Advanced Asymmetric Catalysis for High-Purity Axial Chiral Indole Intermediates
The pharmaceutical industry is constantly seeking robust methodologies to access complex chiral scaffolds that serve as critical building blocks for next-generation therapeutics. A significant breakthrough in this domain is documented in Chinese Patent CN110467555B, which discloses a novel synthetic route for axial chiral aryl indole compounds. These molecules, characterized by their restricted rotation around a single bond creating stable atropisomers, represent a privileged structure in medicinal chemistry due to their unique three-dimensional architecture. The patent specifically highlights the utility of these compounds as potential anticancer agents, noting their cytotoxic activity against MCF-7 breast cancer cell lines. For R&D directors and procurement specialists alike, this technology offers a compelling value proposition: a streamlined, metal-free pathway to high-value intermediates that bridges the gap between academic innovation and commercial viability. By leveraging asymmetric organocatalysis, this method addresses the longstanding challenge of constructing axially chiral indole-naphthalene and indole-benzene skeletons with exceptional stereocontrol.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of axially chiral biaryl systems containing indole moieties has been fraught with synthetic challenges. Traditional approaches often rely on direct coupling reactions between indole rings and naphthalene or benzene rings, which frequently suffer from limited substrate scope and poor stereochemical outcomes. Prior art, such as methods reported in Angewandte Chemie (2017) and Nature Chemistry (2018), typically involves the coupling of 2-indolecarbinols with naphthols or the reaction of 2-substituted indoles with azonaphthalenes. While effective in specific contexts, these legacy methods often demand rigorous reaction conditions, expensive transition metal catalysts, or multi-step sequences that degrade overall atom economy. Furthermore, the reliance on specific functional groups limits the structural diversity accessible to medicinal chemists, creating a bottleneck in the rapid exploration of chemical space for drug discovery programs. The inability to efficiently resolve racemic mixtures into single enantiomers without substantial yield loss has also been a persistent hurdle in the field.
The Novel Approach
The methodology outlined in patent CN110467555B represents a paradigm shift by employing a dynamic kinetic resolution (DKR) strategy mediated by chiral phosphoric acid catalysts. This innovative route utilizes readily available racemic indole derivatives (Formula 1 or Formula 4) and azodicarboxylates (Formula 2) as starting materials. The reaction proceeds smoothly in dichloromethane at ambient temperatures, typically between 20°C and 30°C, eliminating the need for energy-intensive heating or cryogenic cooling. As illustrated in the general reaction scheme below, this single-step transformation efficiently constructs the axially chiral skeleton while simultaneously installing hydrazine functionality, which can be further derivatized. The process is remarkably tolerant of various substituents, allowing for the introduction of halogens, esters, and alkoxy groups without compromising yield or selectivity.
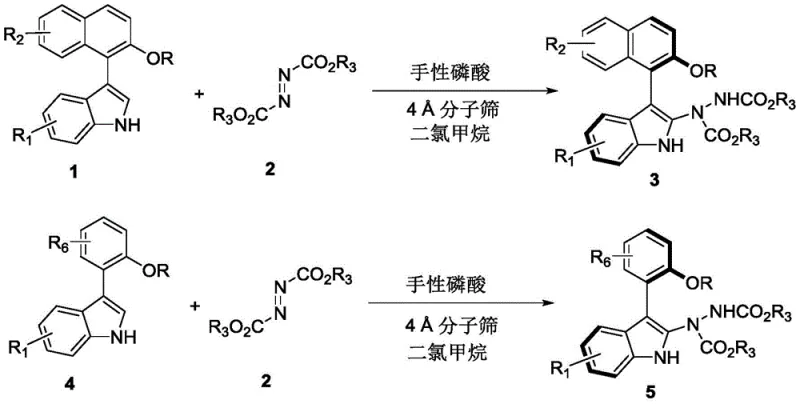
This versatility makes the process an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing, as it simplifies the supply chain by reducing the number of synthetic steps and purification operations required to reach the target molecule.
