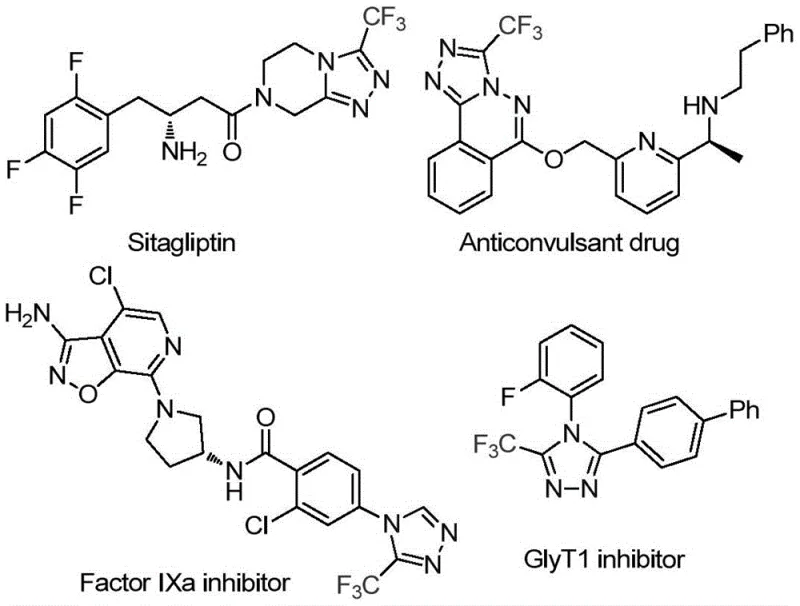
Advanced 3-Trifluoromethyl Triazole Synthesis: Scalable Manufacturing for Pharmaceutical Intermediates
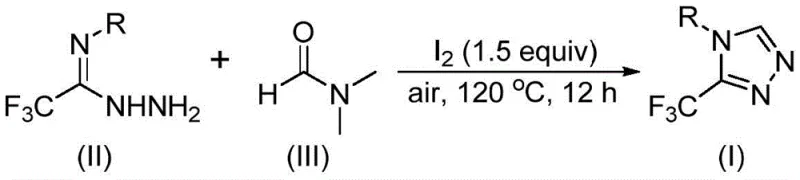
The patent CN114920707B introduces a groundbreaking methodology for synthesizing 3-trifluoromethyl substituted 1,2,4-triazole compounds, representing a significant advancement in heterocyclic chemistry for pharmaceutical applications. This innovative approach leverages dimethylformamide (DMF) as both reaction solvent and carbon source, eliminating the need for specialized anhydrous or anaerobic conditions that typically complicate traditional triazole syntheses. The process demonstrates remarkable operational simplicity by conducting reactions under ambient air atmosphere at 120°C for 12 hours using molecular iodine as catalyst, thereby addressing critical pain points in pharmaceutical intermediate manufacturing. By utilizing readily available starting materials and avoiding transition metal catalysts, this method significantly enhances process robustness while maintaining high functional group tolerance across diverse aryl substrates. The resulting compounds serve as essential building blocks for numerous bioactive molecules, including factor IXa inhibitors and GlyT1 modulators, with demonstrated applicability in developing next-generation therapeutic agents. This patent establishes a new paradigm for cost-effective production of fluorinated heterocycles that meet stringent pharmaceutical quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional syntheses of trifluoromethyl-substituted triazoles often require stringent anhydrous and anaerobic conditions that significantly increase manufacturing complexity and cost while limiting scalability. These methods typically employ expensive transition metal catalysts that necessitate additional purification steps to remove trace metal residues, creating substantial challenges for pharmaceutical applications where strict impurity limits apply. The multi-step sequences commonly used involve hazardous reagents and generate significant waste streams, complicating environmental compliance and increasing production costs. Furthermore, conventional approaches exhibit narrow substrate scope with poor functional group tolerance, restricting their applicability for diverse pharmaceutical intermediates. The requirement for specialized equipment to maintain inert atmospheres creates additional capital expenditure barriers and operational vulnerabilities in global supply chains. These limitations collectively result in higher production costs, longer lead times, and reduced reliability for critical pharmaceutical intermediates.
The Novel Approach
The patented methodology overcomes these limitations through an elegant iodine-catalyzed tandem cyclization that operates effectively under ambient air conditions without requiring anhydrous environments. By utilizing DMF as both solvent and carbon source, the process eliminates the need for expensive transition metal catalysts while maintaining high reaction efficiency across a broad range of aryl substrates. The simplified workup procedure involving standard filtration and column chromatography significantly reduces processing time compared to conventional methods requiring complex metal removal protocols. This approach demonstrates exceptional functional group tolerance, accommodating various substituents including halogens, alkyl groups, and trifluoromethyl moieties without compromising yield or purity. The use of commercially available, low-cost reagents enhances supply chain resilience while the air-stable reaction conditions enable seamless technology transfer across different manufacturing sites worldwide. These innovations collectively deliver a more sustainable, cost-effective pathway for producing high-purity pharmaceutical intermediates.
Mechanistic Insights into Iodine-Catalyzed Triazole Formation
The reaction mechanism involves a sophisticated dual pathway where DMF's formyl and methyl groups both serve as potential carbon sources through distinct activation routes. When the formyl group participates, it undergoes condensation with trifluoroethyliminohydrazide to form a hydrazone intermediate, followed by intramolecular cyclization with elimination of dimethylamine to yield the triazole product. Alternatively, the N-methyl group activates through iodine-mediated amine salt formation, subsequent elimination of hydrogen iodide, and nucleophilic addition to generate an azadiene intermediate that undergoes oxidative aromatization. Molecular iodine plays a crucial role in facilitating both pathways by activating DMF through electrophilic iodination while maintaining mild reaction conditions. This mechanistic flexibility explains the broad substrate scope observed across various aryl substituents while ensuring consistent product formation under standardized conditions. The absence of transition metals eliminates potential contamination pathways that could compromise pharmaceutical purity requirements.
