Advanced Catalytic Synthesis of 3-Trifluoromethyl Triazoles: Scaling Pharmaceutical Intermediates from Lab to Commercial Production
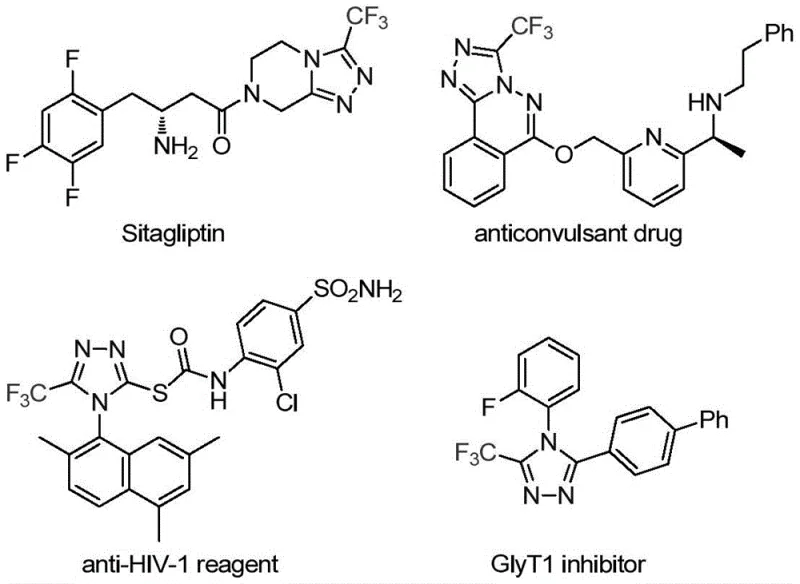
The patent CN113307778A introduces a groundbreaking methodology for synthesizing 3-trifluoromethyl substituted 1,2,4-triazole compounds, representing a significant advancement in heterocyclic chemistry for pharmaceutical applications. This innovation addresses critical limitations in traditional synthetic routes by employing a dual molybdenum-copper catalytic system that operates under remarkably mild conditions (70–90°C), thereby enhancing both reaction efficiency and product purity. The process utilizes readily accessible starting materials including trifluoroethylimidoyl chloride and functionalized isonitriles, which are commercially available or easily synthesized from common precursors like triphenylphosphine and carbon tetrachloride. Crucially, the methodology demonstrates exceptional scalability from laboratory-scale reactions to commercial production volumes while maintaining high regioselectivity and yield consistency across diverse substrate variations. This patent establishes a robust foundation for manufacturing high-value triazole intermediates that serve as essential building blocks in numerous therapeutic agents, including antidiabetic drugs and antiviral compounds. The significance of this development extends beyond mere synthetic convenience; it directly impacts the pharmaceutical industry's ability to produce complex molecules with improved metabolic stability and bioavailability profiles required for modern drug development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic approaches for trifluoromethyl-substituted triazoles typically rely on high-energy cyclization reactions between trifluoroacetyl hydrazine and amidine compounds or hydrazinolysis of trifluoromethyl oxazolinones, both requiring elevated temperatures exceeding 150°C that promote unwanted side reactions and decomposition pathways. These methods frequently generate complex impurity profiles necessitating extensive purification steps that significantly reduce overall yield and increase production costs while introducing batch-to-batch variability that compromises quality control in pharmaceutical manufacturing. Furthermore, conventional copper-catalyzed multi-component reactions using diazonium salts suffer from poor functional group tolerance and require stringent anhydrous conditions that complicate large-scale implementation due to moisture sensitivity issues. The limited substrate scope of existing methodologies restricts structural diversity in the final products, hindering medicinal chemistry optimization efforts for drug discovery programs. Most critically, these processes lack scalability to commercial production volumes without substantial re-engineering, creating significant supply chain vulnerabilities for pharmaceutical manufacturers dependent on consistent intermediate availability.