Mechanistic Insights into Chiral Phosphoric Acid Catalyzed Asymmetric Addition
The core of this technological advancement lies in the precise activation of substrates through dual hydrogen-bonding interactions facilitated by the chiral phosphoric acid catalyst. The catalyst, specifically the binaphthyl-derived phosphoric acid designated as Compound 61 in the patent, acts as a bifunctional organocatalyst. It simultaneously activates the electrophilic azodicarboxylate and the nucleophilic indole species within a well-defined chiral pocket. This supramolecular assembly lowers the activation energy for the C-N bond formation while imposing strict steric constraints that favor the formation of one atropisomer over the other. The result is a highly enantioselective transformation, with experimental data showing enantiomeric ratios (er) reaching as high as 98:2 for certain substrates. Such high optical purity is critical for pharmaceutical applications, where the biological activity is often confined to a single enantiomer, and the presence of the wrong isomer can lead to toxicity or reduced efficacy.
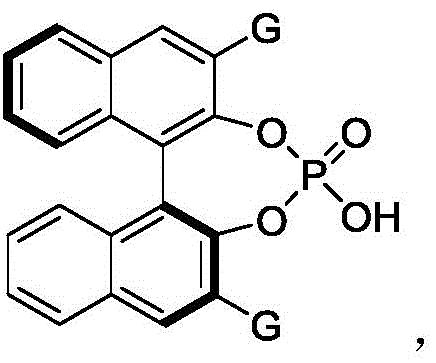
From an impurity control perspective, the mechanism inherently suppresses side reactions. The mild acidic nature of the phosphoric acid prevents the decomposition of sensitive functional groups that might occur under stronger acidic or basic conditions typical of traditional coupling reagents. Furthermore, the use of 4 Å molecular sieves as an additive plays a crucial role in sequestering trace water, which could otherwise hydrolyze the azodicarboxylate reagent or deactivate the catalyst. This attention to reaction hygiene ensures a cleaner crude reaction profile, significantly reducing the burden on downstream purification processes like silica gel column chromatography. For quality assurance teams, this translates to a more consistent impurity profile and easier validation of the manufacturing process.
How to Synthesize Axial Chiral Aryl Indole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and stereoselectivity. The protocol dictates the use of dichloromethane as the solvent of choice due to its ability to dissolve both organic substrates and the catalyst effectively while maintaining a liquid phase at the optimal reaction temperature of 25°C. The stoichiometry is flexible, with a molar ratio of indole substrate to azodicarboxylate ranging from 1:1 to 1:3, though a 1:3 ratio is often preferred to drive the equilibrium towards product formation. The addition of activated 4 Å molecular sieves is non-negotiable for maintaining anhydrous conditions essential for catalyst longevity. Following the reaction, which is monitored via TLC until completion, the workup involves simple filtration to remove the sieves and catalyst residues, followed by concentration and purification. For a detailed breakdown of the standardized operating procedures and safety protocols, please refer to the technical guide below.
- Prepare the reaction mixture by combining the racemic indole substrate (Formula 1 or 4) and the azodicarboxylate reagent (Formula 2) in dichloromethane solvent.
- Add activated 4 Å molecular sieves and the chiral phosphoric acid catalyst (specifically Compound 61) to the reaction vessel under inert atmosphere.
- Stir the mixture at ambient temperature (20-30°C) until TLC indicates completion, then filter, concentrate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this organocatalytic route offers tangible strategic benefits beyond mere chemical elegance. The shift away from transition metal catalysis eliminates the need for expensive and potentially toxic heavy metals like palladium or rhodium, which are subject to volatile market pricing and stringent regulatory limits regarding residual levels in final drug products. By removing these metals from the process, manufacturers can achieve significant cost savings associated with raw material acquisition and, more importantly, the extensive purification steps required to meet ppm-level metal specifications. This simplification of the downstream processing directly contributes to cost reduction in API manufacturing, allowing for tighter margins and more competitive pricing for the final active pharmaceutical ingredient.
