Advanced Electrochemical Manufacturing of High-Purity 4-Selenopyrazole Derivatives for Global Pharma
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and environmentally benign synthetic routes for complex heterocyclic intermediates. A significant breakthrough in this domain is documented in Chinese Patent CN113046772B, which discloses a novel electrochemical synthesis method for 4-selenopyrazole derivatives. This technology represents a paradigm shift from traditional transition metal-catalyzed processes to a sustainable electroorganic approach. By utilizing electrons as a trace-free oxidant, this method circumvents the inherent limitations of chemical oxidants and metal catalysts, offering a pathway to high-purity intermediates essential for drug discovery and development. The process operates under mild conditions, specifically at room temperature, and utilizes a simple undivided cell setup, making it highly attractive for both laboratory-scale optimization and potential industrial scale-up. For procurement and supply chain leaders, this innovation signals a move towards more reliable pharmaceutical intermediates supplier capabilities, where cost reduction in manufacturing is driven by simplified downstream processing and enhanced safety profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-selenopyrazole derivatives has relied heavily on transition metal catalysis, typically employing copper salts such as cuprous bromide (CuBr) alongside ligands like bipyridine. As illustrated in prior art, these reactions often require harsh conditions, including elevated temperatures around 100°C and the use of dimethyl sulfoxide (DMSO) as a solvent with atmospheric oxygen serving as the oxidant. 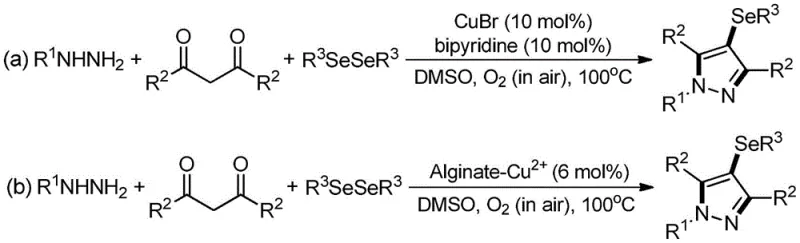 . While effective in generating the target scaffold, these conventional methods introduce significant challenges for large-scale pharmaceutical manufacturing. The primary concern is the inevitable contamination of the final product with transition metal residues, which necessitates rigorous and expensive purification steps to meet regulatory limits for elemental impurities. Furthermore, the use of high temperatures and organic solvents like DMSO poses safety risks related to flammability and thermal stability, while the reliance on stoichiometric or excess chemical oxidants generates substantial chemical waste, complicating environmental compliance and waste treatment protocols.
. While effective in generating the target scaffold, these conventional methods introduce significant challenges for large-scale pharmaceutical manufacturing. The primary concern is the inevitable contamination of the final product with transition metal residues, which necessitates rigorous and expensive purification steps to meet regulatory limits for elemental impurities. Furthermore, the use of high temperatures and organic solvents like DMSO poses safety risks related to flammability and thermal stability, while the reliance on stoichiometric or excess chemical oxidants generates substantial chemical waste, complicating environmental compliance and waste treatment protocols.
The Novel Approach
In stark contrast, the electrochemical methodology described in the patent offers a streamlined, metal-free alternative that addresses these critical pain points. The new process utilizes a solution containing aryl hydrazine, 1,3-propanedione, and diselenide compounds as the electrolyte, subjected to direct current in an undivided cell. 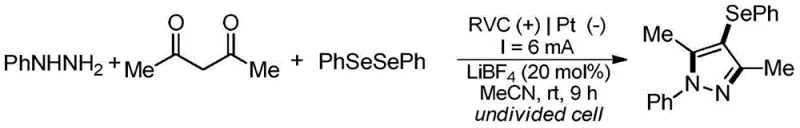 . This approach eliminates the need for external chemical oxidants and transition metal catalysts entirely, relying instead on the anodic oxidation of the diselenide species to drive the cyclization-coupling domino reaction. The reaction proceeds efficiently at room temperature in acetonitrile, a solvent that is easier to recover and recycle compared to DMSO. By removing the metal catalyst from the equation, the resulting 4-selenopyrazole products are inherently cleaner, significantly reducing the burden on downstream purification units. This technological leap not only enhances the atom economy of the reaction but also aligns perfectly with the principles of green chemistry, providing a cost reduction in pharmaceutical intermediates manufacturing through reduced reagent costs and simplified isolation procedures.
. This approach eliminates the need for external chemical oxidants and transition metal catalysts entirely, relying instead on the anodic oxidation of the diselenide species to drive the cyclization-coupling domino reaction. The reaction proceeds efficiently at room temperature in acetonitrile, a solvent that is easier to recover and recycle compared to DMSO. By removing the metal catalyst from the equation, the resulting 4-selenopyrazole products are inherently cleaner, significantly reducing the burden on downstream purification units. This technological leap not only enhances the atom economy of the reaction but also aligns perfectly with the principles of green chemistry, providing a cost reduction in pharmaceutical intermediates manufacturing through reduced reagent costs and simplified isolation procedures.