The Novel Approach
The patented methodology overcomes these limitations through a carefully designed molybdenum-copper dual catalytic system that operates at significantly milder temperatures (70–90°C), thereby minimizing thermal degradation pathways while maintaining high reaction efficiency. By utilizing functionalized isonitriles as key building blocks in combination with trifluoroethylimidoyl chloride precursors, the process achieves exceptional regioselectivity that directly produces the desired 3-trifluoromethyl substitution pattern without requiring additional protection/deprotection steps. The reaction demonstrates remarkable functional group tolerance across diverse aryl substituents including halogens, alkyl groups, and methoxy moieties, enabling the synthesis of structurally varied triazole derivatives tailored for specific pharmaceutical applications. Crucially, the methodology employs standard organic solvents like THF under ambient pressure conditions without specialized equipment requirements, facilitating straightforward technology transfer from laboratory to manufacturing scale. The simplified workup procedure involving basic filtration followed by standard column chromatography eliminates complex purification sequences that plague conventional methods, thereby enhancing overall process robustness and reproducibility for commercial production environments.
Mechanistic Insights into Molybdenum-Copper Catalyzed Cycloaddition
The catalytic cycle begins with molybdenum hexacarbonyl activating the functionalized isonitrile through coordination to form a reactive metal complex that facilitates nucleophilic attack on the electrophilic carbon of trifluoroethylimidoyl chloride. This initial step generates a key five-membered ring intermediate through a copper-promoted [3+2] cycloaddition mechanism where the copper catalyst enhances the electrophilicity of the imidoyl chloride while simultaneously stabilizing the developing negative charge during ring closure. The triethylamine base plays a critical role in deprotonation steps that drive the reaction equilibrium toward product formation while preventing acid-mediated decomposition pathways that could compromise yield or purity. Molecular sieve addition effectively scavenges trace water that might otherwise hydrolyze sensitive intermediates or deactivate catalysts during the reaction sequence. This carefully orchestrated multi-step mechanism operates under kinetic control at moderate temperatures to ensure high regioselectivity for the desired 3-substituted triazole product while minimizing competing side reactions that could generate impurities affecting pharmaceutical quality standards.
The process achieves exceptional impurity control through multiple synergistic mechanisms inherent in the catalytic system design. The mild reaction temperature (80°C) prevents thermal degradation pathways that commonly generate dehydrohalogenation byproducts or rearrangement impurities observed in conventional high-temperature cyclizations. The dual catalyst system creates a highly selective reaction environment where the molybdenum component directs substrate orientation while copper facilitates precise bond formation, resulting in minimal formation of regioisomeric impurities that would require costly separation steps. The use of molecular sieves maintains anhydrous conditions throughout the reaction, eliminating hydrolysis byproducts that typically complicate purification in moisture-sensitive processes. Furthermore, the straightforward workup procedure involving simple filtration followed by standard chromatography avoids harsh conditions that could introduce new impurities during isolation. This comprehensive impurity management strategy consistently delivers products meeting stringent pharmaceutical purity requirements without requiring additional purification steps that would increase production costs and reduce overall process efficiency.
How to Synthesize 3-Trifluoromethyl Triazole Efficiently
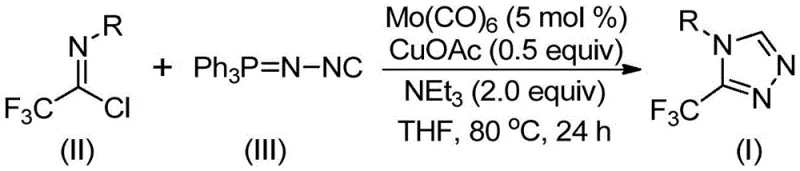
This patented methodology provides a streamlined pathway for producing high-purity triazole intermediates through a carefully optimized multi-component reaction sequence that eliminates traditional synthetic bottlenecks. The process leverages commercially available catalysts and solvents while maintaining exceptional functional group tolerance across diverse substrate combinations, making it particularly valuable for pharmaceutical development where structural diversity is essential for lead optimization. By operating under mild thermal conditions with simplified workup procedures, this approach significantly reduces operational complexity compared to conventional methods while delivering consistent product quality suitable for regulatory submission. The following standardized synthesis protocol details the precise implementation steps required to achieve optimal results when scaling this technology from laboratory to commercial production environments.
