Advanced Electrochemical Manufacturing of High-Purity 4-Selenopyrazole Derivatives for Pharma
The pharmaceutical and fine chemical industries are constantly seeking greener, more efficient synthetic routes that minimize environmental impact while maximizing product purity. A significant breakthrough in this domain is documented in Chinese Patent CN113046772A, which discloses a novel electrochemical synthesis method for 4-selenopyrazole derivatives. These compounds are critical scaffolds in medicinal chemistry, known for their diverse biological activities. Unlike conventional thermal methods that rely on harsh oxidants and toxic transition metals, this invention utilizes electricity as a clean, traceless reagent to drive a cyclization-coupling domino reaction. By employing an undivided cell setup with specific electrode materials and electrolytes, the process achieves high selectivity and excellent yields under remarkably mild conditions. This technological advancement represents a paradigm shift towards sustainable manufacturing, offering a reliable pharmaceutical intermediate supplier pathway that aligns with modern green chemistry principles and stringent regulatory requirements for drug substance production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-selenopyrazole derivatives has relied heavily on transition metal catalysis, typically involving copper salts such as cuprous bromide (CuBr) paired with ligands like bipyridine. As illustrated in prior art, these reactions often necessitate high temperatures, frequently around 100°C, and utilize molecular oxygen from air as the terminal oxidant in solvents like DMSO. 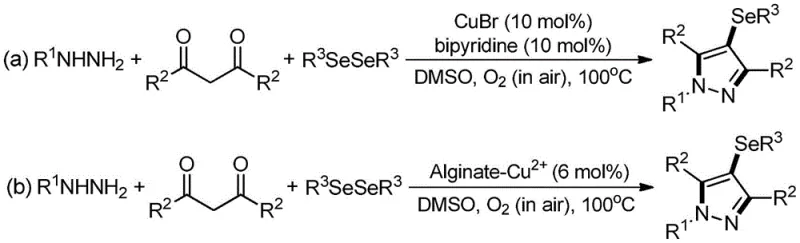 While effective in a laboratory setting, these thermal protocols present substantial hurdles for industrial application. The primary concern is the inevitable contamination of the final product with residual copper, a heavy metal that is strictly regulated in active pharmaceutical ingredients (APIs). Removing these traces requires additional, costly purification steps, such as specialized scavenging resins or repeated recrystallizations. Furthermore, the use of high temperatures and organic solvents increases energy consumption and poses safety risks related to flammability and thermal runaway, making the process less desirable for large-scale operations where safety and cost-efficiency are paramount.
While effective in a laboratory setting, these thermal protocols present substantial hurdles for industrial application. The primary concern is the inevitable contamination of the final product with residual copper, a heavy metal that is strictly regulated in active pharmaceutical ingredients (APIs). Removing these traces requires additional, costly purification steps, such as specialized scavenging resins or repeated recrystallizations. Furthermore, the use of high temperatures and organic solvents increases energy consumption and poses safety risks related to flammability and thermal runaway, making the process less desirable for large-scale operations where safety and cost-efficiency are paramount.
The Novel Approach
In stark contrast, the electrochemical method introduced in CN113046772A offers a transformative solution by replacing chemical oxidants and metal catalysts with electrical energy. The core innovation lies in the use of an undivided electrochemical cell where the anodic oxidation drives the formation of reactive selenium species directly from diselenides. 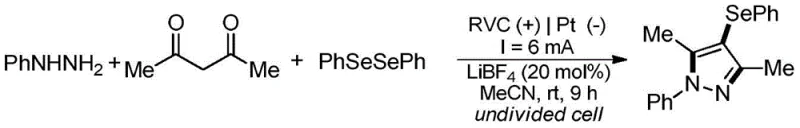 This approach operates at room temperature, drastically reducing energy costs and eliminating thermal safety hazards. The reaction system is optimized with lithium tetrafluoroborate (LiBF4) as the electrolyte and acetonitrile as the solvent, creating a highly conductive medium that facilitates efficient electron transfer. By avoiding transition metals entirely, the resulting 4-selenopyrazole products are inherently cleaner, significantly reducing the burden on downstream purification processes. This metal-free strategy not only enhances the environmental profile of the synthesis but also streamlines the supply chain by removing the dependency on expensive and fluctuating metal catalyst markets, thereby offering a more robust and predictable manufacturing route for high-value heterocyclic intermediates.
