Advanced One-Pot Synthesis of 2-Cyano-3-Arylpyrroles for Commercial Scale-Up
Advanced One-Pot Synthesis of 2-Cyano-3-Arylpyrroles for Commercial Scale-Up
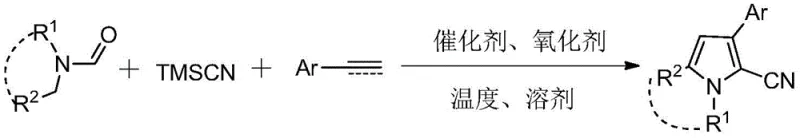
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct nitrogen-containing heterocycles, which serve as critical scaffolds in drug discovery and material science. Patent CN107501156B introduces a groundbreaking three-component tandem synthesis method for producing 2-cyano-3-arylpyrrole compounds, addressing long-standing inefficiencies in heterocyclic chemistry. This innovation leverages a Lewis acid-catalyzed oxidative cyclization strategy that merges N,N-disubstituted formamides, trimethylsilyl cyanide (TMSCN), and aryl alkenes or alkynes into a unified reaction vessel. By eliminating the need for intermediate isolation and utilizing readily available commodity chemicals, this technology represents a significant leap forward for reliable pharmaceutical intermediate supplier networks aiming to optimize their production pipelines. The method not only streamlines the synthetic route but also enhances the purity profile of the final polysubstituted pyrrole products, making it an attractive candidate for high-value API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of substituted pyrrole rings has been plagued by cumbersome multi-step protocols that suffer from poor atom economy and hazardous reagent profiles. Prior art, such as the methods cited in Tetrahedron Letters (1995), often relies on the use of toxic and difficult-to-handle reagents like boron trifluoride etherate or ferric chloride nonahydrate, which generate substantial corrosive waste streams. Furthermore, traditional routes frequently involve the formation of unstable intermediates, such as propargylamino-acetonitrile derivatives, which require rigorous purification before proceeding to the next step. This stepwise approach inherently increases the risk of product loss, extends the overall lead time for high-purity pharmaceutical intermediates, and escalates the operational costs due to the need for multiple reactor setups and extensive solvent consumption. The accumulation of byproducts in these older methodologies often complicates downstream processing, necessitating expensive chromatographic separations that are impractical for large-scale commercial operations.
The Novel Approach
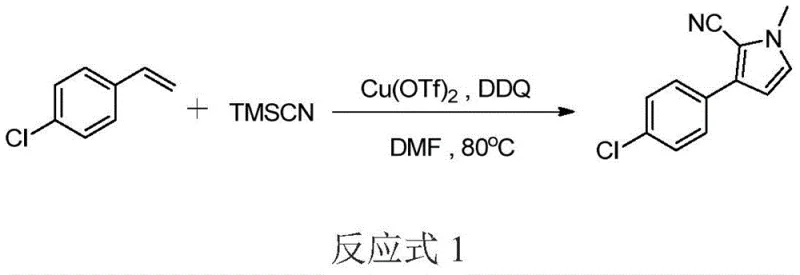
In stark contrast, the methodology disclosed in CN107501156B utilizes a convergent one-pot strategy that dramatically simplifies the synthetic landscape for 2-cyano-3-arylpyrroles. By employing a copper-catalyzed system in the presence of DDQ as an oxidant, the reaction seamlessly integrates C-C and C-N bond formation events without the need to isolate reactive intermediates. This tandem process allows for the direct conversion of simple starting materials—such as styrenes or phenylacetylenes and formamides—into complex heterocyclic architectures with high efficiency. The ability to conduct this transformation in common solvents like DMF or mixed hydrocarbon systems reduces the environmental footprint and simplifies solvent recovery protocols. Moreover, the mild reaction conditions, typically maintained around 80°C, minimize thermal degradation of sensitive functional groups, thereby ensuring a cleaner impurity profile compared to harsher classical methods.
Mechanistic Insights into Cu(OTf)2-Catalyzed Oxidative Cyclization
The core of this technological advancement lies in the synergistic interaction between the Lewis acid catalyst and the oxidant, which facilitates a complex cascade of bond-forming events. The copper trifluoromethanesulfonate acts as a potent Lewis acid, activating the nitrile source (TMSCN) and coordinating with the unsaturated bonds of the aryl alkene or alkyne substrate to initiate nucleophilic attack. Simultaneously, the oxidant, preferably DDQ, plays a critical role in the dehydrogenative aromatization step, driving the equilibrium towards the formation of the stable pyrrole ring system. This mechanistic pathway avoids the generation of stoichiometric metal waste often associated with traditional transition metal couplings, offering a greener alternative for cost reduction in electronic chemical manufacturing and pharmaceutical synthesis. The precise control over the oxidation state prevents over-oxidation of the electron-rich pyrrole core, a common pitfall in heterocyclic synthesis that leads to polymeric tars and reduced yields.
From an impurity control perspective, the one-pot nature of this reaction is particularly advantageous for maintaining high product quality. Because the intermediate species are generated and consumed in situ within the same reaction matrix, there is minimal opportunity for side reactions that typically occur during the handling and storage of isolated intermediates. The use of batched addition for the oxidant further refines the reaction kinetics, preventing localized exotherms that could trigger decomposition pathways. This controlled environment ensures that the final crude product contains fewer structurally related impurities, simplifying the final purification step. For R&D teams focused on regulatory compliance, this inherent process robustness translates to a more consistent impurity profile across different batches, facilitating easier validation and scale-up activities.
