Advanced Synthesis of Axial Chiral Aryl Indoles for High-Value Pharmaceutical Applications
The pharmaceutical industry is constantly seeking efficient pathways to access complex chiral scaffolds that serve as critical building blocks for next-generation therapeutics. Patent CN110467555B introduces a groundbreaking methodology for the synthesis of axial chiral aryl indole compounds, specifically targeting the indole-naphthalene and indole-benzene structural motifs. These privileged structures have garnered significant attention in medicinal chemistry due to their potent biological activities, ranging from glucocorticoid receptor antagonism to HCV NS5B polymerase inhibition. The disclosed technology addresses a critical gap in the current synthetic landscape by providing a robust, organocatalytic route that operates under exceptionally mild conditions. By leveraging a dynamic kinetic resolution strategy mediated by chiral phosphoric acids, this invention enables the direct construction of axially chiral skeletons from racemic precursors with high fidelity. For R&D directors and process chemists, this represents a paradigm shift away from traditional, resource-intensive coupling reactions towards a more streamlined, atom-economical approach that promises to accelerate lead optimization campaigns.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of axially chiral indole-naphthalene and indole-benzene frameworks has been fraught with synthetic challenges that hinder scalable production. Conventional strategies often rely on the direct coupling of indole rings with naphthalene or benzene derivatives, a process that typically suffers from poor regioselectivity and limited scope. Existing literature, such as reports in Angewandte Chemie and Nature Chemistry, describes methods involving the coupling of 2-indolecarbinols with phenols or azonaphthalenes; however, these approaches are often restricted to specific substrate classes and may require stringent reaction parameters. Furthermore, many traditional routes fail to effectively control the stereochemistry at the axial bond, resulting in racemic mixtures that necessitate costly and yield-loss-prone resolution steps. The reliance on transition metal catalysts in some legacy methods also introduces concerns regarding heavy metal contamination, which is a critical quality attribute for pharmaceutical intermediates. These limitations collectively create a bottleneck in the supply chain for high-purity chiral intermediates, driving up costs and extending development timelines for drug candidates targeting oncology and virology.
The Novel Approach
In stark contrast to these legacy techniques, the methodology outlined in CN110467555B utilizes a highly efficient organocatalytic system that transforms simple, commercially available starting materials into complex chiral architectures in a single operational step. The core innovation lies in the use of azodicarboxylates as electrophilic partners reacting with substituted indoles in the presence of a chiral phosphoric acid catalyst. This reaction proceeds smoothly in dichloromethane at ambient temperatures, typically between 20°C and 30°C, eliminating the need for energy-intensive heating or cryogenic cooling. The process demonstrates remarkable versatility, accommodating a wide array of substituents on both the indole and the aryl moieties, including electron-withdrawing groups like trifluoromethyl and halogens, as well as electron-donating groups like methoxy. As illustrated in the general reaction scheme below, this approach not only simplifies the synthetic sequence but also ensures high optical purity directly from the reaction vessel.
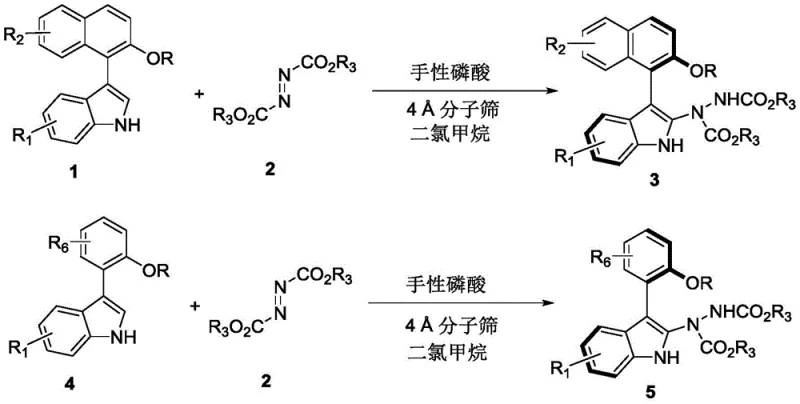
The operational simplicity of this novel approach cannot be overstated; the reaction mixture requires only standard filtration and concentration followed by routine silica gel chromatography to isolate the target compounds in high yields. This stands as a significant improvement over multi-step sequences that generate substantial chemical waste. For procurement managers, the ability to utilize economical raw materials such as substituted indoles and azodicarboxylates, combined with the elimination of expensive transition metals, translates directly into a more favorable cost structure for cost reduction in pharmaceutical intermediate manufacturing. The robustness of the reaction conditions further implies that this chemistry is readily transferable from laboratory scale to pilot plant operations without significant re-engineering of the process parameters.
