Advanced Iridium-Catalyzed Synthesis of Polysubstituted Oxazoles for Pharmaceutical Applications
Introduction to Patent CN115536605A Technology
The pharmaceutical industry continuously seeks more efficient pathways to construct complex heterocyclic scaffolds, particularly those serving as core structures for potent anti-inflammatory agents. Patent CN115536605A introduces a groundbreaking methodology for the preparation of polysubstituted oxazole compounds, which are pivotal intermediates in the synthesis of COX-2 inhibitors. This technology leverages a trivalent metal catalyst, specifically iridium or rhodium complexes, to facilitate multiple dehydrogenation and 1,1-difunctionalization reactions of N-(1-arylvinyl)amides with diverse boron reagents. By employing a weak coordination-directed strategy, the process utilizes the enamine moiety as a traceless directing group, thereby overcoming significant hurdles associated with traditional C-H activation. This innovation not only enhances regioselectivity and chemoselectivity but also expands the substrate scope to include secondary alkyl boron reagents and strongly coordinating heterocycles, representing a substantial leap forward in synthetic organic chemistry for drug discovery.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of multi-substituted oxazole rings, essential for COX-2 inhibitor development, has relied heavily on classical condensation reactions or transition-metal catalyzed cross-couplings like the Suzuki-Miyaura reaction. However, these conventional methods suffer from inherent limitations that impede efficient manufacturing. The Suzuki-Miyaura reaction, while robust for Csp2-Csp2 bond formation, often struggles when extending coupling to sp3 carbon centers due to slow transmetallation rates that mismatch with beta-hydride elimination processes. Furthermore, traditional approaches frequently necessitate the pre-functionalization of starting materials, requiring energy-intensive and waste-generating halogenation or metallation steps prior to the core ring construction. These multi-step sequences reduce overall atom economy and complicate the purification process, leading to higher production costs and longer lead times for critical pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN115536605A offers a streamlined, one-step solution that bypasses the need for pre-functionalized substrates. By utilizing N-(1-arylvinyl)amides as readily available starting materials, the process achieves direct olefin 1,1-dehydrogenative difunctionalization through a cascade of C-H activation events. The use of a trivalent metal catalyst in conjunction with a silver salt oxidant promotes a unique mechanism where the amide group acts as a weak, traceless directing group. This allows for the precise activation of specific C-H bonds without permanent modification of the molecular scaffold. Consequently, this approach enables the direct incorporation of alkyl, alkenyl, and aryl groups from boron reagents, including challenging secondary alkyl variants, into the oxazole core with high fidelity. This represents a paradigm shift towards more sustainable and step-economical synthesis of complex drug-like molecules.

Mechanistic Insights into Ir-Catalyzed C-H Activation and Cyclization
The core of this technological advancement lies in the sophisticated catalytic cycle driven by trivalent iridium or rhodium species. The mechanism initiates with the coordination of the trivalent metal catalyst to the weakly coordinating amide oxygen or nitrogen of the N-(1-arylvinyl)amide substrate. This interaction facilitates the activation of the adjacent vinyl C-H bond, forming a metallacycle intermediate. Subsequently, the boron reagent undergoes transmetallation with the metal center, a step that is critically enabled by the presence of silver salts which act as halide scavengers to generate the active cationic metal species. Unlike traditional systems that might stall with sp3 boron reagents, this catalytic system is uniquely tuned to accommodate the steric and electronic properties of secondary alkyl boron acids. Following transmetallation, reductive elimination or a similar bond-forming step constructs the new carbon-carbon bond, followed by enol tautomerization and intramolecular oxidative cyclization to close the oxazole ring.
Impurity control is inherently managed through the high chemoselectivity of the weak coordination strategy. Because the directing group is traceless and the activation is site-specific, side reactions such as non-selective polymerization or homocoupling of the boron reagent are minimized. The use of specific additives, such as sodium pivalate or sodium trifluoroacetate, further modulates the acidity of the medium and stabilizes the catalytic species, ensuring that the reaction proceeds cleanly to the desired polysubstituted oxazole. This precision is particularly evident when handling substrates with sensitive functional groups like halogens or strong coordinating nitrogens, which remain intact throughout the transformation. The result is a high-purity product profile that reduces the burden on downstream purification processes, a critical factor for maintaining cost-efficiency in commercial pharmaceutical manufacturing.
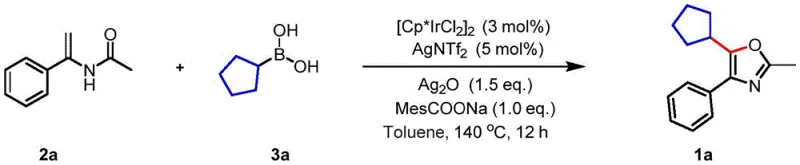
How to Synthesize Polysubstituted Oxazoles Efficiently
Implementing this synthesis route requires careful attention to the stoichiometry of the catalyst system and the choice of oxidant to ensure optimal turnover. The protocol generally involves mixing the N-(1-arylvinyl)amide substrate with an excess of the chosen boron reagent, typically in a 1:1.5 molar ratio, to drive the equilibrium towards product formation. The catalyst loading is kept low, often around 3 mol%, making the process economically viable for large-scale operations. The reaction is conducted in inert solvents such as toluene at elevated temperatures ranging from 120°C to 140°C for a duration of 12 to 24 hours. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by combining N-(1-arylvinyl)amide, boron reagent, trivalent metal catalyst (e.g., [Cp*IrCl2]2), silver salt oxidant, and additive in an inert solvent like toluene.
