridine ester in a molar ratio of 1: (1-2):(0.2-1.5):(1-2) and heat to 80-100°C for 0.5-12h.
- Add base and benzyl bromide to the reaction mixture in a molar ratio of 1: (1-4):(1-4) relative to the starting nitrile, then heat to 80-100°C for 0.5-3h.
- Purify the crude product via silica gel column chromatography using a petroleum ether and ethyl acetate mixture to isolate the final derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this solvent-free technology translates into tangible strategic benefits that extend beyond simple chemical yield. The most immediate impact is seen in the drastic simplification of the downstream processing workflow. By eliminating the need for organic solvents, manufacturers can bypass energy-intensive distillation steps required for solvent recovery and removal. This reduction in unit operations not only lowers utility costs but also decreases the turnaround time for each batch, effectively increasing the throughput of existing production assets. Furthermore, the absence of volatile organic compounds (VOCs) significantly reduces the regulatory burden associated with emissions control and worker safety, aligning production practices with increasingly stringent global environmental standards. These operational efficiencies cumulatively contribute to a more resilient and cost-effective supply chain for critical pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The economic implications of removing solvents from the process are profound. Solvents often constitute a major portion of the raw material cost in fine chemical synthesis, and their disposal represents a significant hidden expense. By operating under solvent-free conditions, the process eliminates these costs entirely. Additionally, the use of dihydropyridine esters as hydrogen sources avoids the need for expensive transition metal catalysts or hazardous metal hydrides, further optimizing the bill of materials. The high yields reported, often surpassing 90% for optimized substrates, mean that less raw material is wasted, directly improving the cost of goods sold (COGS). This lean manufacturing approach allows for competitive pricing structures without compromising on quality or margin.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the availability and price volatility of specialized solvents and reagents. This novel method relies on commodity chemicals such as aromatic aldehydes, benzyl bromide, and common amine bases, which are widely available from multiple global suppliers. The robustness of the reaction conditions, tolerating temperatures up to 100°C without degradation, ensures consistent batch-to-batch quality even with minor variations in raw material specifications. This flexibility reduces the risk of production stoppages due to supply shortages. Moreover, the simplified logistics of handling non-volatile reaction masses reduce transportation and storage hazards, making the supply chain more agile and responsive to market demands for high-purity intermediates.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to industrial reactors often encounters heat transfer and mixing limitations, particularly when solvents are involved. The solvent-free nature of this reaction mitigates many of these engineering challenges, as the reaction mass itself acts as the heat transfer medium. This facilitates easier scale-up from kilogram to multi-ton quantities. From an environmental perspective, the E-factor (mass of waste per mass of product) is significantly improved due to the lack of solvent waste. This aligns perfectly with the principles of Green Chemistry, helping companies meet their sustainability goals and corporate social responsibility targets. The ability to produce complex intermediates with a minimal environmental footprint is a powerful differentiator in the global marketplace.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this solvent-free synthesis technology. These insights are derived directly from the experimental data and beneficial effects outlined in the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their specific product portfolios.
Q: What are the primary advantages of this solvent-free method over traditional solvent-based synthesis?
A: The solvent-free approach eliminates the need for volatile organic compounds like alcohols or DMSO, significantly reducing environmental impact and downstream purification costs while shortening reaction times from 24 hours to under 12 hours.
Q: Which base catalysts provide the highest yields in this tandem reaction sequence?
A: Experimental data indicates that organic amine bases, specifically N,N-diisopropylethylamine (DIEA), outperform inorganic bases like potassium carbonate, achieving yields up to 92% in the initial condensation-reduction step.
Q: Is this synthetic route scalable for industrial production of pharmaceutical intermediates?
A: Yes, the absence of solvents simplifies thermal management and reactor design, making the process highly suitable for commercial scale-up while maintaining high chemoselectivity and product purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Disubstituted p-Nitrophenylacetonitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the solvent-free synthesis technology described in patent CN109694335B for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate such innovative laboratory protocols into robust, commercial-scale manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this green chemistry approach are fully realized in an industrial setting. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of disubstituted p-nitrophenylacetonitrile derivative meets the exacting standards required by the global pharmaceutical industry.
We invite you to collaborate with us to leverage this advanced synthetic route for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. By partnering with us, you gain access to a reliable supply chain capable of delivering high-purity intermediates with reduced lead times. Please contact us today to request specific COA data and route feasibility assessments, and let us help you optimize your manufacturing strategy with cutting-edge, sustainable chemistry solutions.