Mechanistic Insights into Chiral Phosphoric Acid Catalyzed Asymmetric Addition
The success of this synthetic strategy hinges on the precise stereochemical control exerted by the chiral phosphoric acid catalyst. The patent identifies a specific class of binaphthyl-derived phosphoric acids as being particularly effective, with Compound 61 emerging as the optimal catalyst for this transformation. Structurally, Compound 61 features bulky 2,4,6-triisopropylphenyl groups at the 3,3'-positions of the binaphthyl backbone, which create a well-defined chiral pocket. This steric environment is crucial for differentiating between the enantiotopic faces of the reacting species. The mechanism likely involves a dual activation mode where the acidic proton of the phosphoric acid activates the azodicarboxylate via hydrogen bonding, while the phosphoryl oxygen simultaneously interacts with the indole substrate. This cooperative activation lowers the energy barrier for the nucleophilic attack while rigidly locking the transition state into a specific chiral conformation.
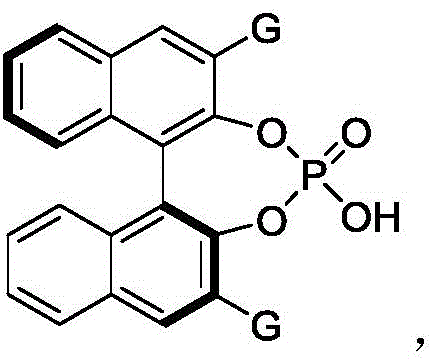
From an impurity control perspective, this organocatalytic mechanism offers distinct advantages over metal-catalyzed alternatives. Since the chirality is induced by the organic catalyst rather than a metal-ligand complex, there is no risk of metal-induced side reactions or racemization pathways that are sometimes observed with transition metals. The high enantiomeric ratios (er) reported, reaching up to 98:2 in optimized examples, indicate that the background non-catalyzed reaction is negligible. This high level of stereocontrol minimizes the formation of diastereomeric impurities, thereby simplifying the downstream purification process. For quality assurance teams, this means a cleaner crude profile and a higher probability of meeting stringent regulatory specifications for chiral purity without the need for repeated recrystallizations or preparative chiral HPLC, which are often cost-prohibitive at large scales.
How to Synthesize Axial Chiral Aryl Indoles Efficiently
Implementing this synthesis in a laboratory or pilot setting requires adherence to specific protocol details to ensure reproducibility and optimal outcomes. The general procedure involves dissolving the indole substrate and the azodicarboxylate reagent in dry dichloromethane, followed by the addition of activated 4 Å molecular sieves to maintain anhydrous conditions which are critical for catalyst performance. The chiral phosphoric acid catalyst is then introduced, typically at a loading of 10 mol%, although optimization may allow for lower loadings depending on the specific substrate reactivity. The reaction is allowed to proceed at room temperature with monitoring via thin-layer chromatography (TLC) to determine the endpoint. Once conversion is complete, the molecular sieves are removed by filtration, and the solvent is evaporated under reduced pressure. The resulting residue is purified using standard flash column chromatography with a petroleum ether and ethyl acetate gradient. For a detailed, step-by-step standardized operating procedure including specific molar ratios and workup instructions, please refer to the guide below.
- Prepare the reaction mixture by combining the indole substrate (Formula 1 or 4) and the azodicarboxylate reagent (Formula 2) in dichloromethane solvent.
- Add 4 Å molecular sieves as an additive and introduce the chiral phosphoric acid catalyst (preferably Compound 61) to the mixture.