- Heat the reaction mixture to a temperature range of 120-140°C and maintain stirring for 12 to 24 hours under atmospheric conditions to facilitate the oxidative cyclization.
- Upon completion, isolate the crude product and purify it using column chromatography with a petroleum ether and ethyl acetate eluent system to obtain the high-purity target oxazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers tangible strategic benefits that extend beyond mere chemical novelty. The primary advantage lies in the drastic simplification of the raw material supply chain. By eliminating the need for pre-halogenated intermediates, manufacturers can source cheaper, more abundant starting materials like simple vinyl amides and commercially available boronic acids. This reduction in precursor complexity directly translates to cost reduction in pharmaceutical intermediates manufacturing, as fewer synthetic steps mean lower labor, energy, and waste disposal costs. Furthermore, the high atom economy of the cross-dehydrogenative coupling, which produces water or hydrogen as the primary byproduct, aligns perfectly with modern green chemistry initiatives and environmental compliance standards.
- Cost Reduction in Manufacturing: The elimination of expensive palladium catalysts often used in traditional cross-couplings, replaced by more robust iridium or rhodium systems that operate at low loadings, significantly lowers catalyst costs. Additionally, the avoidance of pre-functionalization steps removes the need for hazardous halogenating agents and the associated waste treatment expenses. This streamlined process flow reduces the overall cost of goods sold (COGS) for high-purity polysubstituted oxazoles, allowing for more competitive pricing in the global market while maintaining healthy margins.
- Enhanced Supply Chain Reliability: The reliance on commercially available boron reagents, including alkyl, alkenyl, and aryl variants, ensures a stable and diversified supply base. Unlike specialized halogenated intermediates which may have limited suppliers and long lead times, boronic acids are commodity chemicals produced by multiple vendors worldwide. This diversity mitigates the risk of supply disruptions and reduces lead time for high-purity pharmaceutical intermediates. The robustness of the reaction conditions, which tolerate air and moisture better than many sensitive organometallic processes, further enhances operational reliability and batch-to-batch consistency.
- Scalability and Environmental Compliance: The use of common solvents like toluene and the absence of cryogenic conditions make this process highly scalable from gram to ton quantities. The reaction's compatibility with functional groups means that complex molecules can be built later in the synthesis tree, reducing the risk of losing value-added materials in early stages. Moreover, the generation of benign byproducts simplifies effluent treatment, facilitating easier regulatory approval and environmental compliance. This scalability supports the commercial scale-up of complex pharmaceutical intermediates, ensuring that supply can meet demand spikes for critical anti-inflammatory medications without compromising quality.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxazole synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the process capabilities and limitations for potential partners and licensees.
Q: What is the primary advantage of this oxazole synthesis method over traditional Suzuki coupling?
A: Unlike traditional Suzuki-Miyaura reactions which are often limited to Csp2-Csp2 bond formation and require pre-halogenated substrates, this method utilizes a traceless enamine directing group to achieve direct C-H activation and 1,1-difunctionalization. This eliminates the need for prior halogenation or metallation steps, significantly improving atom economy and step efficiency while allowing for the use of secondary alkyl boron reagents which are typically challenging in standard cross-coupling protocols.
Q: Can this process tolerate strong coordinating groups like pyridine?
A: Yes, a significant breakthrough of this patented technology is its ability to tolerate and even utilize boron reagents containing strong coordinating heterocycles such as pyridine. Conventional strategies often fail due to catalyst poisoning by strong nitrogen ligands, but the weak coordination-directed strategy employed here ensures effective catalyst turnover and site-selective activation, enabling the synthesis of complex heterocyclic substituted oxazoles that were previously difficult to access.
Q: What are the typical reaction conditions for scaling this synthesis?
A: The process operates under relatively robust conditions suitable for scale-up, utilizing common inert solvents like toluene and temperatures between 120°C and 140°C. The reaction proceeds under atmospheric air conditions without the need for stringent inert gas protection for the setup, although standard Schlenk techniques are used in the examples. The use of commercially available oxidants like silver oxide and additives like sodium pivalate further simplifies the supply chain requirements for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Oxazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic methodologies described in patent CN115536605A for the production of next-generation COX-2 inhibitors. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to market supply is seamless. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of polysubstituted oxazole intermediates meets the highest international standards for pharmaceutical applications. We are committed to leveraging our technical expertise to optimize this iridium-catalyzed process for your specific needs.
We invite you to collaborate with us to unlock the full commercial potential of this advanced chemistry. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your project volume and timeline. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our manufacturing capabilities can accelerate your drug development pipeline while optimizing your supply chain efficiency.