This approach operates at room temperature, drastically reducing energy costs and eliminating thermal safety hazards. The reaction system is optimized with lithium tetrafluoroborate (LiBF4) as the electrolyte and acetonitrile as the solvent, creating a highly conductive medium that facilitates efficient electron transfer. By avoiding transition metals entirely, the resulting 4-selenopyrazole products are inherently cleaner, significantly reducing the burden on downstream purification processes. This metal-free strategy not only enhances the environmental profile of the synthesis but also streamlines the supply chain by removing the dependency on expensive and fluctuating metal catalyst markets, thereby offering a more robust and predictable manufacturing route for high-value heterocyclic intermediates.
Mechanistic Insights into Electrochemical Cyclization-Coupling
Understanding the mechanistic underpinnings of this electrochemical transformation is crucial for R&D teams aiming to optimize the process for specific substrates. The reaction initiates at the anode surface, where the diselenide compound undergoes a single-electron oxidation to generate a radical cation intermediate. This unstable species rapidly dissociates into a phenylselenyl radical and a phenylselenyl cation. Simultaneously, the aryl hydrazine and 1,3-propanedione undergo a condensation reaction to form a pyrazole intermediate. The phenylselenyl radical then attacks this pyrazole core, forming a new carbon-selenium bond and generating a radical intermediate on the pyrazole ring. 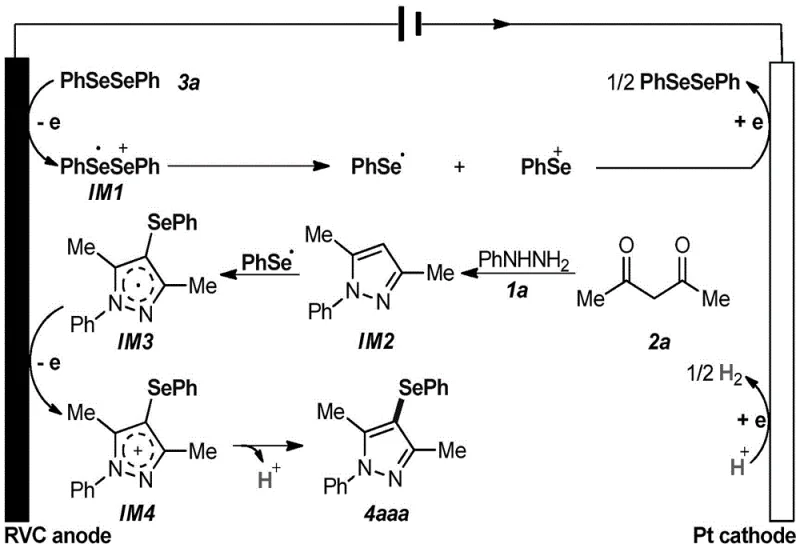 This radical intermediate is subsequently oxidized at the anode to a cationic species, which finally loses a proton to aromatize and yield the stable 4-selenopyrazole product. On the cathode side, protons generated during the reaction are reduced to hydrogen gas, completing the electrical circuit. This elegant mechanism highlights the dual role of the electrodes: the anode acts as the oxidant to generate reactive species and drive the coupling, while the cathode manages the proton balance. The precise control over electron flow allows for high chemoselectivity, minimizing side reactions that are common in thermal radical processes, thus ensuring a clean impurity profile essential for pharmaceutical applications.
This radical intermediate is subsequently oxidized at the anode to a cationic species, which finally loses a proton to aromatize and yield the stable 4-selenopyrazole product. On the cathode side, protons generated during the reaction are reduced to hydrogen gas, completing the electrical circuit. This elegant mechanism highlights the dual role of the electrodes: the anode acts as the oxidant to generate reactive species and drive the coupling, while the cathode manages the proton balance. The precise control over electron flow allows for high chemoselectivity, minimizing side reactions that are common in thermal radical processes, thus ensuring a clean impurity profile essential for pharmaceutical applications.