- Stir the reaction at room temperature (20-30°C) until completion monitored by TLC, then filter, concentrate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement specialists, the adoption of this technology offers tangible strategic benefits beyond mere technical elegance. The shift towards organocatalysis fundamentally alters the cost drivers associated with producing complex chiral intermediates. By removing the dependency on precious metals such as palladium, rhodium, or iridium, manufacturers can insulate their supply chains from the volatility of commodity metal prices and the geopolitical risks associated with their sourcing. Furthermore, the mild reaction conditions reduce the energy footprint of the manufacturing process, aligning with increasingly strict environmental regulations and corporate sustainability goals. The simplicity of the workup procedure—essentially a filtration and evaporation—minimizes solvent consumption and waste generation, contributing to a greener manufacturing profile that is highly valued by downstream pharmaceutical partners.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and ligands results in a direct reduction in raw material costs. Additionally, the high yields and selectivity reduce the amount of starting material required per kilogram of product, enhancing overall process mass intensity. The avoidance of complex purification steps like chiral HPLC further drives down processing costs, making the final high-purity pharmaceutical intermediate more economically viable for large-scale production.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, including various substituted indoles and azodicarboxylates, are commercially available from multiple global suppliers, reducing the risk of single-source bottlenecks. The robustness of the reaction conditions means that the process is less sensitive to minor fluctuations in temperature or mixing, ensuring consistent batch-to-batch quality. This reliability is crucial for maintaining continuous supply lines for critical drug substances, minimizing the risk of production delays that could impact patient access.
- Scalability and Environmental Compliance: The use of dichloromethane as a solvent is well-established in the industry, and the reaction does not generate hazardous byproducts that require specialized disposal. The scalability of the process is supported by the fact that it does not require specialized high-pressure or low-temperature equipment, allowing for easy translation from gram-scale R&D to ton-scale commercial manufacturing. This ease of scale-up facilitates rapid response to market demand surges, ensuring that the commercial scale-up of complex pharmaceutical intermediates can be achieved with minimal capital expenditure.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific projects, we have compiled answers to common inquiries regarding the process parameters and capabilities. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation. Understanding these nuances is essential for integrating this synthetic route into existing development pipelines and for assessing its compatibility with current manufacturing infrastructure.
Q: What are the key advantages of this chiral phosphoric acid catalyzed method?
A: This method offers mild reaction conditions (25°C), high enantioselectivity (er values up to 98:2), and excellent yields (up to 98%), making it superior to traditional coupling methods which often require harsh conditions.
Q: Which catalyst is preferred for this synthesis?
A: The patent specifically highlights Compound 61, a binaphthyl-derived chiral phosphoric acid with 2,4,6-triisopropyl groups, as the most effective catalyst for achieving high optical purity.
Q: Do these compounds exhibit biological activity?
A: Yes, biological testing indicated that the synthesized axial chiral aryl indole compounds possess cytotoxic activity against MCF-7 breast cancer cells, with Compound 3aa showing an IC50 of 4.44 μg/mL.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axial Chiral Aryl Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic methodologies described in CN110467555B for advancing oncology and antiviral drug discovery. As a premier CDMO partner, we possess the technical expertise and infrastructure to translate this academic innovation into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from clinical trials to market launch. We are committed to delivering products that meet stringent purity specifications, utilizing our rigorous QC labs to verify enantiomeric excess and chemical purity at every stage of production. Our facility is equipped to handle the specific requirements of organocatalytic processes, including precise temperature control and advanced chromatographic purification capabilities.
We invite you to collaborate with us to leverage this cutting-edge chemistry for your next drug candidate. By partnering with us, you gain access to a Customized Cost-Saving Analysis that evaluates the economic benefits of switching to this organocatalytic route compared to your current supply chain. We encourage you to contact our technical procurement team today to request specific COA data for similar chiral indole derivatives and to discuss route feasibility assessments tailored to your unique molecular targets. Let us help you secure a reliable supply of high-quality intermediates that will accelerate your path to clinical success.
