Advanced Rhodium-Catalyzed Synthesis of Benzo[a]carbazoles for High-Purity Pharmaceutical and OLED Intermediates
The pharmaceutical and advanced materials industries are constantly seeking robust methodologies for constructing complex nitrogen-containing heterocycles, particularly benzo[a]carbazole scaffolds which are pivotal in drug discovery and organic light-emitting diode (OLED) technologies. Patent CN108929262B introduces a groundbreaking synthetic strategy that addresses long-standing challenges in this domain by utilizing a Rhodium(III)-catalyzed cascade reaction. This innovative approach leverages readily available 2-arylindole compounds and sulfur ylides as primary building blocks to efficiently construct the benzo[a]carbazole core. The significance of this technology lies not only in its chemical elegance but also in its potential to streamline the supply chain for high-value intermediates. By enabling a direct, one-pot transformation under relatively mild conditions, this method offers a compelling alternative to traditional multi-step syntheses, positioning it as a critical asset for manufacturers aiming to enhance production efficiency and reduce environmental impact in the synthesis of functional materials and active pharmaceutical ingredients.
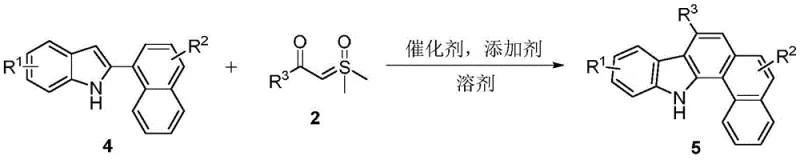
![General reaction scheme for Rh(III)-catalyzed synthesis of benzo[a]carbazoles from 2-arylindoles and sulfur ylides](/insights/img/benzo-a-carbazole-rhodium-catalysis-pharma-supplier-20260303010730-01.webp)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of benzo[a]carbazole frameworks has been plagued by significant synthetic hurdles that hinder large-scale adoption. Traditional routes often rely on harsh reaction conditions, including extremely high temperatures and the use of corrosive or toxic reagents that pose safety risks and complicate waste management. Furthermore, many existing methodologies suffer from poor atom economy, generating substantial amounts of chemical waste due to the requirement for multiple protection and deprotection steps. The starting materials for these conventional processes are frequently expensive, difficult to source commercially, or require complex pre-synthesis, which drives up the overall cost of goods sold. Additionally, the purification of intermediates in multi-step sequences often leads to yield losses and increased processing time, creating bottlenecks in the supply chain that affect the reliability of delivery for downstream clients seeking reliable pharmaceutical intermediates supplier partnerships.
The Novel Approach
In stark contrast, the methodology disclosed in the patent data presents a streamlined, atom-economical solution that fundamentally reshapes the production landscape. By employing a Rh(III)-catalyzed tandem reaction, this novel approach allows for the direct coupling of 2-arylindoles with sulfur ylides in a single operational step. The reaction proceeds efficiently at a moderate temperature of 100°C in common solvents like tetrahydrofuran, eliminating the need for extreme thermal inputs. Crucially, the process is tolerant to air, meaning it does not require rigorous inert atmosphere techniques such as gloveboxes or extensive nitrogen purging, which drastically simplifies the engineering controls needed for reactor setup. This simplicity translates directly into cost reduction in fine chemical manufacturing, as it lowers both capital expenditure on specialized equipment and operational expenditure on energy and gases. The broad substrate scope further enhances its utility, allowing for the rapid generation of diverse analogues essential for structure-activity relationship (SAR) studies in drug development.
Mechanistic Insights into Rh(III)-Catalyzed C-H Activation and Cyclization
The core of this transformative synthesis lies in the sophisticated mechanism driven by the pentamethylcyclopentadienyl rhodium(III) catalyst. The reaction initiates with the coordination of the rhodium species to the nitrogen atom of the 2-arylindole substrate, facilitating a directed C-H activation at the proximal position of the aryl ring. This metallacycle intermediate is highly reactive and undergoes insertion with the sulfur ylide reagent, a key step that forms the new carbon-carbon bond necessary for ring expansion. Following the insertion, a series of elimination and cyclization events occur, ultimately releasing the sulfur byproduct and aromatizing the system to yield the stable benzo[a]carbazole structure. The use of cesium acetate as an additive plays a critical role in regenerating the active catalytic species and neutralizing acidic byproducts, ensuring the catalytic cycle continues efficiently without deactivation. Understanding this mechanistic pathway is vital for R&D directors aiming to optimize reaction parameters for specific substrates.
From an impurity control perspective, the specificity of the Rh(III) catalyst ensures high regioselectivity, minimizing the formation of isomeric byproducts that are common in non-directed C-H functionalization reactions. The mild conditions prevent the degradation of sensitive functional groups such as halogens or esters that might be present on the substrate, thereby preserving the integrity of the molecule for further derivatization. The patent data highlights that even with electron-deficient or electron-rich substituents, the reaction maintains consistent performance, yielding products with high purity profiles as confirmed by NMR and HRMS analysis. This level of control over the impurity profile is essential for meeting the stringent quality standards required for high-purity benzo[a]carbazoles used in electronic materials, where trace impurities can quench luminescence or alter charge transport properties significantly.