From an impurity control perspective, the absence of transition metals fundamentally alters the impurity landscape. In traditional copper-catalyzed routes, metal-ligand complexes and metal-bound byproducts are common contaminants that are difficult to separate due to their similar polarity to the product. In this electrochemical protocol, the primary byproducts are derived from over-oxidation or incomplete conversion, which are generally easier to manage through standard crystallization or extraction techniques. The use of LiBF4 as a supporting electrolyte is particularly advantageous; unlike halide salts which might participate in side reactions or corrode electrodes, tetrafluoroborate is electrochemically stable in acetonitrile and does not introduce nucleophilic impurities. Furthermore, the mild room temperature conditions prevent thermal degradation of sensitive functional groups on the aryl hydrazine or diselenide substrates, preserving the structural integrity of complex molecules. This robustness allows for a broader substrate scope, accommodating electron-withdrawing and electron-donating groups alike, which is vital for generating diverse libraries of analogs during drug discovery phases.
How to Synthesize 4-Selenopyrazole Derivatives Efficiently
To implement this cutting-edge synthesis in a laboratory or pilot plant setting, operators must adhere to specific parameters outlined in the patent to ensure reproducibility and high yield. The process begins with the preparation of the electrolytic solution, where precise molar ratios of the aryl hydrazine, 1,3-propanedione, and diselenide are dissolved in dry acetonitrile. The addition of the electrolyte salt, specifically LiBF4 at approximately 20 mol%, is critical for maintaining sufficient conductivity without overwhelming the system with inorganic salts that could complicate workup. The choice of electrode material is another pivotal factor; the patent specifies a reticulated vitreous carbon (RVC) mesh as the anode and a platinum sheet as the cathode. This combination provides a high surface area for the reaction while maintaining chemical inertness. Once the cell is assembled, a constant direct current of 6 mA is applied. Monitoring the reaction progress via TLC is recommended, with typical completion times around 9 hours. Upon completion, the solvent is removed under reduced pressure, and the crude product can often be purified simply by column chromatography or recrystallization, bypassing the need for metal scavenging.
- Prepare the electrolytic solution by dissolving aryl hydrazine, 1,3-propanedione, and diselenide compounds in acetonitrile with LiBF4 as the electrolyte salt.
- Insert a reticulated vitreous carbon (RVC) anode and a platinum sheet cathode into the undivided cell containing the reaction mixture.
- Apply a constant direct current of 6 mA at room temperature for approximately 9 hours, then isolate the product via solvent removal and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this electrochemical methodology presents compelling economic and logistical advantages over traditional thermal synthesis. The elimination of transition metal catalysts removes a significant cost center associated with purchasing expensive metal salts and ligands, as well as the downstream costs of metal removal and waste disposal. Since the reaction proceeds at room temperature, the energy footprint is drastically reduced compared to processes requiring heating to 100°C, leading to lower utility costs per kilogram of product. Moreover, the simplicity of the reaction setup—an undivided cell with commercially available electrodes—means that existing electrochemical infrastructure can be utilized or easily retrofitted, minimizing capital expenditure for scale-up. The high atom efficiency and selectivity of the process reduce the generation of hazardous waste, aligning with increasingly strict environmental regulations and reducing the costs associated with effluent treatment. These factors collectively contribute to a more resilient and cost-effective supply chain for critical pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The most significant driver for cost reduction is the complete removal of transition metal catalysts such as copper bromide and bipyridine ligands. In traditional manufacturing, the cost of these reagents, combined with the expensive unit operations required to remove trace metals to ppm levels, adds substantial overhead to the cost of goods sold (COGS). By utilizing electrons as the oxidant, this electrochemical process eliminates these material costs entirely. Additionally, the operation at ambient temperature reduces energy consumption significantly, as there is no need for prolonged heating or refluxing of solvents. The simplified workup procedure, which often avoids complex chromatographic purification in favor of crystallization, further lowers labor and material costs associated with isolation. These cumulative savings allow for a more competitive pricing structure for the final 4-selenopyrazole intermediates, enhancing margin potential for downstream API manufacturers.