How to Synthesize 2-Cyano-3-Arylpyrrole Efficiently
Implementing this synthesis requires careful attention to the sequential addition of reagents and the controlled dosing of the oxidant to maximize yield and safety. The protocol generally begins with the dissolution of the aryl alkene or alkyne substrate in the chosen solvent system, followed by the introduction of the formamide and TMSCN. The catalyst is then added to initiate the coordination complex, after which the reaction mixture is heated to the optimal temperature range of 40-120°C. To maintain steady reaction progress and avoid runaway exotherms, the oxidant is introduced in multiple small aliquots over an extended period, typically ranging from 10 to 30 hours depending on the substrate reactivity. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by sequentially adding N,N-disubstituted formamide, trimethylsilyl cyanide (TMSCN), Lewis acid catalyst (Cu(OTf)2), and aryl alkene or alkyne into the solvent.
- Heat the mixture to 80°C and begin the batched addition of the oxidant (DDQ) over a period of 8 to 24 hours to control the oxidation rate.
- Upon completion, extract the organic phase, dry over anhydrous sodium sulfate, and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this three-component tandem method offers tangible strategic benefits that extend beyond mere chemical elegance. The primary value driver is the drastic simplification of the manufacturing workflow, which directly correlates to reduced operational expenditures and enhanced throughput. By collapsing multiple synthetic steps into a single vessel operation, manufacturers can significantly lower their capital expenditure requirements for reactor hardware and reduce the labor hours needed for batch turnover. This efficiency gain is critical in a competitive market where speed to market for new drug candidates is paramount. Furthermore, the reliance on bulk commodity chemicals like DMF and TMSCN ensures a stable and resilient supply chain, mitigating the risks associated with sourcing exotic or regulated reagents that often plague specialty chemical production.
- Cost Reduction in Manufacturing: The elimination of intermediate isolation and purification steps represents a massive opportunity for cost savings in the production of complex heterocycles. Traditional multi-step syntheses incur significant costs related to solvent usage, filtration media, and energy consumption for drying and concentrating intermediate fractions. By performing the entire transformation in a one-pot fashion, the process effectively cuts out these unit operations, leading to a leaner manufacturing bill of materials. Additionally, the high atom economy of the tandem reaction minimizes waste generation, which lowers the costs associated with waste disposal and environmental compliance. This streamlined approach allows for a more competitive pricing structure for the final active pharmaceutical ingredients derived from these pyrrole scaffolds.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route contributes to greater supply chain stability by reducing the number of potential failure points in the production process. In multi-step syntheses, a yield drop or quality issue at any intermediate stage can halt the entire production line, causing delays and inventory shortages. The one-pot nature of this method consolidates risk, as the reaction proceeds directly to the final product with fewer opportunities for human error or equipment malfunction between steps. Moreover, the use of stable, shelf-stable reagents like copper triflate and DDQ ensures that raw material quality remains consistent over time, further securing the continuity of supply for downstream customers who rely on just-in-time delivery models.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to the plant floor often reveals hidden challenges, but this methodology is inherently designed for scalability. The reaction conditions are mild and do not require extreme pressures or cryogenic temperatures, making it compatible with standard stainless steel reactor infrastructure found in most multipurpose chemical plants. The solvent systems employed, such as DMF or heptane/DMAC mixtures, are well-understood in terms of safety and recovery, facilitating straightforward implementation of green chemistry principles. This ease of scale-up ensures that commercial volumes can be reached rapidly without the need for extensive process re-engineering, while simultaneously meeting increasingly stringent global environmental regulations regarding solvent emissions and heavy metal residues.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear understanding of the method's capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers below highlight the versatility of the substrate scope and the practical considerations for optimizing reaction outcomes.
Q: What are the primary advantages of this three-component tandem method over traditional pyrrole synthesis?
A: Unlike traditional methods that require complex multi-step sequences and harsh reagents like boron trifluoride, this patent describes a one-pot process that eliminates intermediate isolation, significantly reducing labor and capital investment while improving overall yield.
Q: Which catalysts and oxidants are preferred for this transformation?
A: The patent identifies copper trifluoromethanesulfonate (Cu(OTf)2) as the preferred Lewis acid catalyst and 2,3-dichloro-5,6-dicyano-1,4-benzoquinone (DDQ) as the optimal oxidant for achieving high conversion rates under mild heating conditions.
Q: Is this process scalable for industrial production of pharmaceutical intermediates?
A: Yes, the use of common solvents like DMF or n-heptane/DMAC mixtures, combined with commercially available starting materials and a simplified workup procedure, makes this method highly suitable for commercial scale-up and continuous manufacturing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Cyano-3-Arylpyrrole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the one described in CN107501156B for accelerating drug development pipelines. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of critical intermediates. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 2-cyano-3-arylpyrrole meets the highest industry standards. We are committed to leveraging our technical expertise to adapt this efficient one-pot process for your specific project needs, delivering high-quality materials that support your R&D and commercial goals.
We invite you to collaborate with us to explore how this innovative synthesis can optimize your supply chain and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and quality targets. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to excellence can drive value for your organization.