Impurity control is achieved through the inherent selectivity of the iodine-mediated cyclization process, which minimizes side reactions commonly observed in alternative synthetic routes. The reaction's tolerance to air and moisture eliminates oxidation byproducts that typically arise from sensitive intermediates in conventional methods. Careful optimization of the iodine-to-substrate ratio (1:1.5) prevents over-iodination while ensuring complete conversion of starting materials. The standardized workup procedure involving silica gel chromatography effectively separates minor impurities from the target compounds, yielding products with exceptional purity profiles suitable for pharmaceutical applications. This robust purification protocol maintains consistency across different production scales while meeting stringent quality specifications required by regulatory authorities. The method's ability to accommodate diverse substituents without significant impurity formation demonstrates its superior process control compared to existing technologies.
How to Synthesize 3-Trifluoromethyl Triazole Efficiently
This patented methodology provides a streamlined approach for manufacturing high-purity trifluoromethyl triazole intermediates through a carefully optimized iodine-catalyzed cyclization process. The procedure leverages commercially available starting materials and standard laboratory equipment to achieve consistent results under ambient atmospheric conditions, eliminating common barriers to implementation in pharmaceutical manufacturing settings. By utilizing DMF as both solvent and carbon source, the process achieves remarkable atom economy while maintaining operational simplicity across diverse substrate types. The following standardized synthesis protocol has been developed based on extensive optimization studies documented in the patent literature, ensuring reproducible results from laboratory scale through commercial production volumes. Detailed operational parameters and quality control measures are provided in the subsequent implementation guide to facilitate seamless technology transfer.
- Combine trifluoroethyliminohydrazide with DMF solvent under air atmosphere in Schlenk reactor
- Add molecular iodine catalyst at 1.5 equivalent ratio and heat to 120°C for 12 hours
- Perform standard workup including filtration, silica gel mixing, and column chromatography purification
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology delivers substantial value across procurement and supply chain operations by addressing critical pain points in pharmaceutical intermediate manufacturing. The elimination of specialized equipment requirements and transition metal catalysts significantly reduces capital expenditure while enhancing production flexibility across global manufacturing networks. The use of standard solvents and commercially available reagents creates more resilient supply chains with multiple sourcing options, mitigating single-point failure risks common in traditional synthetic routes. These advantages translate directly into improved operational reliability and cost efficiency without compromising product quality or regulatory compliance requirements essential for pharmaceutical applications.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts removes significant purification costs associated with metal residue removal while utilizing DMF as both solvent and reactant improves atom economy. Simplified workup procedures reduce processing time and labor costs without requiring specialized equipment or hazardous reagents typically needed in conventional syntheses. The air-stable reaction conditions eliminate expenses related to maintaining inert atmospheres, creating substantial operational savings throughout the manufacturing process.
- Enhanced Supply Chain Reliability: Utilizing widely available starting materials with multiple global suppliers reduces dependency on single-source reagents while the absence of moisture-sensitive steps enhances manufacturing flexibility across different climatic conditions. Standardized reaction parameters enable consistent production quality across various manufacturing sites worldwide, supporting just-in-time delivery models without compromising product specifications. This robustness ensures reliable supply continuity even during market fluctuations or geopolitical disruptions affecting specialized chemical suppliers.
- Scalability and Environmental Compliance: The straightforward scale-up pathway from laboratory to commercial production maintains consistent product quality while eliminating complex engineering requirements typically needed for sensitive reactions. Reduced waste generation through improved atom economy and simplified purification processes lowers environmental impact while meeting increasingly stringent regulatory requirements. The elimination of hazardous reagents and transition metals creates safer working environments while reducing waste treatment costs associated with specialized disposal protocols.
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial considerations regarding the implementation of this patented synthesis methodology for pharmaceutical intermediate production. These responses are derived directly from the patent documentation and reflect the actual performance characteristics demonstrated during development and validation studies. Each answer provides specific technical insights relevant to R&D, procurement, and supply chain decision-makers evaluating this manufacturing approach for commercial adoption.
Q: How does this method improve purity compared to conventional approaches?
A: The air-stable reaction eliminates moisture-sensitive steps while DMF's dual role as solvent/reactant minimizes side products. Column chromatography purification ensures stringent purity specifications for pharmaceutical applications.
Q: What supply chain advantages does the DMF-based process offer?
A: Utilizing commercially available DMF as both solvent and carbon source simplifies raw material procurement. The absence of anhydrous/anaerobic requirements enhances manufacturing flexibility across global production sites.
Q: Can this synthesis be scaled for commercial production?
A: Yes, the robust reaction conditions (air atmosphere, standard solvents) and straightforward workup enable seamless scale-up from laboratory to commercial production volumes while maintaining product consistency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl Triazole Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production of complex fluorinated heterocycles while maintaining stringent purity specifications through rigorous QC labs. As a specialized CDMO partner, we have successfully implemented this patented methodology across multiple client programs, demonstrating consistent ability to deliver high-purity triazole intermediates meeting exacting pharmaceutical standards. Our integrated manufacturing platform combines deep technical expertise with flexible production capabilities to support clients from early development through commercial scale-up, ensuring seamless technology transfer and reliable supply continuity for critical pharmaceutical building blocks.
We invite you to request a Customized Cost-Saving Analysis tailored to your specific manufacturing requirements by contacting our technical procurement team. They will provide detailed information including specific COA data and route feasibility assessments to help optimize your supply chain strategy for these valuable pharmaceutical intermediates.