Mechanistic Insights into Electrochemical Cyclization-Coupling
Understanding the mechanistic underpinnings of this electrochemical transformation is crucial for R&D directors evaluating its feasibility for complex molecule synthesis. The reaction initiates at the anode surface, where the diselenide compound (e.g., diphenyl diselenide) undergoes a single-electron oxidation to form a radical cation intermediate (IM1). This unstable species rapidly dissociates into a phenylselenyl cation and a phenylselenyl radical. Concurrently, the aryl hydrazine and 1,3-propanedione undergo a condensation and cyclization sequence to form a pyrazole intermediate (IM2). The phenylselenyl radical then attacks this pyrazole core to generate a radical intermediate (IM3), which is subsequently oxidized at the anode to a cationic species (IM4). 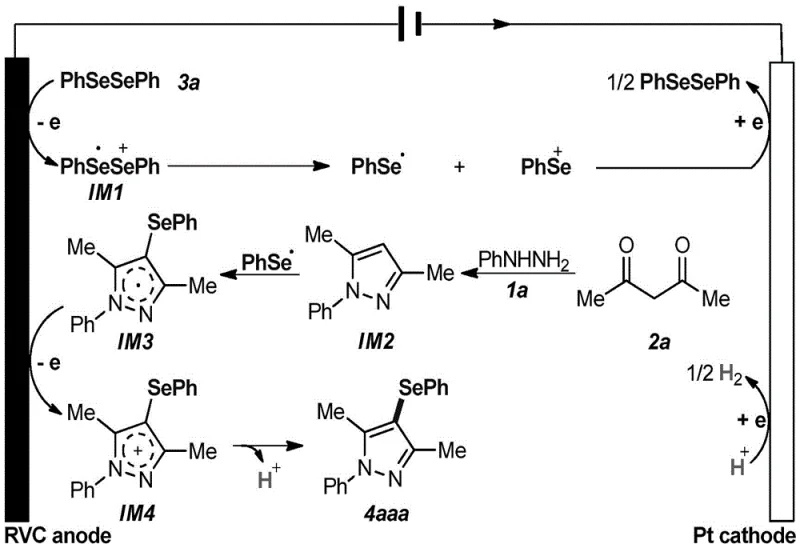 . Finally, deprotonation of IM4 yields the target 4-selenopyrazole product. At the cathode, protons are reduced to generate hydrogen gas, completing the electrical circuit. This elegant mechanism ensures high selectivity because the electrochemical potential can be finely tuned to activate specific bonds without affecting sensitive functional groups elsewhere in the molecule. The absence of aggressive chemical oxidants means that functionalities such as halogens, ethers, and even nitro groups remain intact, as evidenced by the broad substrate scope demonstrated in the patent examples.
. Finally, deprotonation of IM4 yields the target 4-selenopyrazole product. At the cathode, protons are reduced to generate hydrogen gas, completing the electrical circuit. This elegant mechanism ensures high selectivity because the electrochemical potential can be finely tuned to activate specific bonds without affecting sensitive functional groups elsewhere in the molecule. The absence of aggressive chemical oxidants means that functionalities such as halogens, ethers, and even nitro groups remain intact, as evidenced by the broad substrate scope demonstrated in the patent examples.
From an impurity control perspective, this mechanism offers distinct advantages over radical chain reactions initiated by thermal decomposers. Since the generation of reactive selenium species is controlled by the applied current density rather than thermal kinetics, the formation of side products derived from uncontrolled radical propagation is minimized. The patent data indicates that optimizing the current density between 4 mA and 12 mA is critical; currents that are too low fail to drive the reaction, while excessive currents may lead to over-oxidation or solvent decomposition. The use of lithium tetrafluoroborate (LiBF4) as the supporting electrolyte further stabilizes the ionic environment, ensuring consistent conductivity and reaction efficiency. This level of control allows for the production of high-purity OLED material precursors or pharmaceutical intermediates with minimal byproduct formation, reducing the need for extensive chromatographic purification and facilitating a more robust manufacturing process.
How to Synthesize 4-Selenopyrazole Derivatives Efficiently
To implement this electrochemical synthesis effectively, precise control over reaction parameters is essential to replicate the high yields reported in the patent literature. The standard protocol involves charging an undivided cell with the three key components: the aryl hydrazine, the 1,3-dicarbonyl compound, and the diselenide, dissolved in acetonitrile with 20 mol% LiBF4. The choice of electrode materials is paramount; the patent specifies that a reticulated vitreous carbon (RVC) anode paired with a platinum (Pt) sheet cathode provides the optimal surface area and electrochemical stability for this transformation. The reaction is typically conducted under a constant current of 6 mA at ambient temperature for approximately 9 hours. Monitoring the reaction progress via thin-layer chromatography (TLC) is recommended to determine the exact endpoint, although the patent suggests that 9 hours is generally sufficient for complete conversion. Upon completion, the solvent is removed under reduced pressure, and the crude product can be purified using standard column chromatography techniques.
- Prepare the electrolyte solution by dissolving aryl hydrazine, 1,3-propanedione, and diselenide compounds in acetonitrile with LiBF4 as the supporting electrolyte.