- Cost Reduction in Manufacturing: The economic advantages of this method are multifaceted. Firstly, the catalyst loading is relatively low (typically 10 mol%), and the catalyst itself is derived from abundant binaphthol precursors, avoiding the supply chain risks associated with rare earth metals. Secondly, the reaction operates at room temperature, drastically reducing energy consumption compared to processes requiring reflux or cryogenic conditions. Thirdly, the high atom economy and yield (often exceeding 90% for optimized substrates) mean less waste generation and lower disposal costs. The elimination of metal scavenging resins and the associated validation testing further streamlines the cost structure, making the overall production economics highly favorable for large-scale campaigns.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions enhances supply chain resilience. Since the reagents—substituted indoles and azodicarboxylates—are commercially available or easily synthesized from commodity chemicals, the risk of raw material shortages is minimized. The tolerance of the reaction to various functional groups means that a single platform technology can be applied to synthesize a diverse library of analogs without needing to requalify entirely new synthetic routes for each derivative. This flexibility allows supply chain planners to respond rapidly to changes in clinical demand or pipeline priorities, ensuring continuity of supply for critical drug development programs without the long lead times typically associated with developing bespoke metal-catalyzed processes.
- Scalability and Environmental Compliance: From an environmental and scalability standpoint, this process aligns perfectly with green chemistry principles. The use of dichloromethane, while requiring proper handling, is a standard industrial solvent with well-established recovery and recycling infrastructure. The absence of heavy metals simplifies wastewater treatment and reduces the environmental footprint of the manufacturing site. The mild conditions also imply that the reaction can be scaled from gram to kilogram to tonne scales with minimal re-optimization, as heat transfer and mixing issues common in exothermic metal-catalyzed reactions are largely mitigated. This ease of scale-up reduces the time-to-market for new drugs, a critical factor in the highly competitive pharmaceutical landscape.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this axial chiral indole synthesis technology. These answers are derived directly from the experimental data and embodiments provided in the source patent documentation, ensuring accuracy and relevance for technical decision-makers evaluating this route for their own production needs.
Q: What is the primary advantage of this chiral phosphoric acid method over traditional coupling reactions?
A: Unlike traditional coupling methods that often require harsh conditions or specific pre-functionalized substrates, this organocatalytic approach utilizes dynamic kinetic resolution under mild conditions (25°C), achieving high enantioselectivity (up to 98:2 er) without transition metals.
Q: What biological activity do these axial chiral indole compounds exhibit?
A: Biological testing described in the patent indicates significant cytotoxic activity against MCF-7 breast cancer cells, with lead compound 3aa demonstrating an IC50 value of 4.44 μg/mL, highlighting their potential as oncology drug leads.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the process uses economical solvents like dichloromethane, operates at room temperature, and employs robust organocatalysts, making it highly scalable and safer for commercial manufacturing compared to cryogenic or high-pressure alternatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axial Chiral Aryl Indole Supplier
The synthesis of complex chiral intermediates requires not just a patented method, but a partner with the expertise to execute it flawlessly at scale. NINGBO INNO PHARMCHEM stands ready to support your drug development pipeline with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with state-of-the-art reactors capable of maintaining the precise temperature control and anhydrous conditions required for this organocatalytic process. We understand that in the race to bring life-saving oncology therapies to market, consistency is key. That is why our rigorous QC labs enforce stringent purity specifications, ensuring that every batch of axial chiral indole intermediate meets the highest standards for enantiomeric excess and chemical purity required by global regulatory agencies.
We invite you to leverage our technical capabilities to accelerate your project timelines. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. Whether you need specific COA data for a particular analog or a comprehensive route feasibility assessment for a new target molecule, our experts are prepared to provide the data-driven insights you need to make informed sourcing decisions. Let us be your partner in transforming this innovative chemistry into a reliable supply of high-quality pharmaceutical intermediates.