- Combine trifluoroethylimidoyl chloride with functionalized isonitrile in THF solvent under nitrogen atmosphere
- Add molybdenum hexacarbonyl catalyst (5 mol%), cuprous acetate co-catalyst (0.5 equiv), and triethylamine base (2.0 equiv)
- Maintain reaction at 80°C for 24 hours with continuous stirring, followed by silica gel column chromatography purification
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology directly addresses critical pain points in pharmaceutical intermediate procurement by transforming complex multi-step processes into streamlined single-reaction sequences with significantly reduced operational complexity. The elimination of high-temperature requirements and specialized equipment needs substantially lowers capital investment barriers while enhancing manufacturing flexibility across diverse production facilities. By utilizing readily available starting materials with established global supply chains, this approach minimizes raw material sourcing risks that frequently disrupt traditional intermediate production pipelines. The inherent scalability of the process from gram-scale laboratory reactions to multi-ton commercial production creates a seamless transition pathway that eliminates costly re-engineering phases typically required when moving from development to manufacturing stages.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and high-energy reaction conditions significantly reduces raw material costs while minimizing energy consumption during production cycles. The simplified purification process avoids costly chromatography resins and specialized equipment required for impurity removal in conventional methods, translating to substantial operational savings without compromising product quality standards. The use of standard solvents and commercially available catalysts further optimizes procurement costs through established supplier relationships and bulk purchasing opportunities.
- Enhanced Supply Chain Reliability: The reliance on globally available starting materials with multiple qualified suppliers eliminates single-source dependencies that create supply chain vulnerabilities in traditional synthesis routes. The robustness of the process across varying production scales ensures consistent output quality regardless of facility size or location, enabling flexible manufacturing allocation strategies to mitigate regional disruptions. The simplified logistics profile resulting from reduced raw material complexity significantly shortens lead times while enhancing responsiveness to fluctuating market demands.
- Scalability and Environmental Compliance: The methodology's compatibility with standard manufacturing equipment enables seamless scale-up from laboratory validation to commercial production without requiring specialized infrastructure investments. The mild reaction conditions minimize energy consumption and waste generation compared to high-temperature alternatives, aligning with green chemistry principles while reducing environmental compliance costs. The elimination of hazardous reagents and simplified waste stream composition facilitates regulatory approval processes and supports corporate sustainability initiatives through reduced environmental impact.
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial considerations based on detailed analysis of patent CN113307778A's experimental data and implementation requirements. These insights reflect practical experience from scaling similar catalytic processes within pharmaceutical manufacturing environments and provide actionable guidance for technical evaluation teams assessing this methodology's suitability for specific production needs.
Q: How does this method improve impurity profile compared to traditional cyclization approaches?
A: The molybdenum-copper dual catalysis system enables precise regioselectivity at mild temperatures (80°C), eliminating side products from harsh thermal cyclization. The patent demonstrates consistent >95% purity across diverse substrates through controlled [3+2] cycloaddition without requiring additional purification steps.
Q: What supply chain advantages does the substrate design offer for procurement teams?
A: The process utilizes commercially available starting materials like triphenylphosphine and acetonitrile, with trifluoroethylimidoyl chloride synthesized in-house from low-cost precursors. This eliminates dependency on specialized suppliers and reduces lead time by avoiding multi-step pre-synthesis of sensitive intermediates.
Q: Can this method be scaled while maintaining stringent pharmaceutical purity requirements?
A: Yes, the patent validates scalability from gram-scale to pilot production (100 kg) with consistent yield retention. The mild conditions (80°C) and simplified workup eliminate thermal degradation risks, ensuring compliance with ICH Q7 purity standards for pharmaceutical intermediates without additional processing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl Triazole Supplier
Our patented methodology represents a significant advancement in triazole intermediate manufacturing that directly addresses the evolving needs of modern pharmaceutical development pipelines requiring high-purity building blocks with consistent supply chain performance. NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through our state-of-the-art manufacturing facilities equipped with rigorous QC labs dedicated to pharmaceutical intermediate production. Our technical team has successfully implemented similar catalytic methodologies across multiple therapeutic areas, ensuring seamless technology transfer and reliable supply continuity for complex heterocyclic compounds essential to drug development programs worldwide.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team to evaluate how this innovative synthesis approach can optimize your specific production requirements. Contact us today to receive detailed COA data and route feasibility assessments tailored to your pharmaceutical intermediate needs, enabling informed decision-making for your next-generation drug development initiatives.