- Insert a reticulated vitreous carbon (RVC) anode and a platinum sheet cathode into the undivided cell containing the reaction mixture.
- Apply a constant direct current of 6 mA at room temperature for approximately 9 hours, then isolate the product via solvent removal and chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this electrochemical technology translates into tangible strategic benefits that extend beyond mere chemical yield. The elimination of transition metal catalysts removes a major bottleneck in the supply chain associated with the sourcing of high-purity metal salts and the subsequent disposal of metal-contaminated waste streams. This shift significantly simplifies the regulatory compliance landscape, particularly for companies supplying active pharmaceutical ingredients (APIs) where heavy metal limits are strictly enforced. By avoiding copper or other transition metals, manufacturers can drastically simplify their quality control workflows, reducing the time and cost associated with ICP-MS testing and metal scavenging treatments. Furthermore, the ability to run the reaction at room temperature reduces the energy footprint of the manufacturing process, contributing to lower operational expenditures and a smaller carbon footprint, which is increasingly important for corporate sustainability goals.
- Cost Reduction in Manufacturing: The economic impact of switching to this metal-free electrochemical route is profound, primarily driven by the simplification of the downstream processing workflow. In traditional methods, the removal of residual copper often requires specialized scavenger resins or multiple recrystallization steps, both of which add material costs and reduce overall throughput. By eliminating the metal catalyst entirely, this new method removes the need for these expensive purification stages, leading to substantial cost savings in the final product price. Additionally, the use of electricity as the primary "reagent" for oxidation is inherently cheaper and more atom-efficient than purchasing stoichiometric amounts of chemical oxidants, further driving down the variable costs of production. The high atom efficiency of the domino reaction also means that less raw material is wasted as byproducts, maximizing the value extracted from every kilogram of starting material purchased.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commercially available and stable starting materials that do not require special handling or storage conditions associated with sensitive metal catalysts or strong chemical oxidants. The reactants—aryl hydrazines, 1,3-propanediones, and diselenides—are widely available from global chemical suppliers, reducing the risk of single-source dependency. Moreover, the mild reaction conditions (room temperature, atmospheric pressure) reduce the risk of unplanned shutdowns due to equipment failure or safety incidents, ensuring a more consistent and reliable delivery schedule for customers. The robustness of the electrochemical cell setup also allows for easier scaling from pilot plants to commercial production without the need for complex high-pressure or high-temperature reactors, facilitating a smoother technology transfer process.
- Scalability and Environmental Compliance: Scaling electrochemical reactions is becoming increasingly feasible with modern flow chemistry and stackable cell technologies, making this method highly suitable for commercial scale-up of complex pharmaceutical intermediates. The environmental benefits are equally significant; the process generates hydrogen gas as the only byproduct at the cathode, which can be safely vented or captured, avoiding the generation of toxic heavy metal sludge. This aligns with strict environmental regulations regarding waste discharge and hazardous material handling, reducing the liability and disposal costs for manufacturing facilities. The use of acetonitrile, a solvent with well-established recovery protocols, further enhances the environmental profile of the process, making it a preferred choice for companies aiming to achieve green chemistry certifications and reduce their overall ecological impact.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrochemical synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on reaction scope, optimization, and practical application. Understanding these details is vital for technical teams evaluating the feasibility of integrating this method into existing production lines or R&D pipelines.
Q: Why is the electrochemical method superior to copper-catalyzed synthesis for pharmaceutical intermediates?
A: The electrochemical method eliminates the need for transition metal catalysts like copper bromide, thereby avoiding costly and complex metal residue removal steps which are critical for meeting stringent pharmaceutical purity standards.
Q: What are the optimal reaction conditions for maximizing yield in this electrochemical process?
A: Optimal conditions involve using acetonitrile as the solvent, LiBF4 as the electrolyte salt at 20 mol%, and a constant current of 6 mA with an RVC anode and Pt cathode at room temperature, achieving yields up to 96%.
Q: Does this synthesis method support diverse functional groups on the substrate?
A: Yes, the method demonstrates excellent functional group tolerance, successfully accommodating substituents such as halogens (F, Cl, Br), trifluoromethyl, methoxy, and nitro groups on the aryl rings without significant yield loss.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Selenopyrazole Derivatives Supplier
As the demand for high-quality heterocyclic intermediates continues to grow in the pharmaceutical and agrochemical sectors, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM ensures access to cutting-edge synthetic technologies. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our facilities are equipped with state-of-the-art electrochemical reactors and stringent purity specifications, supported by rigorous QC labs that guarantee every batch meets the highest international standards. We understand the critical nature of timeline and quality in drug development, and our team is dedicated to providing seamless technology transfer and process optimization services tailored to your specific molecular targets.
We invite you to engage with our technical procurement team to discuss how this innovative electrochemical route can be adapted for your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this metal-free process for your supply chain. We encourage potential partners to contact us directly to obtain specific COA data for similar intermediates and to receive detailed route feasibility assessments. Let us help you optimize your manufacturing strategy with safer, greener, and more cost-effective solutions for the production of complex 4-selenopyrazole derivatives.