How to Synthesize Benzo[a]carbazole Derivatives Efficiently
Implementing this synthesis route requires careful attention to reagent stoichiometry and reaction monitoring to maximize yield and throughput. The standard protocol involves dissolving the 2-arylindole starting material in tetrahydrofuran, followed by the sequential addition of the sulfur ylide, the rhodium dimer catalyst, and the cesium acetate additive. The mixture is then sealed and heated to 100°C for approximately 12 hours. Post-reaction workup is straightforward, involving aqueous extraction and standard silica gel chromatography, which facilitates easy isolation of the target compound. For process chemists looking to adapt this for larger batches, maintaining the correct molar ratios—specifically a slight excess of the sulfur ylide (1:1.5 ratio)—has been shown to drive the reaction to completion effectively. The detailed standardized synthesis steps see the guide below for specific operational parameters.
- Dissolve the 2-arylindole substrate in tetrahydrofuran (THF) within a reaction vessel.
- Add the sulfur ylide reagent, [RhCp*Cl2]2 catalyst, and cesium acetate additive sequentially to the mixture.
- Seal the reaction tube under air conditions and heat to 100°C for 12 hours to complete the cascade cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this Rh(III)-catalyzed technology offers tangible strategic benefits beyond mere chemical novelty. The primary advantage is the significant simplification of the manufacturing process, which directly correlates to enhanced supply chain reliability. By reducing the number of unit operations from multiple steps to a single pot, the risk of batch failure is minimized, and the lead time for production is drastically shortened. The use of commercially available and inexpensive starting materials like 2-arylindoles ensures a stable supply base, mitigating the risks associated with sourcing exotic or custom-synthesized precursors. Furthermore, the ability to run the reaction under air conditions removes the dependency on high-purity inert gases, reducing utility costs and logistical complexities associated with gas supply chains in remote manufacturing sites.
- Cost Reduction in Manufacturing: The elimination of multiple synthetic steps and the avoidance of expensive protecting group chemistry result in substantial cost savings. The high atom economy of the cascade reaction means less raw material is wasted, and the simplified workup reduces solvent consumption and waste disposal fees. Additionally, the catalyst loading is relatively low, and the robustness of the system allows for potential catalyst recovery strategies, further driving down the cost per kilogram of the final API intermediate.
- Enhanced Supply Chain Reliability: The operational simplicity of this method allows for greater flexibility in manufacturing scheduling. Since the reaction does not require specialized high-pressure equipment or cryogenic conditions, it can be performed in standard glass-lined or stainless steel reactors available in most multipurpose plants. This compatibility with existing infrastructure ensures that production can be scaled up rapidly to meet market demand without significant capital investment, ensuring consistent availability of critical intermediates for downstream drug substance manufacturing.
- Scalability and Environmental Compliance: The mild reaction conditions and the use of common organic solvents like THF make this process highly scalable from gram to tonnage levels. The reduced generation of hazardous waste aligns with modern green chemistry principles and increasingly strict environmental regulations. By minimizing the environmental footprint, manufacturers can avoid regulatory delays and maintain a sustainable production profile, which is becoming a key differentiator in securing contracts with major multinational pharmaceutical companies focused on ESG goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this benzo[a]carbazole synthesis technology. These answers are derived directly from the experimental data and claims within the patent documentation to provide accurate guidance for technical teams evaluating this route for adoption. Understanding these nuances is critical for assessing the feasibility of integrating this chemistry into existing production lines or new product development pipelines.
Q: What are the optimal reaction conditions for this Rh(III)-catalyzed synthesis?
A: According to patent CN108929262B, the optimal conditions involve using tetrahydrofuran (THF) as the solvent at 100°C for 12 hours, with [RhCp*Cl2]2 as the catalyst and cesium acetate as the additive.
Q: Does this method require strict inert gas protection?
A: No, the patent specifies that the reaction can be conducted under air conditions by simply sealing the reaction tube, which significantly simplifies the operational requirements for large-scale manufacturing.
Q: What is the substrate scope for this benzo[a]carbazole synthesis?
A: The method demonstrates a wide substrate scope, tolerating various substituents such as fluoro, chloro, methyl, and trifluoromethyl groups on both the indole and the aryl rings, as well as naphthalene-fused variants.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzo[a]carbazole Supplier
As the demand for complex heterocyclic scaffolds grows in the pharmaceutical and optoelectronic sectors, having a partner with deep technical expertise is essential. NINGBO INNO PHARMCHEM stands ready to support your development needs with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle sensitive organometallic reactions safely and efficiently, ensuring that every batch meets stringent purity specifications. We utilize rigorous QC labs to verify the identity and quality of our products, providing full traceability and documentation that satisfies the compliance requirements of global regulatory bodies. Our commitment to quality ensures that you receive high-purity benzo[a]carbazoles suitable for the most demanding applications.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a reliable supply chain partner dedicated to accelerating your time-to-market while optimizing your overall manufacturing costs through innovative chemical solutions.