- Enhanced Supply Chain Reliability: Supply chain stability is often threatened by the volatility of raw material markets, particularly for specialty catalysts and ligands. This electrochemical method relies on commodity chemicals—aryl hydrazines, 1,3-diketones, and diselenides—which are widely available from multiple global suppliers, reducing the risk of single-source bottlenecks. The electrolyte, LiBF4, is a standard salt in the battery and electrochemical industries, ensuring a secure and stable supply. Furthermore, the robustness of the reaction conditions means that minor variations in raw material quality are less likely to cause batch failures compared to sensitive metal-catalyzed systems. This reliability ensures consistent delivery schedules and reduces the need for excessive safety stock, allowing procurement teams to operate with leaner inventories and improved cash flow while maintaining continuous production lines for key intermediates.
- Scalability and Environmental Compliance: Scaling electrochemical reactions has historically been challenging, but the use of an undivided cell with simple electrode geometries in this patent simplifies the engineering requirements for commercial production. The process does not require high-pressure vessels or specialized gas handling systems for oxidants like oxygen, which simplifies reactor design and safety protocols. From an environmental perspective, the absence of heavy metals and stoichiometric chemical oxidants significantly reduces the toxicity of the waste stream. This facilitates easier compliance with environmental discharge regulations and lowers the costs associated with hazardous waste treatment. The generation of hydrogen gas at the cathode is the only gaseous byproduct, which can be safely vented or captured. These attributes make the process highly attractive for large-scale manufacturing facilities aiming to reduce their carbon footprint and meet sustainability goals without compromising on production volume or product quality.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the electrochemical synthesis of 4-selenopyrazole derivatives, based on the detailed experimental data provided in the patent literature. These insights are designed to clarify the operational feasibility and strategic benefits of adopting this technology for industrial applications. Understanding these nuances helps stakeholders make informed decisions about integrating electrochemical methods into their existing manufacturing portfolios.
Q: Why is the electrochemical method superior to traditional copper-catalyzed synthesis for pharmaceutical intermediates?
A: Traditional methods utilize transition metal catalysts like CuBr, which pose significant risks of heavy metal residue in the final API. The electrochemical approach described in CN113046772A eliminates transition metals entirely, using electrons as a traceless oxidant, thereby simplifying purification and ensuring compliance with strict pharmaceutical impurity standards.
Q: What are the optimal reaction conditions for maximizing yield in this electrochemical process?
A: The patent identifies lithium tetrafluoroborate (LiBF4) as the optimal electrolyte salt, used at 20 mol% relative to the substrate. Acetonitrile serves as the preferred solvent, and the reaction proceeds best at room temperature with a constant current of 6 mA using an RVC anode and Pt cathode pair, achieving yields up to 96%.
Q: Is this electrochemical synthesis method suitable for large-scale industrial production?
A: Yes, the method is highly scalable. It operates under mild conditions (room temperature, atmospheric pressure) in an undivided cell, avoiding the need for complex high-pressure equipment or hazardous chemical oxidants. The simple workup procedure, which often does not require column chromatography, further supports cost-effective commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Selenopyrazole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in modern pharmaceutical manufacturing. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN113046772A are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with advanced electrochemical reactors and rigorous QC labs capable of meeting stringent purity specifications required by global regulatory bodies. We understand that the transition to metal-free synthesis is not just a technical upgrade but a strategic move towards sustainability and cost-efficiency. Our team of expert chemists and engineers is dedicated to optimizing these electrochemical parameters to maximize yield and minimize impurities, delivering high-quality 4-selenopyrazole derivatives that meet your exacting standards.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Whether you require custom synthesis of novel analogs or large-scale production of established intermediates, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific needs. We encourage you to reach out to request specific COA data and route feasibility assessments to see how our electrochemical capabilities can enhance your supply chain resilience and reduce your overall manufacturing costs. Partner with NINGBO INNO PHARMCHEM to secure a reliable, high-purity supply of 4-selenopyrazole derivatives that drives your drug development forward.
